- Home
- Biopharmaceuticals
- Biopharmaceuticals (general category)
CPHI Online is the largest global marketplace in the pharma ingredients industry
-
Products551,744
-
Companies7,781
-
Articles11,636
-
Events8
-
Webinars342
Biopharmaceuticals (general category)
Biopharmaceuticals (general category) Companies (100)
Biopharmaceuticals (general category) News
-
News First offers for pharma from Medicare drug price negotiations
Ten high-cost drugs from various pharma manufacturers are in pricing negotiations in a first-ever for the US Medicare program. President Biden’s administration stated they have responded to the first round of offers.5 Apr 2024 -
News Special Edition Women in Pharma: Women’s health and the pharmaceutical industry
International Women’s Day is an annual celebration of women around the world every March 8. March also marks Women’s History Month. This March, our Women in Pharma series takes a look at how women’s healthcare has developed, and the r...25 Mar 2024 -
News Informa Markets International Women’s Day Panel Discussion
On March 7, 2024, the Informa Markets Amsterdam office hosted an International Women’s Day Breakfast and Panel Discussion to celebrate the women who drive the B2B events industry forward, including members of the CPHI team.8 Mar 2024 -
News Innovative Salts for Biopharma
With more than 135 years of experience, Dr. Paul Lohmann® is an expert in high quality Salt manufacturing and provides DPL-BioPharm Salts for upstream, downstream and fill & finish processes of the biopharmaceutical industry.24 Aug 2023 -
News Humira biosimilars at discounted prices to meet market demand for accessible treatment
Several biosimilars to AbbVie’s monoclonal antibody therapeutic Humira were launched by various pharmaceutical giants this week in the US, with more expected to be released by the end of the year as the patent expires.3 Jul 2023 -
News Biogen receives accelerated approval for first-of-its-kind ALS treatment
Biogen has announced that they have received approval from the US FDA for the amyotrophic lateral sclerosis (ALS) treatment, QALSODY™ (tofersen). This is the first approval of a treatment that targets a genetic cause of ALS.26 Apr 2023 -
News Eli Lilly gets ready to launch five new drugs in 2023
Eli Lilly, the American pharmaceutical company (IN, USA) are gearing up for a big year ahead, with hopes to launch five new drugs and capitalise on growing obesity and Alzheimer’s disease markets.15 Dec 2022 -
News From the floor - CPHI Frankfurt 2022
The live blog from the people on the ground at CPHI Frankfurt - covering all stand-out news, memorable moments, and keynote updates from the 2022 event.1 Nov 2022 -
News LG Chem to acquire AVEO Oncology with USD $566 million investment
South Korean chemical company LG Chem is set to acquire AVEO Oncology, an oncology-focused biopharmaceutical company based in the USA, as part of an investment to increase its footprint in North America and expand its development and commercialisation ...18 Oct 2022 -
News Thermo Fisher Scientific expands viral vector manufacturing capabilities with Plainville facility
Thermo Fisher Scientific’s new Plainville, MA viral vector manufacturing facility has opened, expanding the company's cell and gene therapy capabilities with advancing bioproduction and analytical instrumentation technologies.25 Aug 2022 -
News Lonza adds X-ray powder diffraction to micronisation portfolio in Monteggio (CH)
Lonza expands solid-state characterisation services for polymorph and solid-state properties of APIs with the addition of X-ray powder diffraction to its portfolio.25 Aug 2022 -
News Establishment of antiviral development centre for pandemic-level viruses
Researchers across several US institutions will come together to establish a centre for the research and development of antiviral drugs and therapeutics against pandemic-level viruses.10 Aug 2022 -
News Thermo Fisher Scientific announce expansion of cell culture media manufacturing site
The expansion of Thermo Fisher Scientific’s Grand Island, New York cell culture media manufacturing site offers increased redundant capacity in support of vaccine and biologic therapy development and manufacturing.5 Aug 2022 -
News mRNA production partnership to scale-up COVID-19 vaccine development
GreenLight Biosciences and Samsung Biologics announce mRNA production partnership to scale mRNA vaccine production to a commercial level with Samsung's expanded mRNA drug substance manufacturing site.3 Aug 2022 -
News The Japanese Pharma Market – Top Four Trends Shaping the Sector
Drug pricing, generics, investment, innovation - how these trends are shaping the face of Japanese pharma20 Jul 2022 -
News Investors turn attention to biotechs with clear path to market
Companies that are further down the road to market are more attractive to investors than those in early development29 Jun 2022 -
News Samsung Biologics fully acquires Samsung Bioepis for $2.3B
Samsung Biologics has full ownership over Samsung Bioepis after purchasing Biogen’s stake22 Apr 2022 -
News Asahi Kasei Medical to acquire US-based CDMO Bionova Scientific
Asahi plans to use the CDMO to propel its bioprocess business19 Apr 2022 -
News TraceLink to launch AI solution for predicting drug shortages
TraceLink’s Product Availability Intelligence will enable stakeholders to predict drug shortages months in advance5 Apr 2022 -
News KKR-backed Biosynth Carbosynth acquires Vivitide
KKR closes its previously-announced acquisition of Biosynth Carbosynth and announces Biosynth's acquisition of vivitide.18 Mar 2022
Biopharmaceuticals (general category) Products (169)
-
Product MAST® Autosampling Solution
A Versatile & Modality-Agnostic Aseptic Autosampling Solution for Reliable On-Line and Near Real-Time Sample Analysis
-
Product Mobius® Gold High Cell Density Cryopreservation Assemblies (-80 °C)
A single-use assembly designed to facilitate the freeze and thaw of cryopreserved cells at -80 °C. Mobius® Gold High Cell Density Cryopreservation Assemblies enable seed train inoculation with cells at a higher cell density and larger volume, which significantly accelerates the cell expansion process. The ...
-
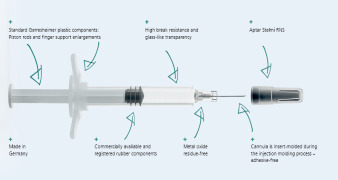
Product Gx RTF® ClearJect® Polymer syringes
Gx RTF® ClearJect® syringes are made from COP (Cyclo Olefin Polymer). They are glue-free as the cannula is not glued in, but is instead insert-molded during the injection molding process, no aggregation due to metal or tungsten residues, high viscosity silicone oil for reduced particles, minimal extractabl...
-
Product Biologic drug substance CDMO services
From pre-clinical development to commercial supply, Patheon by Thermo Fisher Scientific is an industry leader in the development and manufacture of mammalian cell culture drug substances. Patheon offers biotech and pharmaceutical companies the ability to pursue opportunities around the globe with a fully i...
-
Product Bioinformatics
We offer integrated bioinformatics services to biotech and pharmaceutical companies across the drug discovery pipeline. Our bioinformatics services span the entire gamut from building data packages, pathway databases and knowledge bases, to analysis of high throughput data, predictive modeling, structural ...
-
Product Clinical Development
Syngene’s Clinical Development services group supports early phase through late phase clinical trials across a wide range of therapeutic areas.Our Clinical Development Services includes clinical trial management, PK Analysis/Bioanalytical - Small and large molecules, Central lab, Biometrics & Clinical ...
-
Product Data System Integration
Data System Integration
Catalyx offers comprehensive data analytics solutions and integration for organizations, addressing the increasing demand for efficient data management. Our expertise covers a wide spectrum of data-related areas, from batch variable data to integration with various enterp...
-
Product Biologic Quality Control and Release Testing
We deliver responsive QC analysis for complex biologic products from our cGMP laboratories. Our scientists develop and validate methods or perform technology transfer of a sponsor's method for a wide range of analytical methods required for batch release testing. We also routinely carry out testing to Phar...
-
Product Biopharmaceutical Analysis
Solvias has a solid reputation with market-leading biopharmaceutical companies for the characterization and QC analysis of monoclonal antibodies (mAbs), glycoproteins, PEGylated proteins and peptides and biosimilars (follow-on biologics).. This means that businesses of all sizes, even smaller biotechs, can...
-
Product Sodium Hyaluronate Excipients Grade
Sodium Hyaluronate Excipients Grade has the registration of pharmaceutical excipients and with high stability. It has the advantages of ultra-low impurity level, ultra-low endotoxin level, ultra-low metal impurity residue and ultra-low microbial pollution.
• Microbial Fermenta...
-
Product Supply Chain Management
• GMP Compliant Storage of a wide range of client products from API and excipients to consumables as well as both Stability and Retention sample programs. • Facility and Staff certified for storage, handling, and shipment of hazardous materials. Including Flammable, Toxic, Potent, etc. products. • Envir...
-
Product Tannic acid
Name: Tannic Acid
Alias: Tannins
CAS #1401-55-4Molecular Formula: C76H52O
Appearance:Light brown irregular powder

-
Product Hoses and Hygenic Connections
Our hoses for pharma biotech, cosmetics, food, beverage and chemical industry
We offer you a wide range of hoses specially designed for the pharmaceutical, biotech, cosmetic, food, beverage and chemical industries. We also ensure their proper use through training in the co...
-
Product Tris Hydrochloride low endotoxin, pharma grade CODE 633654
https://www.itwreagents.com/iberia/en/product/tris-hydrochloride-low-endotoxin-pharma-grade/633654
-
Product Nitroalkanes and derivatives solvents and building blocks for pharmaceutical synthesis
The effectiveness of nitroalkanes lies in their ability to provide alternative synthetic routes to existingcompounds, as well as highly efficient routes to new compounds. The exceptional versatility and highreactivity of nitroalkanes provide a means to conduct synthetic transformations under mild condition...
-
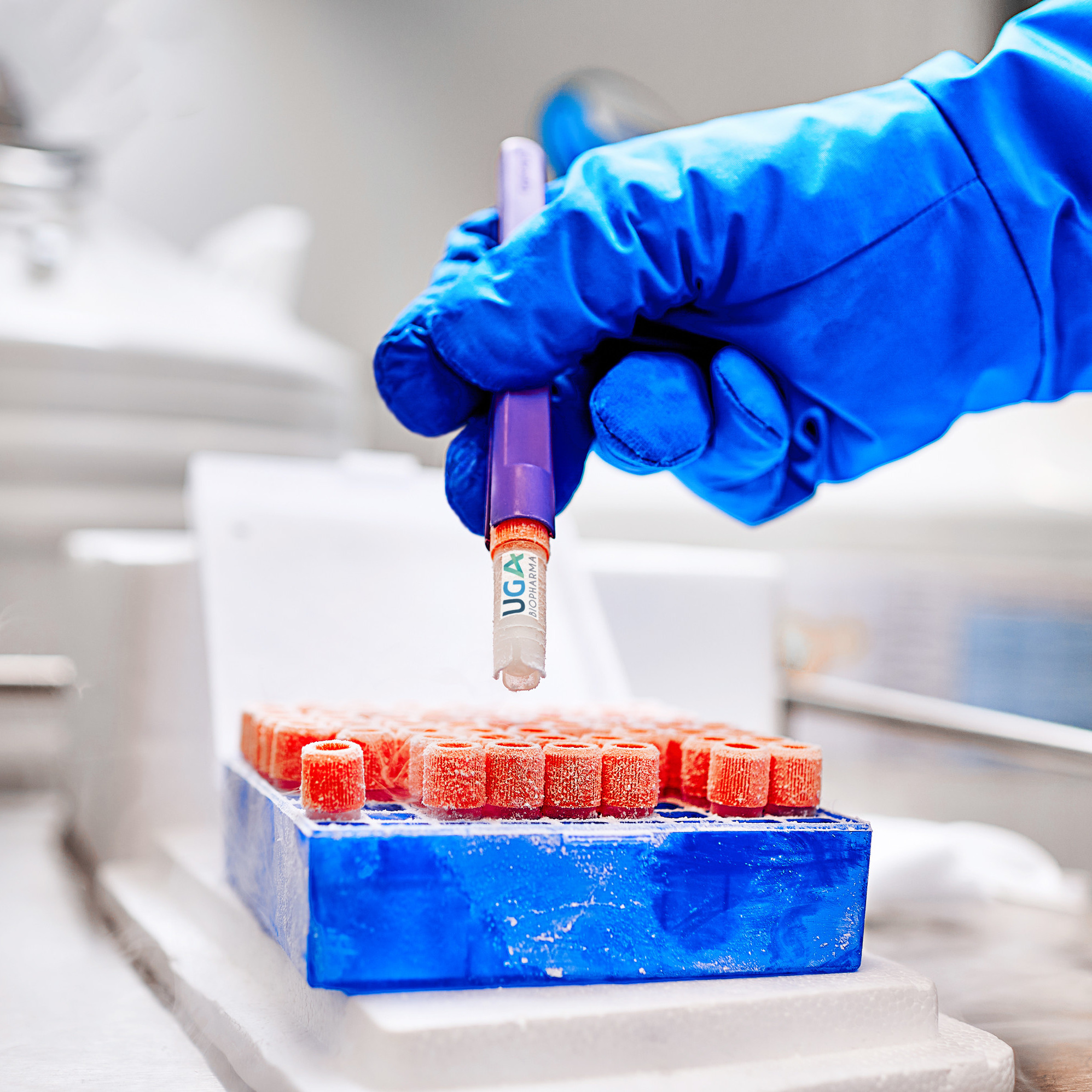
Product Ready-to Use CHO cell lines
UGA Biopharma GmbH offers to their clients so called Ready-to-use Biosimilar Cell Lines. This are in-house developed CHO cell lines stably expressing different biosimilar molecules, that are instantly available for in-licensing. The client profits, because the cell lines are directly available, and develop...
-
Product Sodium Hyaluronate - Injection grade
Hyature® is a pharmaceutical grade sodium hyaluronate which can be used as an API or excipient for drugs and medical devices in ophthalmic preparations, intra-articular injections, anti-adhesive preparations, topical preparations for wound healing and soft tissue filler, etc. Hyature® sodium hyalur...
-
Product Cortellis CMC Intelligence™
Cortellis CMC Intelligence™ product is a comprehensive database of guidelines / granular collection of CMC data requirements for initial registration of small-molecule and biologics drugs around the world that can be used to avoid delays in product approval and successfully bring a drug to the market (laun...
-
Product RoSS®: Protecting single-use bags
Protect your preferred single-use bioprocess containers: RoSS shells - abbreviated for Robust Storage and Shipping - reduce product loss towards 0%. Compatible with all available sizes and types of single-use bags, RoSS enables standardized and scalable end-to-end process solutions for fluid managemen...
-
Product Sterility Test Isolator
1. Built-in Vaporized Hydrogen Peroxide (VHPS) generator for bio-decontamination which can reach 6-log Sterility Assurance Level
2. The VHPS residual concentration is less than 1ppm after aeration
3. leakage rate is less than 0.5% /vol/hour under 2 times working pressure test
qq...
-
Product BioPharm
Caldic is a global, full-service solution provider to the biopharmaceutical market. Our technical expertise, global cGMP manufacturing and distribution capabilities, quality systems, tailored solutions, and extensive product portfolio enable biopharmaceutical customers to accelerate their speed to market o...
-
Product KLEPTOSE® - Betacyclodextrin
KLEPTOSE® Betacyclodextrins are efficient solubilisation, stabilization enhancers and taste-masking agents. A full range is available for use in many dosage forms, from solid to liquid.
-
Product PIPELINE AVAILABLE FOR OUR LICENSING AND SUPPLY
Genovior is a dedicated CMO/CDMO, focusing on providing clients excellent, efficient, one stop services and solutions, from clinical trials (IND/NDA) to commercialization.
Our services include:
1. Process Development
2. Formulation Development
3. Analytical Deve...
-
Product BioHale® Trehalose Dihydrate
BioHale® Trehalose Dihydrate is a non-reducing disaccharide consisting of two glucose molecules which are linked by an α, α-1,1 bond. It is used in the pharmaceutical industry as a stabilizer of biologics.
Trehalose Dihydrate has proven to be highly effective in protecting cell membranes...
-
Product WHEATON Roller Apparatus by DWK
WHEATON® roller apparatus are used for roller bottle cell culture, an established cell culture method and relatively inexpensive way to set up a flexible and scaleable biopharmaceutical operation. In addition to the standard product DWK also offer a wide range of customized options to accommodate the most ...
-
Product Proton drops
Rickets prophylaxis.
DOSAGE8 drops (400 I.U.) per day for a 18-months period.1 drop contains: 50 I.U. of Vitamin D3.
INGREDIENTS
Vitamin D3 (Cholecalciferol).
CHARACTERISTICSMicellar solution in physiological solution easy to be mixed with all liquids or foods. Tasteless...
-
Product Sterile Grade at ISO Class 5 facility
Kikkoman can offer "sterile grade” products, purified through a super purification process at our ISO Class 5 facility, approved by the Japanese Authority.
-
Product Eposis
2000, 3000, 4000, 5000, 6000, 8000, 10000IU / Pre-filled syringe Inj.
rhEPO (recombinant human erythropoietin)
Indication:
Anemia in chronic renal failure
- EposisTM demonstrated significant improvement and safety for patients with anemia in Chronic Renal Failure (CRF).
...
-
Product Icatibant
is a synthetic peptidomimetic drug consisting of ten amino acids, and acts as an effective and specific antagonist of bradykinin B2 receptors. It has been approved in the EU for use in hereditary angioedema, and is under investigation for a number of other conditions in which bradykinin is thought to play ...
-
Product Dopamine hcl
① Low blood pressure regulator
② Antishock agent
③ Raw material for the synthesis of norepinephrine

-
Product Liveo™ Pharmaceutical-Grade Tubing
These high-purity tubings are available in both platinum-cured silicone and thermoplastic elastomer (TPE) options and are designed for ultra-pure fluid transfer applications in pharmaceutical and biotechnological manufacturing processes.
With more than 30 options to choose from, they are t...
-
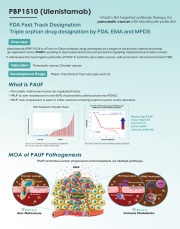
Product PBP1510 (Ulenistamab) Anti-PAUF Antibody for Pancreatic Cancer
Ulenistamab (PBP1510) is a First-in-Class antibody drug developed as a target of pancreatic adenocarcinomaup-regulated factor (PAUF) resulting in decreased autocrine and paracrine signaling implicated pancreatic cancer.
It attenuates the tumorigenic potential of PAUF in both the pancreatic cancer ce...
-
Product Gene Therapy Services
Our gene therapy process development and manufacturing capabilities are designed to confidently and rapidly progress your products from gene to market through development of robust and reproducible cGMP practices.
-
Product Recombinant Trypsin
In compliance with pharmacopoeia standards: in compliance with relevant standards for recombinant proteases in pharmacopoeias.No animal origin: produced by recombination, free of exogenous viral contamination, and no animal-derived materials are used in the production process.
Quality stability: mass pr...
-
Product PharmASep™ Filter Dryer
The PharmASep™ Filter Dryer combines SWECO’s patented separation process with vacuum drying technology to efficiently and effectively filter and dry product while significantly increasing convenience, safety, precision and productivity in critical applications. This documented aseptic pharmaceutical fi...
-
Product Guanidine Thiocyanate Excipient and Intermediate
BioSpectra offers a wide range of products, which includes guanidine thiocyanate. Features: it is a chaotropic agent. Contact us for more information.
-
Product Development Sciences
Our development laboratories are well equipped for conducting a diverse range of experiments. These encompass formulation development, cycle design and process refinement, as well as evaluating finished product. Ease of scale-up is accomplished by completing process development studies within a pilot scale...
-
Product Biological Activity
This test provides a direct measure of the effectiveness of the active ingredient.
-
Product Cellicon® Perfusion Filter and Controller
A Superior Cell Retention Solution for N-1 Seed Train Intensification with Optimized Process Control. The benchtop Cellicon™ Perfusion Solution is designed to meet your perfused seed train challenges. It consists of a controller and a flat sheet cell retention filter with a single-use assembly running in t...
-
Product Cellicon® Cell Retention Solution for Process Scale
Increase Your Bioreactor Productivity with our superior solution for upstream process intensification. In combination with your bioreactor, the Cellicon® Cell Retention Solution allows you to achieve significantly higher viable cell densities, improving your upstream productivity and manufacturing flexibil...
-
Product Ultimus® Film
An innovative single-use film offering strength and leak resistance for your toughest liquid bioprocessing applications. Ultimus® film provides enhanced bag strength, improved durability and leak resistance through a novel strength layer reinforced by woven nylon. The inert fluid contact layer promotes hea...
-
Product Novaseptum® SURe Sterile Sampling Assemblies
A single-use sterile sampling assembly that takes representative samples while protecting your bioprocess from cross-contamination. NovaSeptum® SURe sampling assemblies can be easily integrated into your single-use manufacturing process. Available in five popular container formats and with two connect...
-
Product Mobius iFlex® Bioreactor
The next generation of high performance single-use modular bioreactors for fed-batch and perfusion processes
-
Product ProCellics™ Raman Analyzer with Bio4C® PAT Raman Software
An Easy-to-Use GMP Process Analytical Technology (PAT) Platform to Monitor Cell Cultures In-Line and in Real-Time, from Process Development to Manufacturing
-
Product Pellicon® Capsules with Ultracel® Membrane
Accelerate your therapy using our innovative single-use tangential flow filters with superior flux performance. Ideal for single-use tangential flow filtration (TFF), Pellicon® Capsules provide high flux and linear scalability in a plug ‘n play format for optimum process flexibility and productivity. Pelli...
-
Product MilliStak+® Depth Filters
Innovative, High-Performance Pod Filters are Ideal for Primary and Secondary Clarification in Lab, Pilot and Process-scale Applications. Millistak+® depth filter media is offered in a scalable, disposable format: the Pod Filter System.
-
Product Natrix® Q Chromatography Membrane
Natrix® Q Chromatography Membrane for single-use, scalable purification, Natrix® chromatography membrane is a high capacity, high throughput strong anion exchange device designed to purify biomolecules. It combines the capacity of high performing resins with the exceptional flow properties of chromatograph...
-
Product Millistak+® HC Pro Synthetic Depth Filter
Millistak+® HC Pro (high-capacity synthetic media) is a family of synthetic depth filters providing a cleaner and more consistent depth filtration media over current diatomaceous earth (DE) and cellulose (CE) based filter offerings. Multiple media grades are available for primary and secondary clarificatio...
-
Product Chemical Development
Experience in providing end-to-end integrated services from process design and development to large scale manufacturing
-
Product Monitoring, Notifications, and Alarm Integration
Catalyx is experienced in deploying Monitoring and Notification Systems across a wide variety of industries, bringing cutting-edge technologies for a flexible and customized yet standardized solution. We have developed sophisticated EMS and BAS systems with Alarm Management, including user-based hierarchy ...
-
Product Biosimilars Testing Services
Testing services for biosimilars: Our biosimilar analytical comparability programs provide highly relevant data for early stage characterisation and later stage comparison. These programs evaluate and compare all pertinent features of the biosimilar product and are based on the ICH Q6B. Programs encompa...
-
Product Antibody / Monoclonal antibody therapeutics services
Monoclonal antibody therapeutics: We continually invest in advanced analytical instrumentation that allows us to deliver mAb analytical data with the highest degree of sensitivity, accuracy and resolution. Our experience spans recombinant monoclonal antibodies and related products such as biosimilars, ...
-
Product Protein Aggregation Analysis and Characterization
Analysis and characterization of protein aggregates: The services provided by our experts include a range of protein aggregation analysis techniques that are used to detect and quantify aggregates in solution in support of formulation development, quality control, stability studies, comparability, rel...
-
Product cGMP Cell-based Bioassays
Cell-based bioassays that comply with cGMP: Using a variety of techniques, including cell-based assays, ligand and receptor binding assays, and flexible multiple assay approaches – our potency testing experts offer tailored bioassay method development, method transfer and method validation to ICH Q2 (R1)...
-
Product Oligonucleotide Analytical Development Services
Analytical development services for oligonucleotides: For oligo-based drugs, our capabilities range from GLP bioanalysis to GLP/cGMP characterisation. We support product development, from quality control testing of amidite starting materials and early stage product characterization through to GMP batch r...
-
Product 2,3,4-Trihydroxybenzaldehyde
Name: 2,3,4-Trihydroxybenzaldehyde
CAS No: 2144-08-03
EINECS No.:218-404-2
Chemical Name: Pyrogallolaldehyde
Structure Formula:
Molecular formula&Molecular Weight: C7H6O4; 154.1201
...
-
Product Methyl 3,4,5-trimethoxybenzoate
Name: Methyl 3,4,5-trimethoxybenzoate
CAS No: 1916-07-0
Chemical Name: 3,4,5-Trimethoxybenzoic acid methyl ester
Structure Formula:
Molecular formula&Molecular Weight: C11H14O5; 226.2259
Appearance...
-
Product Pyrogallol
Product Name:Pyrogallol
Alias: Pyrogallic acid, 1, 2.3-trihydroxybenzene
CAS No: 87-66-1
Molecular Formula:C6H6O3;
Molecular Weight:126.11
Appearance: White glossy crystalline powder.
Storage: Moisture-proof and a...
-
Product N-Acetyl-DL-Tryptophan (Ph. Eur, BP) low endotoxin, pharma grade CODE 637763
https://www.itwreagents.com/iberia/en/product/n-acetyl-dl-tryptophan-ph-eur-bp-low-endotoxin-pharma-grade/637763
-
Product HEPES and HEPES Sodium Salt Buffers
Our latest investment in new manufacturing capacity significantly expands our portfolio of in-house-produced biological buffers and moves Advancion into a leading global supply position for the HEPES molecule. HEPES Biologics Plus grade buffer products are produced by Advancion in its Sterlington facility ...
-
Product Sodium Hyaluronate Medical Device Grade
We have different grade like medihya-T, medihya-S,medihya-M,medihya-I to meet customer' requirement. Application in Intra-articular injection, viscosupplementation,dermal filler, eye drops, topical preparation, anti-adhesive agent, medical lubricant, mucosa healingetc.
-
Product Sodium Hyaluronate Injection Grade
Sodium Hyaluronate Injection Grade has GMP certification in China and the status of API registration status is A. The nucleic acid level is 0.01 (A260nm) and bacterial endotoxins is 0.005IU/mg, which are 1/50 of the value specified in the standard of Ph.Eur. And the content of protein is lower than t...
-
Product Sodium Hyaluronate Eye drop Grade
Sodium Hyaluronate Eye-drop Grade has GMP certification in China and the status of API registration status is A. It has the advantages of ultra-low impurity level, ultra-low endotoxin level, ultra-low metal impurity residue and ultra-low microbial pollution.
• Microbial ...
-
Product Hyatrue® — Pharmaceutical Grade Sodium Hyaluronate
Hyatrue® is a pharmaceutical grade sodium hyaluronate which can be used as an API or excipient for drugs and medical devices in ophthalmic preparations, intra-articular injections, anti-adhesive preparations, topical preparations for wound healing and soft tissue filler, etc. Hyatrue® sodium hyaluronate ...
-
Product KLEPTOSE® HP HPB - Hydroxypropyl betacyclodextrin
Kleptose HP and HPB for pharmaceutical applications: parenteral use, syrup oral suspensions. HPBCD increase water solubility and bioavailability.
-
Product Proton Tablets
The cholecalciferol contributes to normal absorption and use of calcium and phosphorus, the normal function of the immune system and the maintenance of normal muscle function.
DOSAGE
1 tablet a day.
INGREDIENTS
Cholecalciferol (Vitamin D3).
CHARACTERISTICS
Vitamin ...
-
Product Equidral sachets
Equidral sachets is a dietary supplement indicated in the treatment of the dehydration caused by acute diarrheas, altered electrolytic equilibrium, vomit, gastroenteritis, hyper-sweating.
DOSAGEDissolve the content of a sachet each 250ml of water and shake until the solution is well mixed. Admin...
-
Product Multicompendial TRIS AMINO and TRIS Amino Ultra Pure reagents
ANGUS is the world’s only fully integrated manufacturer of globally compliant TRIS AMINO™ reagents foruse in a variety of demanding pharmaceutical and biotechnology applications. With more than 50 years ofTRIS manufacturing experience, we continue providing TRIS AMINO grades that meet the dynamic needs –as...
-
Product Biochemicals, Biomolecules and Salts
At ANGUS Life Sciences, we consistently deliver products through a traceable and auditable supply chain of high-quality fine chemicals tailored to help you successfully move your products from pilot to commercial-scale production. Choose with confidence from our selection of product offe...
-
Product Single Use Products
We offer a very wide range of single-use products for the pharmaceutical and biopharmaceutical industries. We also offer customised solutions that can be easily adapted to your installations.
single use Assemblies, Back pressure regulators, pinch valves, plastic closure etc...

-
Product Protocatechuic acid
Synonyms: 3,4-Dihydroxybenzoic acid
CAS: 99-50-3
Molecular Formula: C7H6O4
Molecular Weight: 154.12
Properties: White to slightly brown needle shaped crystals; soluble in hot water, ethanol, and ether, slightly soluble in water, insoluble in benzene and p...
-
Product Drug Product & Finished Goods
End-to-endservices designedfor patient safetyand centricity while offering contracting convenience, cos tefficiencies and time savings. Our globally integrated drugproduct (DP) and finished goods (FG) services offer agileand flexible solutions for clinical to commercial products. Our DP capabilities includ...
-
Product PharmASep™ Filter Dryer
The PharmASep™ Filter Dryer combines SWECO’s patented separation process with vacuum drying technology to efficiently and effectively filter and dry product while significantly increasing convenience, safety, precision and productivity in critical applications. This documented aseptic pharmaceutical filtra...
-
Product Heparin calcium
From mucosa to API-all at one production site. EU-GMP audit applied for 2018
-
Product Enoxaparin Sodium
Production starts from our own Heparin Sodium.. EU-GMP inspection applied for 2018.
-
Product HEPES, Free acid Excipient and Intermediate
BioSpectra offers a wide range of products, which includes HEPES, free acid. Features: it is a zwitterionic buffer used to maintain pH of media used in cell cultures. Contact us for more information.
-
Product MES, Monohydrate Excipient and Intermediate
BioSpectra offers a wide range of products which includes MES, monohydrate. Features: it is a zwitterionic buffer that is not absorbed through cell membranes and is virtually transparent in UV light. Contact us for more information.
-
Product MOPS, Free acid Excipient and Intermediate
BioSpectra offers a wide range of products, which includes mops, free acid. Features: it is a zwitterionic buffer used as a running buffer for denaturing gel electrophoresis and as a buffering agent in many biological and biochemical applications. Contact us for more information.
-
Product Tromethamine (TRIS) API
BioSpectra offers a wide range of products, which includes tris / tromethamine. Features: it is commonly used as a buffer in biochemistry and molecular biology, stabilizes the pH of solutions in electrophoresis and other biological applications. Contact us for more information.
-
Product Guanidine Hydrochloride Excipient and Intermediate
BioSpectra offers a wide range of products, which includes guanidine hydrochloride. Features: it is a strong protein denaturant that functions as a chaotropic agent. Contact us for more information.
-
Product Potassium Bromide API
In microbiology, Potassium bromide may be used for the removal of peripheral membrane proteins and in the centrifugation and separation of lipoproteins in high density solvents.
-
Product Urea API
Urea is used in biochemistry and molecular biology as a protein denaturant with low UV-absorptivity.
-
Product Tyramine
Synonyms: 4-(2-aminoethyl)phenol CAS: 51-67-2
Molecular Formula: C8H11NO
Molecular Weight: 137.18
Properties: White to off-white powder

-
Product Anti-MMAE monoclonal antibody
Monomethyl auristatin E (MMAE) is a synthetic antineoplastic agent. Because of its toxicity, it cannot be used as a drug by itself; instead, it is conjugated to a monoclonal antibody (MAB) that directs it to the cancer cells. In international non-proprietary names for MMAE-MAB conjugates, the name vedotin ...
-
Product CDMO Biopharmaceutical Production
With an extensive offering of development services, state-of-the-art manufacturing equipment, and project support, GBI is truly a full-service CDMO biologics partner. Our approach pairs our expert knowledge base with an open mind towards scientific discovery so that together we can develop an optimal way t...
-
Product Somactive LF / Somatropine
Somatropin 5 mg/1,5 ml // 10mg/1,5 ml 15 mg/1,5 ml // 30 mg/3ml For subcutaneous administration Injectable Solution
-
Product Fluid Transfer
A full range of tubing and hoses best suited for the biopharma and pharmaceutical markets. From TPE to silicone, each material type offers a unique set of features and benefits. ValPlus™, an enhanced level of tubing validation certification, is available for select formulations.
-
Product Anti-tuberculosis vaccine BCG10
BCG 10 Anti-Tuberculosis Vaccine is used for protective vaccinations against tuberculosis. Catch-up (supplementary) vaccinations in subjects unvaccinated after birth should be performed as early as possible, no later than at 15 years of age. In case of BCG immunisation booster vaccine doses are not recomme...
-
Product Onko BCG 100
BCG for immunotherapy.
The product is intended for treatment of superficial, epithelial non-invasive urothelial carcinomas (carcinoma urotheliale Ta, Tis, T1)
1 ampoule or 1 vial with the powder contains:
Live attenuated bacilli Bacillus Calmette-Guerin, Brazilian BCG Moreau substrain ...
-
Product BIS-TRIS, LBLE
BIS-TRIS is a zwitterionic biological “Goods” buffer often used in biological pharmaceutical manufacturing and biochemistry for extraction, separation and purification, as well as an intermediate in synthetic reactions for both proteins and nucleic acid. BIS-TRIS is also used as a process aid in peptide sy...
-
Product HEPES Free Acid, LBLE
HEPES is a zwitterionic buffer used to maintain pH of media used in cell cultures. It is one of Good’s twelve buffers that has a pKa value similar to its pH value making it an ideal buffer for pH maintenance. It has only one restriction, it interferes with the Folin protein assay. This buffer can form radi...
-
Product HEPES, Sodium Salt
HEPES Sodium Salt is a zwitterionic salt which is used in the preparation of a HEPES buffer solution. Its pH range of 9.5-11.5 makes an ideal buffer for pH maintenance. HEPES Sodium Salt interferes with the Folin protein assay. The tendency of this buffer to form radicals makes it unsuitable for redox stud...
-
Product Quadrivalent Subunit Influenza Vaccine
As the latest generation of influenza vaccines produced in China,Ab&B Bio-Tech CO.,LTD.JS Hui'erkangxin contains four types of influenza viruses: H1N1 A-type, H3N2 A-type, B-type BV, and B-type BY.This vaccine adopts a safer subunit process, milder extraction agents, and more efficient injectors. ...
-
Product CIP unit [CS25] and CIP/SIP unit [CS25-S]
Two (2) units in our test facility…....but what is the difference?
(Left) CIP unit [CS25] for in-situ cleaning of centrifuges, homogenizers and fermenters in a US based biotech facility.
(Right) CIP/SIP unit [CS25] for in-situ cleaning and sterilization of formulation and holding vessels for a ...
-
Product CIP systems [MEC+CSV1]
The CIP systems were delivered to a pharmaceutical Oral Solid Dosage (OSD) facility in the US, where they are applied for in-situ cleaning of a Fluid Bed Dryer and V-blenders.
The CIP systems are installed in a technical space and are hard-piped to process suites that include 'remote' valve manifolds ...
-
Product 16beta/alpha-Methyl Glucocorticoid Series
Including Betamethasone,16beta-methyl epoxide(DB11) ,Clobetasol, Clobetasol propionate,Diflorasone, Dexamethasone (GMP),8DM acetate ,Mometasone, Mifepristone, Dydrogesterone etc.

-
Product 19-Nor Steroids
Including Mifepristone, Ulipristal,Estradiene dione-3-keta,17β-hydroxy-estra-4,9-dien-3-one,Estrone etc

-
Product Fermentation Products From Phytosterol,
Including 4-androstenedione; 4-AD ,δ-sitolactone,(20S)-21-hydroxy-20-methylpregn-4-en-3-one; BA etc.

-
Product Beromun®
Beromun® is brand name for recombinant TNF-alpha – TASONERMIN (Produced by genetically modified E. Coli) Beromun® is indicated in adults as an adjunct to surgery for subsequent removal of the tumor to prevent or delay amputation, or in the palliative situation, for non resectable STS of the limbs, used in...
-
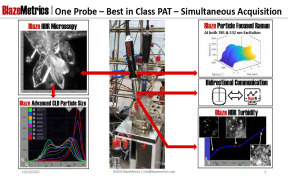
Product PAT Probe
BlazeMetrics USA for Next generation ONE Probe PAT Solution & Application, InSitu probe for simultaneous measurement of Particle Shape & Size , Microscopy , HDR Turbidity & Particle focused Raman in order to optimise the process precisely during the development phase itself. Single probe from 2...
-
Product ilon Classic Ointment - No.1 in German pharmacies
Herbal medical product effectively helps with symptoms of skin inflammation such as redness, tightness, swelling and pain with pus. Top-selling blistering ointment in Germany.
Active ingredients: Larch turpentine, turpentine oil of the maritime pine type, eucalyptus oil
...
-
Product CDMO
• Cytotoxic Injection (Liquid , Lyophilized) • ONCOLOGY • CYTOTOXIC API • EU GMP • JGMP (Japan) • KGMP (Korea) • PIC/S
-
Product FPFs
• Genexol®PM Inj.(Paclitaxel) • Genexol® Inj.(Paclitaxel) • Nanoxel®M Inj.(Docetaxel) • Docetaxel Inj.(Docetaxel) • Nexatin® Inj.(Oxaliplatin) • Everose® Tab.(Everolimus) • Pemed®S Inj.(Pemetrexed) • Azalid® Inj.(Azacitidine) • Decilid® Inj.(Decitabine) • Lenalid® Inj.(Lenalid...
-
Product Biotest Human Serum Albumin
• Human Serum Albumin for the biotechnology and pharmaceutical industry • We offer a human serum albumin (HSA) product that complies with the regulatory requirements of multiple health authorities. • It is manufactured in a GMP compliant, state-of-the-art manufacturing plant and supplied und...
-
Product Distreptaza
Rectal suppositories indicated for:
1) Supportive treatment of pelvic inflammatory disease (PID) – inflammatory diseases of the ovaries, fallopian tubes and endometrium,
2) Adhesions after abdominal surgical procedures,
3) Acute haemorrhoidal disease and chronic haemorrhoidal dis...
-
Product Goserelin Boston Biopharma
Goserelin is hormone therapy, classified as "LHRH agonist." (Endocrine Therapy)
It is indicated:
- in advanced breast cancer in pre and perimenopausal women suitable for hormonal manipulation
- as an alternative to chemotherapy in the standard of care for pre and perimenopausal women with estrogen...
-
Product elogbook : Harmonized logbooks.
Presenting Electronic Logbook, complying with 21 CFR Chapter 11 requirements, a simple yet controlled platform for all Pharmaceutical manufacturing to manage logbooks, digitally.
Complying with GMP guidelines, Electronic Logbook provides capability of defining flexible workflow, SOPs on area...
-
Product DuoDERM
Cartridge - Tubular Glass & Aluminium Foil provide a sustainable and minimalist packaging solution.AppTivator - Reusable, this device enables easy activation, reconstitution and application of your product through the (SPP) Sintered Porous Polymer filter Tip.
-
Product Biomedtracker
Biomedtracker provides real-time analysis of major market-moving events in the pharma and biotech industries. With catalyst tracking and expert insights, Biomedtracker helps you stay on top of breaking events, the drugs pipeline, upcoming milestones, companies, and deals – and their impact on the marke...
-
Product Datamonitor Healthcare
Support the development and validation of your strategic plans and ensure commercial decisions are based on marketplace realities. Datamonitor Healthcare offers expert coverage of key diseases, treatments, companies, drugs, market access and market trends. Results from in-depth KOL and payer interviews...
-
Product Pharma360
Search for comprehensive disease, drugs and company information from one convenient location with the all-new Pharma360. Updated daily using the power of the entire Pharma Intelligence suite of products, Pharma360 immediately delivers critical intelligence from Trialtrove, Pharmaprojects, Datamonitor H...
-
Product MK96 Sanitary PRV
Pharma process and clean utility pressure reducing valves, 1/2" - 3" (DN15 - DN80) with tri-clamp or weld ends, low to high flow, manual or air augmented, all with the Jorlon™ lifetime diaphragm warranty.
-
Product MK95 Sanitary BPRV
Pharma process and clean utility back pressure regulating valves, 1/2" - 3" (DN15 - DN80) with tri-clamp or weld ends, low to high flow, manual or air augmented, all with the Jorlon™ lifetime diaphragm warranty.
-
Product MK93 Sanitary Steam traps
Clean steam traps and accessory devices for process SIP and distribution line condensate removal, 1/2" - 2" (DN15 - DN80), tri-clamp, weld end or NPT connections, low flow to very high flow, all with low subcooling
-
Product Intiline Powder
Semi-Automatic powder or granulate filling machine
Production of up to 1.500 units/hour.

-
Product Fundraising
Operating across all key life science sectors, PharmaVentures offers deep sector expertise to help you derive optimal value from your funding transactions.
We support our clients with strategic advice and full end-to-end process management capabilities to achieve the best outcome from their stra...
-
Product Vial Washing - Effective and Efficient
The SP i-Dositecno vial washer range consists of four models to cover a wide range of production rates including a compact footprint option for laboratory and small batch applications to clean vials with as little as 1 liter per minute WFI (Water-For-Injection) usage, as well as high-speed production a...
-
Product Strike 20 rotary evaporator
20 Lt rotary evaporators designed for industrial systems and R&D purposes.Completely safe instruments as well as performance optimization. STRIKE 20 is a 20lt rotary evaporator designed for industrial systems and R&D purposes. It meets all the process needs using 3.3 borosilicate glass and PTFE all...
-
Product 50L PHARMA JACKETED GLASS REACTORS DN400
Complete jacketed glass reactors for pharmaceutical use on our unique free standing movable frame.
Industrial design, high performing industrial stirring system and connections.
Main features:
- Gmp compliant;
- Total safety: no residues or stagnant liquids;
- For pharmaceuti...
-
Product AUTOCOATER
Design is cGMP – Current Good Manufacturing Practices compliance. Fully automatic film & sugar coating process by PLC Controls with HMI Touch screen.Extremely user friendly software and operation through PLC The machine structure comprising of Perforated Pan, water collection tank...
-
Product FLUID BED DRYER
EQUIPMENT WILL COMPLY WITH THE LATEST GMP STANDARD & NORMS.Basic equipment with Single Skin Air Handling unit witho Pre/micro filter 20/10 Micron.
o Micro Filter of 5 Micron.
o HEPA Filter of 0.3 Micron. (Optional)
Steam ...
-
Product LIQUID MANUFACTURING PLANT
Liquid Manufacturing Plant with fully automatic operations as designed
per CGMP-Current Good Manufacturing Practice compliance.
All contact parts SS 316 & Non Contact Parts SS 304.
Liquid Manufacturing plant consi...
-
Product IMMUCOTHEL®
Active substance - Standardised immunocyanin
Indications - Relapse prevention of superficial carcinoma of the bladder [Tis, Ta–T1(G1–G3)] after transurethral resection. IMMUCOTHEL® constitutes only second line treatment in immunological treatment for relapse prevention.
• Patients appreciate the...
-
Product Chitosan Pharma Grades, low/medium/high molecular weight
Kraeber & Co GmbH offers a wide range of pharmaceutical products. We are GMP, GDP and ISO 9001:2015 certified. You can find our product list in the download files. For more information, please contact: www.kraeber.de
-
Product GB-5001
Duration / Indication: 1-Month / Alzheimer’s disease
Development Status: Nonclinical
-
Product GB-5112
Duration / Indication: 1-Month / Alzheimer’s disease
Development Status: Lead-optimization
-
Product 汉利康® (pronounced as Han-Li-Kang) Rituximab Injection
汉利康® (pronounced as Han-Li-Kang) Rituximab Injection. It is the first-ever China-manufactured biosimilar approved by the NMPA for the treatment of non-Hodgkin lymphoma (NHL). Meanwhile, we are also conducting a Phase 3 clinical trial for rheumatoid arthritis (RA) for HLX01 in China. ...
-
Product Human Hepatitis B Immunoglobulin
The manufacturing process for human hepatitis B immunoglobulin contains a step of low pH incubation for viral inactivation. It contains a suitable amount of glycine as stabilizer, but does not contain any antiseptic or antibiotic.
Indications:
-Post-exposure prophylaxis should be c...
-
Product Human Albumin
The main component of this preparation is human albumin, which is prepared by cold ethanol fractionation of human plasma from healthy donors. The manufacturing process of human albumin contains a step of heating the final solution at 60℃ for 10 hours to inactivate the viruses present in it. It contains...
-
Product Heparin sodium
Anticoagulant drug.100% are extracted from the intestinal mucosa of pigs.
White or almost off-white powder, odorless, hygroscopic; soluble in water; insoluble in ethanol, acetone and other organic solvents.
1kg/drum or 5kgs/drum
-
Product Enoxaparin Sodium
Enoxaparin Sodium complies with USP. Injectable Grade Definition
Enoxaparin sodium is the sodium salt of low molecular weight heparin obtained by alkaline depolymerization of the benzyl ester derivative of heparin from porcine intestinal mucosa.
Usage
Enoxaparin sodium is i...
-
Product Sterile Products Contract Manufacturing
Our main CDMO activity is the large scale manufacturing of injectable products in our internationally reputed site in Pau (France). The site is organized around 9 independent workshops, specialized in the aseptic filling under isolators of potent molecules such as anticancer cytotoxics, monoclonal ant...
-
Product Recombinant human erythropoietin injection (cho cell)
Shandong kexing bioproducts offers a wide range of products which includes recombinant human erythropoietin injection (cho cell). It is colorless and transparent liquid with ph6.9±0.5. Packaging: pre-filled syringe or vial. Storage: stored at 2-8?. Contact us for more information.

-
Product General Portfolio
We produce medicines, cosmetics and supplements in our own Anvisa GMP facilities.
Our sales team promote our product lines for General Practition, Pediatrics, Gynecology and Obstetrics, Gastroenterology, Psychiatry, Neurology and Physioterapy.
-
Product Albumin-Biopharma 20%
Albumin 20% is the gold standard of natural colloidals Albumin's profile of the safety and efficacy exceeds synthetic colloids in volume resuscitation of critically ill patients Quality, purity, and safety of Albumin-Biopharma correspond with the European Pharmacopoeia regulations for the finished pro...
-
Product Bioven Mono 5%
Quality, purity, and safety of BIOVEN MONO correspond with the European Pharmacopoeia regulations for the finished product and raw materials Using the method of ion-exchange chromatographic purification relates these medicines to the last (4th) generation of IVIG, due to BIOVEN MONO have a high ...
-
Product Immunoglobulin Human Anti Rhesus RH0 (D)
Quality, purity, and safety of Immunoglobulin anti-rhesus Rh (D) human correspond with the European Pharmacopoeia regulations for the finished product and raw materials 2 validated stages of virus inactivation, including SD treatmentDonors were examined for the absence of antigen and antibodies to HIV...
-
Product Pre-assemble processing skids
Briggs design and manufacture pre-assembled processing skid units. We have examples where Briggs have supported lab scale trails to pilot trails which have then enabled the design and manufacture of manufacturing scale units.Our skids are usually bespoke design and Briggs offer a turnkey package which can ...
-
Product Sterile Manufacturing
Avara Pharmaceutical Services offers liquid and lyophilized fill-finish of sterile injectable products from state-of-the-art facilities in North America and Europe. Contact us for more information.
-
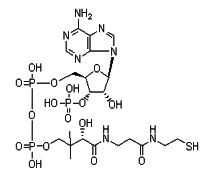
Product Coenzyme A, free acid
Molecular formula: C21H36N7O16P3SMolecular weight: 767.5 Code no.: 8001 Product group: Coenzymes Application: &nbs...
-
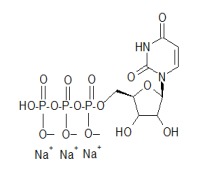
Product Uridine 5'-triphosphate,trisodium salt hydrate(UTP)
Molecular formula: C9H12N2Na3O15P3 Molecular weight: 550.09Code no.: 2016 Product group: Nucleotides Application: &nb...
-
Product 2',5'-Dihydroxyacetophenone
Molecular formula: C8H8O3Molecular weight: 152.15 Code no.: 9013Product ...
-
Product Monomaterial Packaging Specialist
Pharma and Biotech toploading and sideloading cartoning systems from the monomaterial specialistDividella from Switzerland provides pharma and biotech packaging systems for the secondary packaging of parenterals. Dividella's patented top-loading and recently also side-loading cartoners package syringes, vi...
-
Product Product Handling and Infeeding Systems for all your products
The power of Dividella's NeoTOP & NeoTRAY cartoning solutions is enhanced by modular feeding systems that allow packaging lines to be optimized and tailored for all packaging formats and objects. Dividella feeding modules can be selected to support Top Loading of sensitive parenteral p...
-
Product SNEC 30 - Self Nano Emulsifying Curcumin
The product SNEC30 (Self Nano Emulsifying Curcumin 30mg) is based on Indigenously developed drug delivery system for Curcumin by us in collaboration with Jamia Hamdard University and support from Department of Science and Technology, Government of India. We have been granted US Patent (US Patent No. US88...
-
Product Sanitary Shell & Tube Heat Exchangers
Exergy sanitary shell & tube heat exchangers are designed to meet the high quality requirements and hygienic standards of the pharmaceutical industry.
-
Product Sanitary Tube - in - Tube Heat Exchangers
Exergy Sanitary tube-in-tube heat exchangers consist of two tubes, an inner and an outer coiled together.
-
Product PUREX Point-of-Use WFI/PW Dispensing Systems
Exergy Point-Of-Use systems dispense instant, temperature controlled WFI or PW, and are designed to meet the high quality requirements and hygienic standards of the pharmaceutical industry.
-
Product 3'-Sialyl Lewis X
3'-Sialyl Lewis X is a tetrasaccharide carbohydrate, known to play a vital role in cell-to-cell recognition processes.
-
Product Xalost Ophthalmic Solution
Active ingredient: Latanoprost 50mcg/mL Indications: reduction of intraocular pressure of the following disease: -adults: open-angle glaucoma, chronic angle-closure glaucoma, ocular hypertension; pediatric glaucoma, ocular hypertension. - pediatric patients: pediatric glaucoma, ocular hypertension.&n...
-
Product testo 184 T3 - Temperature Data Logger
USB tranport data logger testo 184 T3 for temperature
• Monitoring transport in compliance with GxP, 21 CFR Part 11, HACCP and EN 12830
• All in one temperature data logger: PDF measurement data, configuration file, acceptance test certificate 3.1 according to DIN ISO 10204 and instructio...
-
Product testo Saveris
testo Saveris – your all-in-one solution for climate monitoring • Safe: in the event of critical climate data, you will receive alerts via e-mail, SMS or directly on site • Economic: testo Saveris saves staff costs for measurement and documentation work...
-
Product Centrifuge Liners & Bags
Sefar products for centrifugal filters are matched to the filter machines used, whether they are band centrifuges, bag centrifuges or inverting centrifuges. Our ready-made solutions are based on optimized Sefar designs that have been proven in use over many years.
Thanks to our wide range of fab...
-
Product Fluid Bed Dryer Bags
Thanks to their welded seams, Sefar’s bags for fluid bed dryers guarantee minimal particle loss.
The high-quality finish ensures precise and durable products.
Our bags for fluid bed dryers are subjected to a final testing of their permeability and certified accordingly – a unique serv...
-
Product SEFAR PHARMA GMP BAGS
SEFAR PHARMA GMP BAGS are the certified filter bag solution for the pharmaceutical industry.
The design has been developed to avoid contamination from the needle felt.
The high filtration efficiency is granted by an ePTFE microporose membrane laminated on polyester or PTFE needle felt...
Biopharmaceuticals
Biopharmaceuticals, also known as biologic medical products, are drug products manufactured using biotechnology. Biopharmaceutical drugs are structurally similar to various compounds within the body. Over time, drug development has progressed from small-molecule compounds to large molecule proteins and other accompanying biopharmaceuticals. The discovery of biopharmaceuticals has significantly improved the healthcare delivery. It has provided a shift in providing cures for chronic illnesses such as refractory cancers, diabetes, autoimmune diseases, etc., thereby improving the affected patients' quality of life.
The use of biopharmaceuticals for therapeutic purposes dates as far back as the 19th century, with recombinant human insulin being the first approved biopharmaceutical used for therapeutic purposes in human beings. Over the last three decades, the biopharmaceutical industry has experienced notable growth and is expected to contribute to some remarkable pharmaceutical industry developments. Biopharmaceutical offers a wide range of advantages and benefits to pharmaceutics, and here, we would be exploring these benefits. We would also examine the biopharmaceutical manufacturing process, notable examples of biopharmaceuticals, and more.
What are biopharmaceuticals?
These complex medicines are proteins (monoclonal antibody), sugars (glycan), nucleic acids (RNA, DNA), living cells, or tissues extracted or semi synthesized from a biologic source by biotechnology for in vivo diagnosis or therapeutic purposes.
The majority of biopharmaceuticals are synthesized or derived from living organisms or genetically modified cells and organisms. They cannot only lessen symptoms or treat chronic diseases but also cure them with little or no side effects because of their origin and specificity. Some biopharmaceuticals include enzymes, antisense drugs, cytokines, monoclonal antibodies, gene therapy, cell therapy, etc.
Biopharmaceuticals have helped personalize the treatment process to meet the specific health needs of patients. This is achieved through the modification of drugs genetically through DNA-recombinant technologies or cell fusion to alter specific factors to cure, prevent, or diagnose individual diseases.
How are biopharmaceuticals made, and why?
As opposed to chemical synthesis through which conventional medicines and drug products are made, biopharmaceutical drugs are synthesized through more complex processes from living cells. With the rapid growth of the biopharmaceutical industry and the discovery of new biologic therapies, the term biopharmaceutical (biologics) refers to a variety of medicines and drug products. The diversified portfolio contains various drugs produced by different biotechnological methods and techniques.
These drug products are not made from chemical mixtures but rather from living organisms. Experts in cellular biology, biochemistry, and several other fields carry out biologic testing and analysis (using analytical methods) to ensure that the desired end product is made.
For example, biologics used to treat joint pain are made by adding a DNA into a living cell such as a yeast or mammalian cell to enable the production of a specific molecule in large amounts. These molecules are usually therapeutic proteins that are isolated and utilized as active ingredients.
When newly introduced, biopharmaceuticals focused solely on developing recombinant protein such as enzymes (TPA) and hormones (insulin, erythropoietin, growth hormone, etc.) through recombinant DNA techniques. With time, they have delved into the design and development of chemically defined cells, manufacturing of monoclonal antibodies, and genome-based technology. These new techniques have improved the pharmaceutical manufacturing process and created new treatment options for different diseases.
Like every other drug, biopharmaceutical drugs are produced for therapeutic purposes, but with a catch. They are produced majorly to cure chronic and dilapidating diseases such as cancer, diabetes, rheumatoid arthritis, and several others. The manufacturing of these drug products also aims at providing effective, safe, and cost-effective drugs for therapeutic purposes.
What is the biopharmaceutical industry?
The biopharmaceutical industry is an essential subset of the pharmaceutical industry. It is comprised of biopharmaceutical companies that discover, develop, produce, and market biopharmaceuticals. The drug products made are used to diagnose, treat, prevent, and cure diseases when administered to patients. This industry deals majorly in biopharma drugs and medicines produced via the biotechnological process from living entities such as micro-organisms, living cells, tissues, or organs of humans and animals. The biopharmaceutical industry successfully merges between biotech and pharmaceutical companies to provide better and safer healthcare to patients.
What is the difference between biopharmaceutic and biopharmaceuticals?
Biopharmaceutics is simply pharmaceutics that deals with biopharmaceuticals. It is a branch of pharmaceutical science that examines the physiochemical properties of drugs, their toxicology, pharmacology, dosage forms, and patients' clinical response to drugs, biopharmaceuticals inclusive. The recommended dosage and dosing interval varies for different drugs, and it significantly affects their efficacy and safety. Biopharmaceutics provide adequate information and understanding of the pharmacodynamics and pharmacokinetics of biopharmaceutical drugs, their metabolites, and their effects on the body. Through biopharmaceutics, biopharmaceutical drugs can be evaluated based on their properties and effects on the body.
On the other hand, biopharmaceuticals refer to inherently biologic pharmaceuticals extracted, synthesized, or semi-synthesized from live forms using biotechnological methods. They constitute a significant part of the drug products examined and evaluated in biopharmaceutics to improve healthcare delivery and, in turn, the quality of patients' lives.
How are biopharmaceuticals manufactured?
The rapid advancement of genetics and molecular biology has played a significant role in the biopharmaceutical manufacturing process. The introduction of new and refined techniques such as protein purification, host system, etc., has positively impacted the efficacy, capacity, and safety of biopharmaceuticals produced.
There are many biopharmaceutical manufacturing processes, and they vary based on the type of drug product to be produced.
Where is biopharmaceutical manufacturing heading?
With several emerging technologies in the biopharmaceutical manufacturing process, the biopharmaceutical manufacturing process is expected to take a huge turn. With over 1000 biopharmaceuticals in clinical trials, the majority of drugs in the future may be biopharmaceuticals. The increased need for biopharmaceuticals and the constant advancement in biotech will impact biopharmaceutical manufacturing. Given the various advantages of gene therapy, biopharmaceuticals' manufacturing process is likely to focus more on gene therapy in the future.
What is the process involved in the process of manufacturing biopharmaceuticals?
The process of small-scale and large-scale biopharmaceutical production varies. However, biopharma companies produce to indulge in large-scale biopharmaceutical production. The processes involved in manufacturing these drugs are complex, lengthy, and largely differ depending on the end-product. Generally, the processes include biosynthesis, purification, formulation, and final dosage preparations.
Materials and equipment are sterilized at the biosynthetic stage, and a cell culture or biologic extract is harvested or collected in a bioreactor to grow and multiply. During the purification process, cells, tissues, enzymes, or desired products are separated from surrounding nutrients and byproducts to produce the bulk product. Unlike conventional drugs mixed with other chemical components during formulation, biopharmaceuticals are made and sold as sterile products. Although they are sold as sterile products, biopharmaceuticals are packaged in similar ways as conventional drugs before distribution and sales.
This description gives a general knowledge of the manufacturing process carried out. Nevertheless, there are special intricate biotech processes that can be carried out and some include continuous manufacturing, and other processes requiring single use technology.
What special considerations must be made when manufacturing biopharmaceuticals?
Consistent safety, effectiveness, and quality are the essential factors that must be considered during the biopharmaceutical manufacturing process.
The manufacturing processes of each biopharmaceutical product must be designed to produce consistent quality and safe biopharmaceutical drug products. This involves the complete removal of contaminants and impurities such as viral contamination, endotoxins, cell membranes, and others from the separated product. Also, regular rigorous and well-documented checks and examinations on the efficiency and performance of the materials and equipment must be carried out to validate the manufacturing process.
Benefits of biopharmaceuticals
Given the structural composition and origin of biopharmaceuticals, these bio drugs provide several health benefits to patients and, in extension, the healthcare industry and healthcare professionals. The emergence of biopharmaceuticals has provided patients with a better shot at quality life and longevity.
How do patients benefit from biopharmaceuticals?
Biopharmaceutical drugs, unlike conventional medicines, mimic the structures of human compounds, and this gives biopharmaceuticals the potential and ability to cure diseases more effectively. They also produce fewer side effects due to their high specificity, unlike the traditional medications that are capable of affecting multiple systems simultaneously. Given their similarities with human compounds, they are very safe and highly effective.
Also, these drugs can be genetically modified to allow healthcare professionals to tailor treatments to meet patients' specific health needs. In addition, some generic drugs play crucial roles in providing patients with curative and diagnostic benefits.
However, one of the greatest potentials of biopharmaceuticals that also serves as an essential benefit to patients is gene therapy. Through gene therapy, conditions characterized by rapidly dividing cells due to mutated or defective genes, such as cancer, can be cured. Unlike some cancer drugs and other medications also, biopharmaceutical drugs are non-carcinogenic. The innovation of biopharmaceuticals also aids in producing varieties of vaccine to prevent infection and the spread of infectious diseases in patients.
Generally, bio drugs improve longevity, increase life expectancy, provide welfare to the population, and provides several public health benefits.
How does the healthcare industry benefit from biopharmaceuticals?
The impact of biopharmaceuticals and biopharmaceutical innovation on health care professionals also tells on the healthcare industry. Biopharmaceuticals have a massive positive economic impact, and other healthcare industry benefits that cannot be ignored. The development and availability of the various biopharmaceuticals in the pharmaceutical market will increase access to bio drugs, cost-effective medicines, and a boost in the global pharmaceutical market. Also, the ease of commercial production of biopharmaceuticals boosts the healthcare industry's economy and market growth.
How do healthcare professionals benefit from biopharmaceuticals?
Biopharmaceuticals and biopharmaceutical innovations provide outstanding benefits to healthcare professionals. It provides healthcare professionals with a variety of safe and efficient curative and therapeutic options for their patients. Therefore, in cases where a particular bio drug doesn't produce the desired therapeutic effect in a specific patient or has undesired side effects, other alternatives within the same drug family can be administered. Also, biopharmaceutical innovation provides job opportunities for healthcare professionals and individuals in different fields of practice.
Examples of common biopharmaceuticals
There are various biopharmaceutical products, and the majority of these products are widely known. Some common biopharma products include insulin, blood factor IX and VIII, human growth hormone, erythropoietin, tissue plasminogen activator, therapeutic antibodies, glucocerebrosidase, interferon α, and β, and GSCF. These form the most common examples, but they fall into the different biopharmaceutical classes, including hormones, clotting factors, blood products, vaccines, cytokines, and growth factors.
Where can some common biopharmaceuticals be found?
Biopharmaceuticals are derived and produced from various cells. They can be extracted and synthesized mammalian cell lines, microbial cells (yeast culture or recombinant E. coli), moss plants, and plant cell cultures. The majority of these sources are readily available and accessible at all times. These sources are origins from which the drug development process spans.
Who commonly uses biopharmaceuticals?
Biopharmaceuticals are primarily used for patients. However, most of the patient population that uses biopharmaceuticals are patients with long-term diseases such as psoriasis, diabetes, Crohn’s disease, rheumatoid arthritis, etc. The elderly constitute the majority of patients affected by either of these chronic conditions. Also, cancer patients form a significant percentage of the patient population using biopharmaceutical products.
What industries can common biopharmaceuticals be found in?
The common biopharmaceuticals are produced majorly by the biopharmaceutical and biotechnology industries. Within these industries, these biopharmaceutical products are developed and manufactured by biopharmaceutical companies. Some of the top pharma companies that manufacture the most common biopharma products include Gilead Sciences, Amgen, Genentech, Biogen, Riotech, Samsung biologics, Teva Pharmaceuticals, Celgene, Pfizer, United Therapeutics Corporation, UCB, and Sanofi. Although these companies form the existing biopharmaceutical manufacturing companies, a new biopharmaceutical company, Clover biopharmaceuticals, presently undergoing clinical trial is set to become a top tier pharmaceutical company in this industry.
References
https://medcraveonline.com/OAJS/biopharmaceutical-innovation-benefits-and-challenges.html
References
Upcoming Events
-
CPHI Japan 2024
East Halls 4, 5 & 6, Tokyo Big Sight, Tokyo, Japan
17 Apr 2024 – 19 Apr 2024 -
CPHI North America 2024
Pennsylvania Convention Center, Philadelphia
07 May 2024 - 09 May 2024 -
Pharmaceutical Industry Webinars
-
Webinar Exploring Three Technological Trends in the Future of Pharmaceutical Manufacturing
-
23rd May 2024
-
4pm CET / 10am EST
-
-
Webinar Achieving Manufacturing Excellence Through Digital Transformation
-
16th April 2024
-
4pm CET / 10am EST
-
-
Webinar Made in Africa: What’s Driving Pharma Manufacturing
-
28th March 2024
-
4pm CET / 10am EST
-
-
Webinar Case Study: Risk Management for Annex 1 Sterile Production EMS
-
28th February 2024
-
4pm CET / 10am EST
-
-
Webinar Innovative Strategies for B2B Pharma Marketeers: Driving Value through Content
-
20th February 2024
-
4pm CET / 10am EST
-
-
Webinar Revolutionizing Pharma: Data and AI Unleashed
-
18th January 2024
-
4pm CET / 10am EST
-
-
Webinar Optimal Temperature: Elevating Biologics Cold Chain Excellence
-
16th January 2024
-
4pm CET / 10am EST
-
-
Webinar Market Outlook – The Biggest Pharma Trends of 2024
-
12th December 2023
-
4pm CET / 10am EST
-
-
Webinar The Next Frontier – Emerging Opportunities in the LATAM Pharma Market
-
21st November 2023
-
4pm CET / 10am EST
-
-
Webinar Vistamaxx™ MED - imagine the possibilities for healthcare product performance
-
10th October, 2023
-
4pm CET / 10am EST
-
-
Webinar Co-processing: A Multifaceted Approach for Enhancing Density & Powder Flow
-
19th September 2023
-
4pm CET / 10am EST
-
-
Webinar The Outlook for Cell & Gene Therapy Manufacturing
-
28th June 2023
-
4pm CET / 10am EST
-
-
Webinar Contract Packaging Outlook: Growth Trends within the Commercial Packaging Sector
-
25th May 2023
-
4pm CET / 10am EST
-
-
Webinar The HPAPI Boom: How Pharma is Rising to the Manufacturing Challenge
-
23rd February 2023
-
4pm CET / 10am EST
-
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance

-comp316140.jpg)













































































.png)
.png)
.png)











































































































.png)


.png)



.jpg)