8
Oct
2024

Stevanato Group SPA
Exhibitor at CPHI Milan 2024 stand 20C1, Packaging and Drug Delivery
About Us
Categories

-
IT
-
2015On CPHI since
-
5000+Employees

Company types
Primary activities
Event information
CPHI Milan 2024
-
08 Oct 2024 - 10 Oct 2024
-
Fiera Milano, Italy
-
Visit us at stand 20C1, Packaging and Drug Delivery
Products Featured at CPHI Milan 2024
-
Product Vertiva™ on-body delivery system platform
Vertiva™ is an on-body delivery system platform composed of a single-use pod with pre-filled and pre-loaded cartridge and a multi-use, smart controller. The device is able to deliver micro-precision basal doses and full-content bolus injections, resulting in a highly flexible,... -
Product Alba®: a breakthrough solution for biologics
Alba® represents a best-in-class solution to address the development of sensitive biologics, such as highly concentrated drugs prone to silicone aggression and ophthalmic drugs. Medicines are particularly sensitive to all the components of the primary packaging they are in contact with,... -
Product EZ-fill® Platform
Many Pharmaceutical companies are looking for ways to lower their footprint and reduce total costs by relying on experienced external partners for non-core activities. When it comes to containment solutions for biopharmaceutical products, no solution matches SG EZ-fill®, whose proven advan... -
Product Aidaptus® auto-injector
Owen Mumford's device expertise is joining forces with Stevanato Group's unique engineering and manufacturing experience. The collaboration brings the full value of the Aidaptus® auto-injector platform to the market, helping patients self-administer their therapy using a simple and easy-to-use device. ... -
Product MAVIS Platform
MAVIS Platform is our latest innovative suite for best-in-class inspection results.Developed to match every drug perfectly, MAVIS Platform guarantees easy and accessible operations for maintenance and control. It combines advanced inspection performances with a small footprint.
Platform Highligh... -
Product Nexa Flex™: Pre-sterilized Polymer Syringe
Extremely versatile, outstanding cosmetic quality, high degree of break resistance and glass-like transparency, Nexa FlexTM is the perfect answer when looking for polymer syringes.
Available as 1mL long*, Nexa FlexTM is delivered in EZ-fill®... -
Product Pen Injector Assembly Line
Thanks to the modularity, we can provide tailor-made solutions designed to deliver high-quality standards even when scaling-up.Our Pen Injector Assembly Line is an example of a flexible mid-high speed pen injector line assembling different formats and integrating extensive in-process controls.
... -
Product Vision Robot Unit (VRU)
We present a modular, robotic, proof-of-concept system for inspecting parenterals. By employing artificial intelligence principles and applying the technology to the inspection task, we engineered the Vision Robot Unit (VRU).
The VRU is a reliable, autonomous island machine, capable o... -
Product Analytical and Testing Services
With our Technology Excellence Centers (TEC) we can offer analytical and device testing services to support you all the way, from early-stage to launched combination product.
2 locations, 1 goal: assisting customers to anticipate challenges and help you navigate the regulatory landscape.
q... -
Product EZ-fill Smart™
Leveraging our market-leading EZ-fill® technology, we developed EZ-fill Smart™: an enhanced version of our pre-sterilized platform aiming to make RTU vials a standard, ultimately increasing patient safety. With EZ-fill Smart™ we provide you with a highly standardized platform, featuring increased qual... -
Product Alina® - Pen Injector
Alina® is a variable-dose pen injector developed by Stevanato Group based on Axis-D intellectual property and technology exclusively licensed from pen injector device expert Haselmeier, for the treatment of diabetes, obesity, and other therapeutic areas.
Alina® features an appealing and functiona... -
Product SG EZ-fill ISS - Integrated Safety System
The continuing desire to reduce needlestick injuries led us to develop the SG EZ-fill Integrated Safety System. This groundbreaking, fully passive needle shield comes ready to fill and requires no additional assembly steps. SG ISS is both easy to use and intuitive to handle. Activating the automatic shield... -
Product SG EZ-fill ITC - Integrated Tip Cap
The Integrated Tip Cap has been developed to be the ideal match for SG EZ-fill(R) luer lock cone syringes with its twist-off closure system. The Integrated Tip Cap comprises of a rubber component inserted in a rigid plastic cap, screwed onto a luer lock adaptor and then pre-assembled on the SG EZ-fill syri... -
Product SG Nexa 3x Cartridges
Stevanato Group has launched SG Nexa 3x Cartridges, a full range of products that offer up to 3 times the mechanical resistance when compared to a standard glass container. Thanks to an external anti-friction and anti-scratches silicone coating, SG Nexa 3x better withstands the stresses of fill/finish proc...
Stevanato Group S.p.A. Resources (12)
-
Whitepaper Meeting Quality Demands Through Integrated Products and Services
Lars Keinicke Hansen, Chiara Mussoi, and Odra Pinato, all of Stevanato Group, describe how the streamlining of processes and harmonisation of products and services can better serve pharmaceutical companies. A case study highlights how a unique combination of expertise in automation and glass primary packaging benefited pharma giant Merck Serono in a recent project. -
Video Modular and flexible Assembly Equipment
Assembly Equipment combining transport platforms and process modules: an offer of manual, semi-automatic or fully automatic assembly lines customized to your device program. -
Video Pen Injector Assembly Line
A flexible mid-high speed pen injector line assembling different formats and integrating extensive in-process controls. -
Brochure SG Alina : A user-friendly disposable pen injector platform for diabetes care
SG Alina is a variable-dose and fixed-dose and pen injector offered by Stevanato Group in collaboration with Cambridge Design Partnership.
It is based on Axis-D intellectual property and technology exclusively licensed from pen injector device expert Haselmeier, for the treatment of diabetes.
-
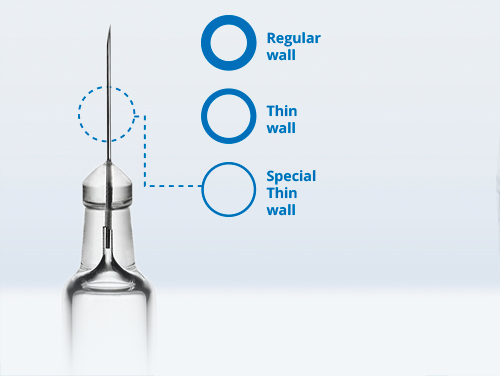
Brochure Innovative Needle Geometry Accommodating Viscous Biologics
In this article, Stevanato Group discusses the challenges faced in administering highly viscous biologics to patients via subcutaneous injection and how its special thin-wall needles can tackle them. -
Whitepaper mRNA therapies on the rise
Learn more from this White Paper providing insights on tests and solutions performed to overcome mRNA challenges. -
Brochure A forensic approach for combination devices
Discover how Stevanato Group helps pharma companies identify relevant testing to de-risk the product development lifecycle and quickly uncover the root causes of product failures. -
Brochure Vertiva™️, Next-generation On-Body Delivery System
In this article, Stevanato Group, introduces Vertiva™️, the company’s next-generation on-body delivery system platform, and discusses how it meets the needs of pharma companies looking to deliver large-volume doses in a patient-centric manner. -
Video Early stage considerations for the manufacture and delivery of vaccines webinar
The COVID-19 pandemic has placed a spotlight on the readiness and requirements of pharma packaging companies to support the distribution and delivery of a potential vaccine and selecting the most suitable primary packaging options for drug products is one of the most crucial considerations in the drug development process, according to industry experts speaking in this CPHI China Virtual Expo Connect Webinar, sponsored by Stevanato Group. Daniel Martinez, Product Manager, Stevanato Group pointed out that speed to market is a primary consideration for vaccine developers, particularly in the current pandemic environment, and keeping the same type of container is essential to reaching this target. “The advantage of using a vial is that it is a pretty versatile container – it’s good for liquids, it’s good for lyophilized products, powders and cryogenic,” he told the audience, noting that some of the COVID-19 vaccines being developed need to be stored at -70 degrees. Watch the webinar to hear more from Daniel and experts from NRL Enterprise Solutions & IQVIA. -
Video Crystal Clear - The Rise of Pharmaceutical Glass Packaging Solutions
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 Glass primary packaging solutions offer safety, stability, recyclability and protect drug integrity. The glass packaging market is predicted to grow exponentially in the next few years, driven in part by the huge demand for glass vials for COVID-19 vaccines. What are the emerging trends in this sector? Which ones are here to stay, or which will prove to be transitory? Unprecedented packaging demands: the impact of COVID19 on the glass packaging market Glass primary packaging to answer customers’ requirements Smart Containers: A new vision ahead for Pharmaceutical Packaging How can pharma manufacturers prepare for future trends? -
Webinar CPHI Webinar Series Optimising Pharma Visual Inspection - Unlocking the Potential of AI
Artificial Intelligence (AI) has triggered a fundamental shift within the innovation paradigm for the pharmaceutical industry and has been implemented in almost every aspect of the value chain, from drug discovery and development to manufacturing. One application of AI which offers particular value is the realm of visual inspections, which present a challenging step in the packaging process. This is particularly true for products with complex characteristics where manual inspections can sometimes fall short, leading to errors and inconsistencies. AI learning models can optimise the packaging process by detecting minute errors, resulting in fewer rejects, less intervention and a more efficient process overall. This webinar will tackle some of the key drivers and benefits to implementing AI for pharma visual inspections to enhance performance, improve accuracy, efficiency and overall quality. Why attend? Common challenges encountered with Pharma Visual Inspections Key Drivers and Benefits to Implementing AI for Pharma Visual Inspections Case Study: Enhancing Pharma Visual Inspection Processes with AI Validation of Artificial Intelligence (AI)-Based Systems WATCH FOR FREE -
Webinar Collaborating to Simplify Device Development and Optimize Glass Solutions for Auto-Injectors
Advances in auto-injector design now provide the option to use both 1ml and 2.25mL syringes plus employ technology to adapt to varying fill volumes and viscosities all whilst maintaining the same device form. This provides a solution for the formulation changes that take place during drug development, clinical trials, and life cycle management. When looking at the performance of any parenteral injection device, the drug containment systems play a crucial role. Auto-injector performance and functionality is strictly linked to key dimensional tolerances of glass syringes. We will illustrate the importance of syringe design to ensure these requirements and how monitoring dimensional parameters at an early stage of the development is key to ensure the intended use and ultimately work to preserve patients’ safety. Benefits of tight collaboration between device designer and manufacturer will also be discussed.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance