Bosch at Achema - New Inspection Technologies for Higher product Quality and Safety
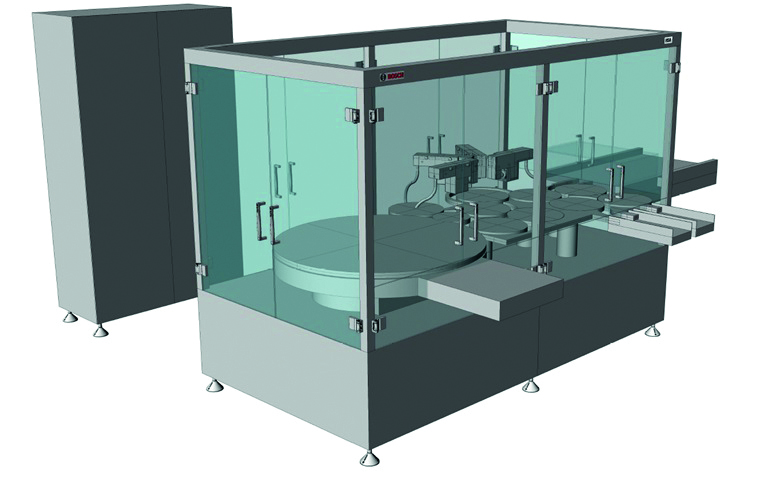
At Achema 2015, Bosch Packaging Technology showcases two new pharmaceutical inspection machines. The KHS 1 is a new development emerging from a co-operation of Bosch with Lighthouse Instruments. It is designed for container closure integrity testing of ampoules, vials, bottles, cartridges and syringes using laser headspace analysis (HSA). The AIM 3, in turn, is the new member of the well-established AIM series. It offers the possibility to perform both visual inspection and high-voltage leak detection (HVLD) for ampoules and vials on one single platform. “These new platforms underline the expertise of Bosch Packaging Technology in all inspection areas. Independent of the product or application, we can offer extensive consulting and the appropriate technology to ensure the highest product safety and quality for all requirements,” Mahmoud Hamada, business development manager at Bosch Inspection Technology, explains.
Successful Co-operation with HSA Pioneer
To further expand its inspection portfolio, Bosch collaborated with Lighthouse Instruments, the leading provider of non-destructive laser spectroscopy headspace analysis systems. This expertise in leak detection fits ideally with the highest quality requirements of the pharmaceutical industry. “By combining this expertise with our market-leading machinery for pharmaceutical inspection applications, we have developed a highly flexible and customizable high-speed platform,” says Joachim Baczewski, head of Inspection Technology and president of Bosch Packaging Technology K.K. in Japan. Jim Veale, president of Lighthouse Instruments, adds: “We are excited to offer the pharmaceutical industry best-in-class leak detection machines together with Bosch Packaging Technology.”
The KHS 1 is the new high-end leak detection system from Bosch for closure integrity testing with laser headspace analysis. It measures the absorbed light passing through the headspace region via laser spectroscopy. HSA is applicable to lyophilized products and medicines filled under vacuum or purged with inert
gas. The new KHS 1 inspects both standing and non-standing containers at outputs of up to 600 per minute, and can combine HSA with optional near infrared (NIR) measurement and container coding. In order to ensure the highest reliability and accuracy, a built-in automatic re-calibration is continuously performed using certified reference containers.
AIM 3: Compact Platform for Visual Inspection and Leak Detection
Eisai Machinery (now Bosch Inspection Technology) developed the first model of the AIM series 40 years ago. In 1985, the successful KLD series using HVLD technology was introduced. It detects leaks by measuring the electrical resistance of containers with conductive solutions. At Achema, Bosch now launches the new AIM 3, which combines both visual inspection of the AIM series and high-voltage leak detection of the KLD series. It inspects ampoules and vials containing solutions and suspensions at outputs of up to 400 containers per minute. “Our aim was to offer pharmaceutical producers and contract manufacturers from all over the world an economical solution for medium speeds at high Bosch quality standards,” Mahmoud Hamada explains.
To sort out damaged containers before they enter the main inspection turret, the AIM 3 is equipped with a pre-inspection station. The core module features a high-resolution CMOS camera with high-speed interface for particle and cosmetic inspection, as well as a re-inspection function. The customizable platform can be retrofitted on site to add further visual inspection stations or the HVLD module. The latter delivers equivalent measurement results for all glass qualities, such as molded and tubing, as well as clear and amber glass. Both
inspection steps are controlled via one common Human Machine Interface (HMI).
Related News
-
News Patients vs Pharma – who will the Inflation Reduction Act affect the most?
The Inflation Reduction Act brought in by the Biden administration in 2022 aims to give better and more equitable access to healthcare in the USA. However, pharma companies are now concerned about the other potential costs of such legislation. -
News CPHI Podcast Series: What does the changing US Pharma market mean for industry and patients alike?
In this week's episode of the CPHI Podcast Series Lucy Chard, Digital Editor for CPHI Online is joined by James Manser to discuss the political and market changes in the US pharma field. -
News CPHI Barcelona Annual Report illuminates industry trends for 2024
The CPHI Annual Survey comes into it’s 7th year to report on the predicted trends for 2024. Over 250 pharma executives were asked 35 questions, with their answers informing the industry landscape for the next year, spanning all major pharma marke... -
News Which 10 drugs are open to price negotiation with Medicare in the USA?
The Centres for Medicare & Medicaid Services, under the Biden administration in the USA, has released a list of the 10 drugs that will be open to price negotiations as part of the new legislation under the Inflation Reduction Act (IRA). -
News EU Medical Devices Regulation causes unintended disappearances of medical devices for children, doctors state
Doctor groups and associations have appealed to the EU to correct the EU Medical Devices Regulation law that may cause unintended shortages of essential drug and medical devices for children and rare disease patients. -
News 10 Major Drug Approvals So Far in 2023
Last year, 37 novel drugs were approved by the FDA, this was a high number for such a category, and covered many fields including oncology, demonstrating how promising further research is, and how it is only continuing to build. To date, there are alre... -
News Detecting Alzheimer's disease with a simple lateral flow test
A novel rapid diagnostic test for early-stage Alzheimer's disease has been developed using a biomarker binder from Aptamer Group along with technology from Neuro-Bio, the neurodegenerative disease experts. -
News CPHI Podcast Series: outsourcing and manufacturing trends
Listen to the CPHI Podcast Series this June to hear Gil Roth of the PBOA speak with Digital Editor Lucy Chard about the biggest trends and topics to watch in pharma outsourcing and manufacturing at the minute.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







