Aptar Pharma Twister Chosen for Asthma Therapy
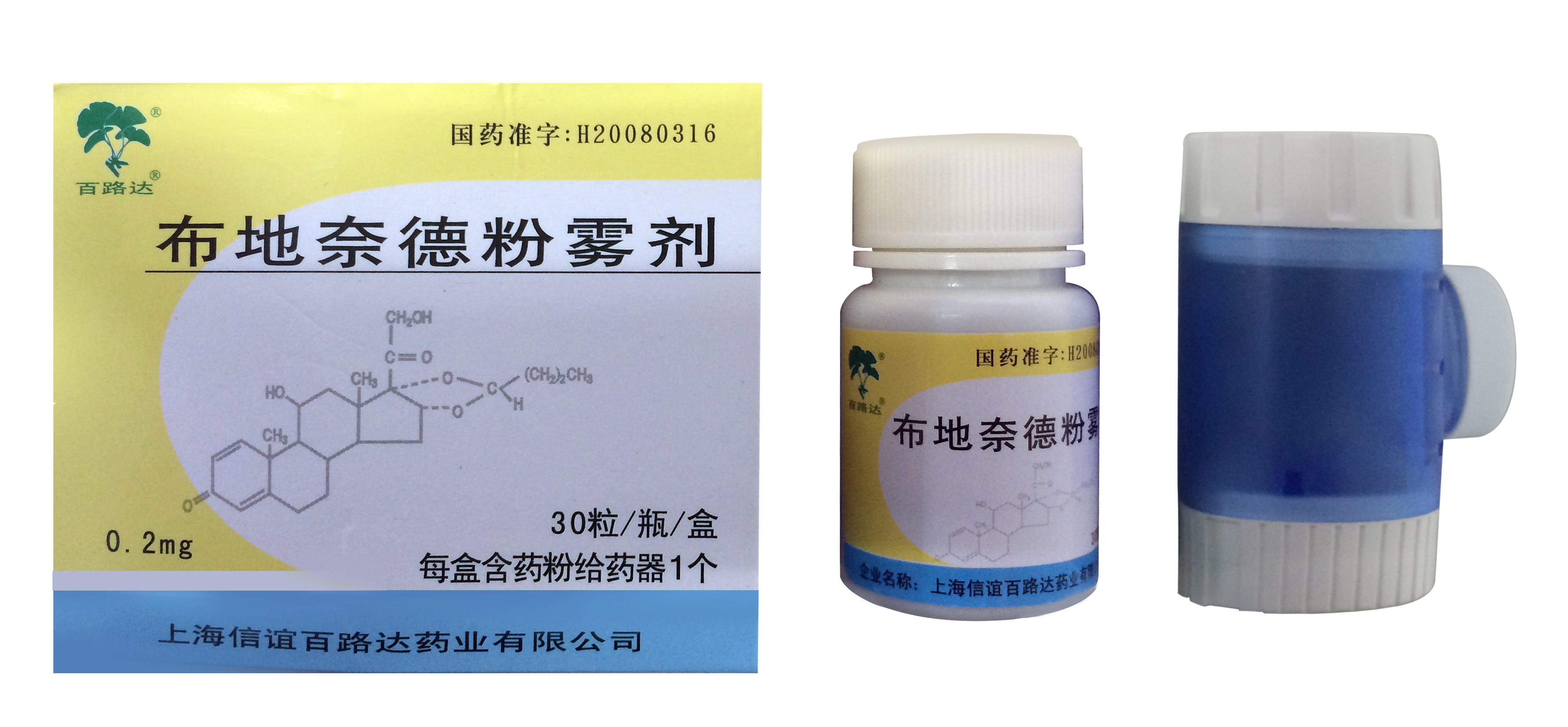
Aptar Pharma has partnered with Shanghai Sine Promod Pharmaceutical to develop and launch its new Budesonide Dry Powder Inhaler (DPI), which uses Aptar Pharma’s novel, user-friendly and cost-effective Twister DPI.
The first drug product marketed with Twister Budesonide DPI is designed for maintenance therapy in the treatment of asthma. Budesonide DPI is one of the first drug products manufactured locally and marketed in China. This cost-effective drug product incorporates Aptar Pharma’s Twister technology. Budesonide DPI contains an inhaled corticosteroid for the treatment of asthma in adults and children age six and over. The product is marketed in two versions, one version containing 30 capsules and the other, 60 capsules.
Each capsule consists of 200µg of budesonide blended with lactose monohydrate as the carrier.
Key benefits of Aptar Pharma’s Twister
Twister is a new capsule-based DPI, designed and developed by a multidisciplinary technical team based at Aptar Pharma’s Centre of Excellence in Le Vaudreuil, France to specifically address unmet medical needs in fast-growing markets.
Twister has a simple and robust design with few components, making it costeffective for asthma and COPD treatments, and has been tested and validated with a number of different dry powder drug formulations.
Twister is designed to be patient-friendly and easy to use, allowing patients easy access to their medication in three simple steps: insert, twist and inhale. To help improve patient compliance with the prescribed treatment, Twister is transparent, allowing the capsule and powder to be seen in the device as they are administered. In addition to these visual cues, the patient is also guided by audible feedback during inhalation.
“Shanghai Sine Promod Pharmaceutical is one of Aptar Pharma’s key customers in China. We are particularly proud to have supported their Budesonide DPI program through the development and commercialization of Twister, manufactured locally in our Suzhou facility. This is a very important milestone for Aptar Pharma China,” stated Mrs Yan Jiang, Vice President Northeast Asia for Aptar Pharma.
Related News
-
News Patients vs Pharma – who will the Inflation Reduction Act affect the most?
The Inflation Reduction Act brought in by the Biden administration in 2022 aims to give better and more equitable access to healthcare in the USA. However, pharma companies are now concerned about the other potential costs of such legislation. -
News CPHI Podcast Series: What does the changing US Pharma market mean for industry and patients alike?
In this week's episode of the CPHI Podcast Series Lucy Chard, Digital Editor for CPHI Online is joined by James Manser to discuss the political and market changes in the US pharma field. -
News CPHI Barcelona Annual Report illuminates industry trends for 2024
The CPHI Annual Survey comes into it’s 7th year to report on the predicted trends for 2024. Over 250 pharma executives were asked 35 questions, with their answers informing the industry landscape for the next year, spanning all major pharma marke... -
News Which 10 drugs are open to price negotiation with Medicare in the USA?
The Centres for Medicare & Medicaid Services, under the Biden administration in the USA, has released a list of the 10 drugs that will be open to price negotiations as part of the new legislation under the Inflation Reduction Act (IRA). -
News EU Medical Devices Regulation causes unintended disappearances of medical devices for children, doctors state
Doctor groups and associations have appealed to the EU to correct the EU Medical Devices Regulation law that may cause unintended shortages of essential drug and medical devices for children and rare disease patients. -
News 10 Major Drug Approvals So Far in 2023
Last year, 37 novel drugs were approved by the FDA, this was a high number for such a category, and covered many fields including oncology, demonstrating how promising further research is, and how it is only continuing to build. To date, there are alre... -
News Detecting Alzheimer's disease with a simple lateral flow test
A novel rapid diagnostic test for early-stage Alzheimer's disease has been developed using a biomarker binder from Aptamer Group along with technology from Neuro-Bio, the neurodegenerative disease experts. -
News CPHI Podcast Series: outsourcing and manufacturing trends
Listen to the CPHI Podcast Series this June to hear Gil Roth of the PBOA speak with Digital Editor Lucy Chard about the biggest trends and topics to watch in pharma outsourcing and manufacturing at the minute.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







