Cherwell makes an impact with new continuous monitoring microbial air samplers
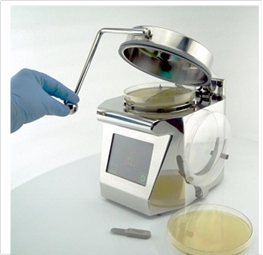
The new ImpactAir high performance microbial air sampler.
ImpactAir high performance air sampler range joins Cherwell’s portfolio of cleanroom microbiology products.
Cherwell Laboratories has signed a distribution agreement with Pinpoint Scientific for the ImpactAir range of microbial air samplers. The new ImpactAir range of high performance microbial air samplers has been designed to meet the demanding environmental monitoring requirements of aseptic processing in the pharmaceutical and healthcare industries.
ImpactAir is a state-of-the-art range of slit-to-agar microbial air samplers that fully complements Cherwell’s existing portfolio of environmental monitoring products, including SAS microbial air samplers and Redipor prepared microbiological media range. Whereas SAS samplers are ideal for rapid and frequent routine sampling regimes, ImpactAir is designed for continuous monitoring in high-grade areas where in-process sampling of viable particles is critical. Due to its unique design, ImpactAir does not shed particles, so it can be used alongside particle counting devices, and as it runs with extremely low levels of vibration, it can also be operated near balances.
By using larger 140-mm petri dishes, such as those in the Redipor range, the ImpactAir can continuously sample to a single dish over longer periods. This sampling time can be easily programmed on the touch screen user interface, from minutes up to several hours, depending on the process to be monitored. The petri dish rotates slowly under the inlet slit to present fresh agar throughout– dramatically reducing the probability of ‘twin impingement’ (where two microbes impact on an agar plate at the same point to incubate as a single colony) for enhanced accuracy and enabling an estimate of any event times during key processes.
ImpactAir is the first sampler of its kind to employ precision sensing and control to automatically adjust and maintain the distance between air inlet and agar surface, further enhancing sampling efficiency. Critically, this ensures a consistent d50 value of <0.5µm is maintained throughout an entire sampling run, as independently verified by Public Health England (PHE) following physical and biological validation.
Andy Whittard, Cherwell’s Managing Director, commented, “The new ImpactAir microbial air sampler is an exciting addition to our ‘cleanroom microbiology solutions’ offering. It fully supports our existing portfolio of quality environmental monitoring products for our pharma customers. Effective viable monitoring within high-grade areas is vitally important and ImpactAir allows users to perform high efficiency, continuous environmental monitoring. We look forward to working with the manufacturer, Pinpoint Scientific, in developing the UK and Irish markets.”
Related News
-
News Patients vs Pharma – who will the Inflation Reduction Act affect the most?
The Inflation Reduction Act brought in by the Biden administration in 2022 aims to give better and more equitable access to healthcare in the USA. However, pharma companies are now concerned about the other potential costs of such legislation. -
News CPHI Podcast Series: What does the changing US Pharma market mean for industry and patients alike?
In this week's episode of the CPHI Podcast Series Lucy Chard, Digital Editor for CPHI Online is joined by James Manser to discuss the political and market changes in the US pharma field. -
News CPHI Barcelona Annual Report illuminates industry trends for 2024
The CPHI Annual Survey comes into it’s 7th year to report on the predicted trends for 2024. Over 250 pharma executives were asked 35 questions, with their answers informing the industry landscape for the next year, spanning all major pharma marke... -
News Which 10 drugs are open to price negotiation with Medicare in the USA?
The Centres for Medicare & Medicaid Services, under the Biden administration in the USA, has released a list of the 10 drugs that will be open to price negotiations as part of the new legislation under the Inflation Reduction Act (IRA). -
News EU Medical Devices Regulation causes unintended disappearances of medical devices for children, doctors state
Doctor groups and associations have appealed to the EU to correct the EU Medical Devices Regulation law that may cause unintended shortages of essential drug and medical devices for children and rare disease patients. -
News 10 Major Drug Approvals So Far in 2023
Last year, 37 novel drugs were approved by the FDA, this was a high number for such a category, and covered many fields including oncology, demonstrating how promising further research is, and how it is only continuing to build. To date, there are alre... -
News Detecting Alzheimer's disease with a simple lateral flow test
A novel rapid diagnostic test for early-stage Alzheimer's disease has been developed using a biomarker binder from Aptamer Group along with technology from Neuro-Bio, the neurodegenerative disease experts. -
News CPHI Podcast Series: outsourcing and manufacturing trends
Listen to the CPHI Podcast Series this June to hear Gil Roth of the PBOA speak with Digital Editor Lucy Chard about the biggest trends and topics to watch in pharma outsourcing and manufacturing at the minute.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







