CHO and cell lines
CHO and cell lines Companies (4)
CHO and cell lines News
-
News ProBioGen and LAVA Therapeutics sign cell line development agreement
The agreement marks the second cell line development collaboration between the two companies -
News ProBioGen selected for development and large-scale manufacturing services
The Contract Development and Manufacturing Organization (CDMO) and technology provider will use its CHO.RiGHT expression platform for high-titer cell line development. -
News FUJIFILM Irvine Scientific launches BalanCD Gal Supplement for biotherapeutic development
Delivers enhanced galactosylation for improved protein quality, and antibody binding and function. -
News Sphere Fluidics expands into additional facility
New offices at leading science hub support the company’s rapid growth following recent investment.
CHO and cell lines Products (21)
-
Product Cell Line Development
KBI offers a full suite of CLD across both mammalian and microbial expression systems. KBI can apply established mammalian recombinant protein expression systems to provide rapid and industry-leading cell line generation services using: CHO-M Selexis SUREtechnology, CHO-ZN, CHO-DG44, CHO-S, and CHO-K1 GS. ...
-
Product Biologic drug substance CDMO services
From pre-clinical development to commercial supply, Patheon by Thermo Fisher Scientific is an industry leader in the development and manufacture of mammalian cell culture drug substances. Patheon offers biotech and pharmaceutical companies the ability to pursue opportunities around the globe with a fully i...
-
Product A CDMO PARTNER FOR LIFE
FUJIFILM Diosynth Biotechnologies is an industry-leading cGMP Contract Development and Manufacturing Organization (CDMO) supporting the biopharmaceutical industry in the development and production of biologics, vaccines and cell and gene therapies. Our focus is to combine technical leadership in process d...
-
Product Specialty Cell Culture Media, Cell Lines and Contract Services for Baculovirus Expression Vector Systems
Expression Systems, now an Advancion company, is dedicated to supplying and servicing the cell culture and bio-industrial markets with innovative cell culture media formulations, as well as cell lines, molecular tools, reagents, and contract services. The company specializes in the baculovirus expression v...
-
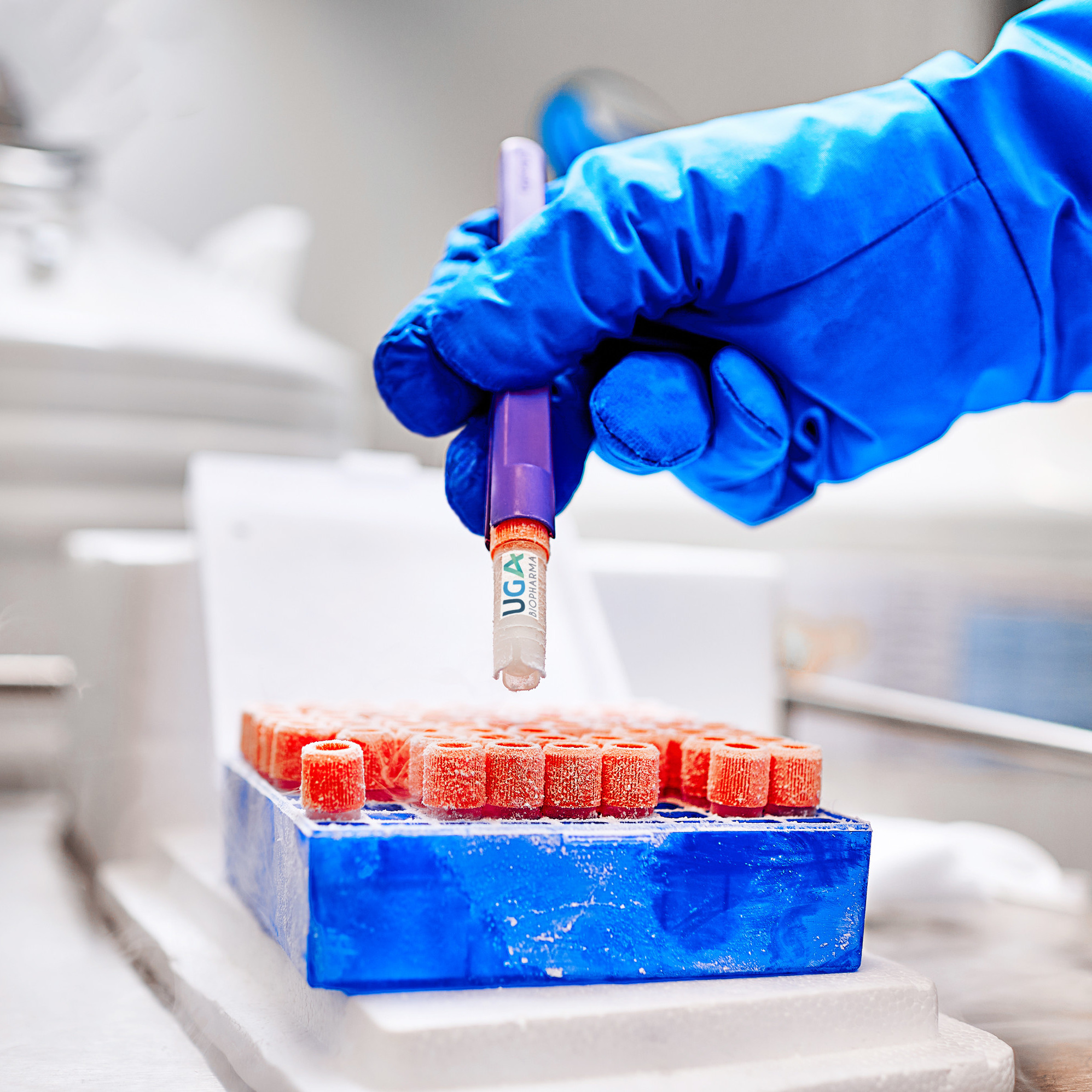
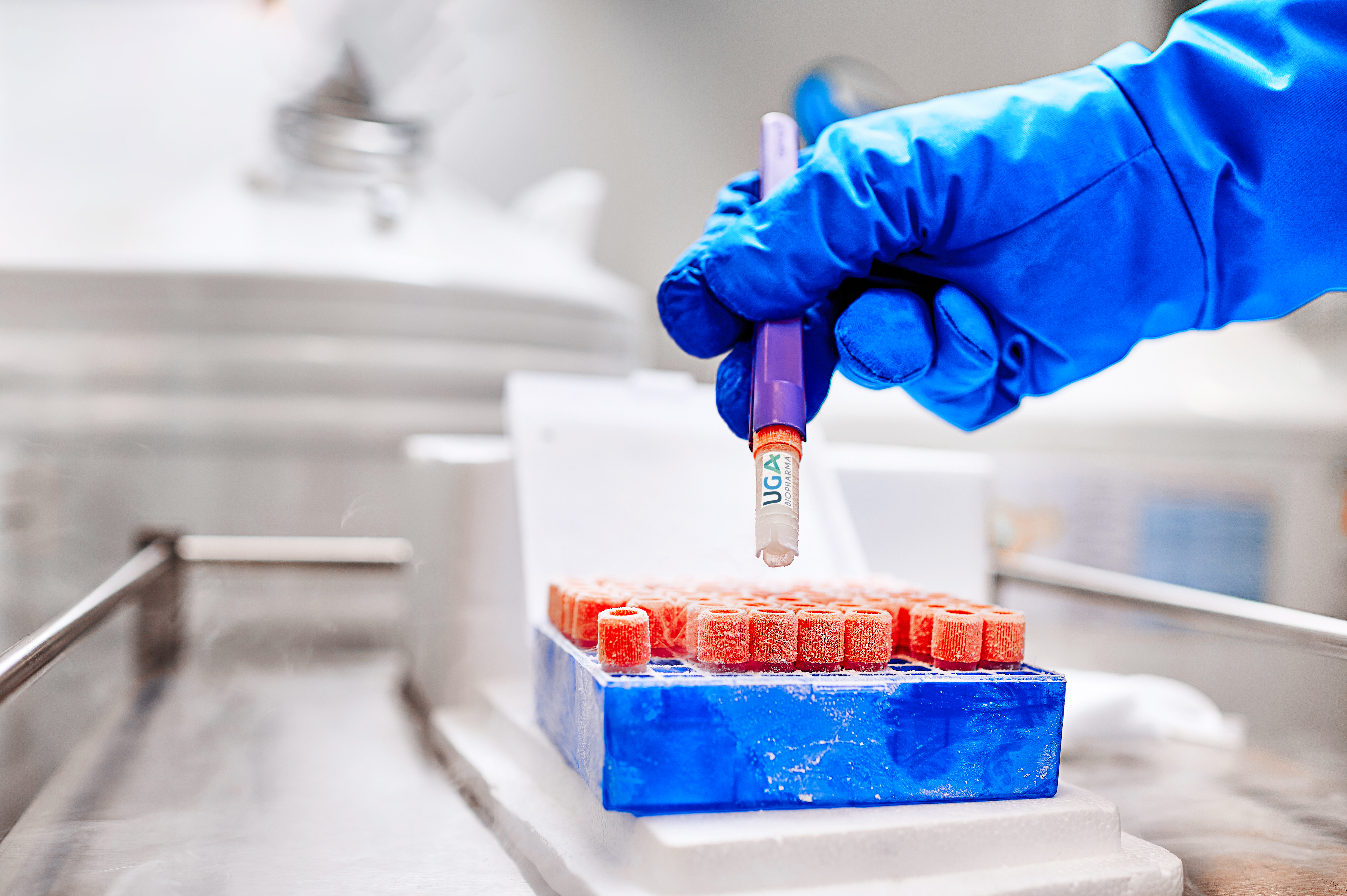
Product Ready-to Use CHO cell lines
UGA Biopharma GmbH offers to their clients so called Ready-to-use Biosimilar Cell Lines. This are in-house developed CHO cell lines stably expressing different biosimilar molecules, that are instantly available for in-licensing. The client profits, because the cell lines are directly available, and develop...
-
Product LN₂ Generator
1. Product Introduction
1) Various sensor monitoring and alarm programs, two-way communication capabilities
2) Vacuum insulation piping for cryogenic
3) Self-generated liquid nitrogen in Cryogenic environments
4) One-touch company size
5) Low noise
6) Boil off g...
-
Product E. Coli Host Cell Proteins
ELISA methods are used to test for the presence of residual host cell proteins left in a drug or therapeutic protein following purification.
-
Product Media & Feeds for CHO cell line
The First CHOice® Medium is a high-performance cell culture medium optimized for mammalian suspension cells ensuring high product quality and cell viability during all cultivation phases. In combination with the First CHOice® Feeds Alpha & Beta, offer a platform to achieve excellent cell growth, an...
-
Product Cell line development services
Cell line development services, including molecular cloning, CHO cell line development and lead clone selection, bioprocess optimization, DSP and analytical characterization of the target molecule.

-
Product Chinese Hamster Ovary Host Cell Protein Testing
ELISA methods are used to test for the presence of residual host cell proteins left in a drug or therapeutic protein following purification.
-
Product Incubator shaker
The ISF1-Z incubator shaker is suitable for applications in laboratories, research centers and production facilities. The ISF1-Z complies with GMP (good manufacturing practice) requirements and regulations. It offers a large shaking capacity with a small footprint and many features allowing straightforward...
-
Product Biosimilars
Portfolio of high-producing biosimilar cell lines and processes, off-the-shelf and generation of such from scratch. Biosimilarity optimisation of your recombinant cell line. Your benefit: Cost and time-savings, while bringing your product to the market. https://excellgene.com/offerings/biosimilars/
-
Product Cell Host Technologies
Outlicensing of superior cell hosts: CHOExpress® & HEKExpress®, cell line generation with best in class transposon technology. Your benefit: In-house state of the art tech revolutionizing your development programs. https://excellgene.com/technologies/
-
Product Bioprocess Development
Integrated upstream and downstream process development for high-yield manufacturing of biologics of desired quality. Your benefit: Cost-effective and scalable bioprocess with robust and reliable tech transfer. https://excellgene.com/offerings/downstream-process-development-dsp/ https://excellgene.com/offer...
-
Product Cancer Cell Lines- research use only
Cancer cells are transformed cells that have acquired a series of changes that permit them to form tumors. They arise when the genes responsible for regulating cell division are damaged. The uncontrolled and often rapid proliferation of cells can lead to benign or malignant tumors. Beni...
-
Product GMP Cell & Virus Bank Storage
Our GMP-compliant storage area offers dedicated or shared storage in standard freezers (-80°C), ultra-low freezers (-130°C), or in cryotank for storage in liquid nitrogen vapor phase at temperature below -150°C.
-
Product Cell & Virus Bank Characterization
The safety testing of cell or virus banks at the level of the Master, Working and End of Production Cell Bank is a key element for cell- or virus-derived biopharmaceutical products. Characterization tests aim to confirm identity, genetic stability and purity of the cell or virus bank.
-
Product GMP manufacturing of MCB/WCB/MVSS/WVSS
Clean Cells is a GMP-licensed Contract Development and Manufacturing Organization providing R&D, production and quality control testing services to the biopharmaceutical industry.
Our two-part site located in Western France holds an innovative process development platform, BSL2 and BS...
-
Product Defined Media and Reagents
Product portfolio for CHO, HEK, Vero, Hybridoma, Insect and more.
Customized media and reagents are also part of our manufacturing capabilities.

Upcoming Events
-
CPHI North America 2024
Pennsylvania Convention Center, Philadelphia
07 May 2024 - 09 May 2024 -
CPHI & PMEC China 2024
Shanghai New International Expo Center
19 - 21 June 2024 -
CPHI South East Asia 2024
Queen Sirikit National Convention Center, Bangkok, Thailand
10 Jul 2024 - 12 Jul 2024
Pharmaceutical Industry Webinars
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







.jpg)














.png)


.png)



.jpg)