CMC
CMC Companies (16)
CMC News
-
News Onyx Scientific expands small molecule CMC services with new UK facility
The contract development and manufacturing organisation (CDMO) has completed the first phase of its multi-million-pound development. -
News Inaugural Bio Integrates conference highlights industry's inefficiency in developing products
Industry leaders give voice to issues and trends shaping the biotech sector, including the importance of collaboration.
CMC Products (18)
-
Product Biologic drug substance CDMO services
From pre-clinical development to commercial supply, Patheon by Thermo Fisher Scientific is an industry leader in the development and manufacture of mammalian cell culture drug substances. Patheon offers biotech and pharmaceutical companies the ability to pursue opportunities around the globe with a fully i...
-
Product GMP and CMC Laboratory Services
Laboratory services according to GMP and CMC: We provide regulatory-driven, phase-appropriate laboratory services, supporting CMC programs from preformulation to formulation to product release. Among our capabilities are centres of excellence for method development and validation, analysis, stability s...
-
Product A CDMO PARTNER FOR LIFE
FUJIFILM Diosynth Biotechnologies is an industry-leading cGMP Contract Development and Manufacturing Organization (CDMO) supporting the biopharmaceutical industry in the development and production of biologics, vaccines and cell and gene therapies. Our focus is to combine technical leadership in process d...
-
Product Cortellis CMC Intelligence™
Cortellis CMC Intelligence product is a comprehensive database of guidelines / granular collection of CMC data requirements for initial registration of small-molecule and biologics drugs around the world that can be used to avoid delays in product approval and successfully bring a drug to the market (launc...
-
Product Hi-End Resource On Demand Solutions
Project resource management includes forecasting the need to hire and/or outsource some tasks. When projects are high-value and require strategic decisions, in-house resources may be insufficient. Similarly, administrative action items are better left to contract workers so as to not overburden your staff....
-
Product New Drug & Chemical Services
1.Custom Synthesis 2.R&D of Synthetic Processes and Custom Manufacturing 3.Contract Manufacturing Service 4.CMC Service for New Drugs

-
Product Formulation Development
BioDuro-Sundia holds more than 25 years of experience formulating poor solubility and poor permeability drugs for the clinical studies. Our comprehensive suite of advanced formulation technologies and extensive scientific know-how supports >95% of marketed dosage form...
-
Product Oligonucleotide Analytical Development Services
Analytical development services for oligonucleotides: For oligo-based drugs, our capabilities range from GLP bioanalysis to GLP/cGMP characterisation. We support product development, from quality control testing of amidite starting materials and early stage product characterization through to GMP batch r...
-
Product Nasal Drug Development
Development of nasal drugs: Scientists from our nasal drug development team provide method development, validation, and testing services to help you optimise the performance of your aqueous, powder, and propellant-driven nasal drug products. Conducted in Good Manufacturing Compliant (GMP) laborato...
-
Product Cortellis CMC Intelligence™
Cortellis CMC Intelligence™ product is a comprehensive database of guidelines / granular collection of CMC data requirements for initial registration of small-molecule and biologics drugs around the world that can be used to avoid delays in product approval and successfully bring a drug to the market (laun...
-
Product Analytical Development
BioDuro-Sundia’s Analytical Testing team offers high quality analytical services including method development and validation, qualification of reference standards, testing and release studies, stability studies, and CMC dossier preparation services. ...
-
Product Process Development
BioDuro-Sundia’s process development services support drug substance production, providing an efficient path to manufacture API or intermediates. Our extensive services support production of API for GLP toxicology studies, IND-enabling studies, clinical studies, and commercialization efforts. qq...
-
Product Hydroxypropyl Methyl Cellulose HPMC
Hydroxypropyl Methylcellulose(HPMC)
CAS NO.:9004-65-3
Hydroxypropyl Methylcellulose (HPMC) are non-ionic cellulose ether & its derivatives that have hyrdroxyl groups on the cellulose chain substituted for a methoxy or hydrox...
-
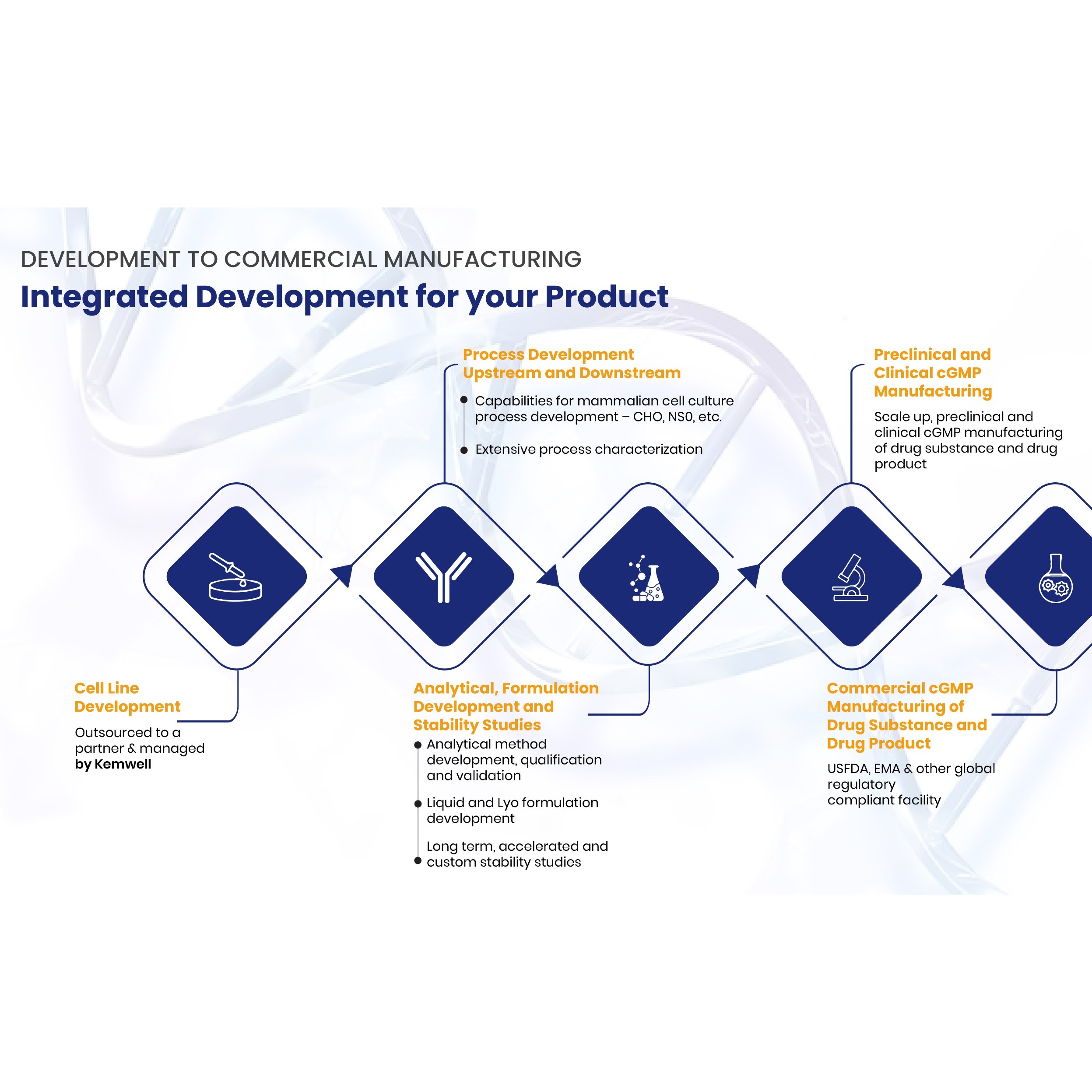
Product Kemwell Biopharma
SERVICES Kemwell provides integrated development and manufacturing services for companies that require one-stop solution for mammalian cell-culture based protein therapeutics The team is experienced to undertake end-to-end activities right from cell line development till cGMP clinical and commercial manufa...
-
Product Quality & Compliance
Our services for Quality & Compliance:
- CMC Pre and Postapproval
- Quality Systems Consulting and Operational Quality Assurance
- Audits and Inspection Readiness
- Management of Compliance

-
Product Regulatory Affairs
Our team of Regulatory affairs experts make our service offerings stronger.
Following are our Regulatory offerings:
• Regulatory Operations, Affairs
• Regulatory Intelligence, Information Management COTS
• Regulatory Strategy & Business Consulting
• Health Autho...
-
Product Submissions Assistance
EAS can assist with all pharmaceutical submissions and amendments to FDA and Health Canada. Our scientific experts in toxicology, radiology, chemistry, and biology are authorities in government laws and regulations. EAS can lead your company through the process of obtaining necessary background data, filin...
-
Product CMC Services - Chemistry, Manufacturing and Controls
BlueReg has a dedicated team of CMC consultants with significant expertise in all areas of CMC development.
Our experts can advise you on pharmaceutical development strategy and provide oversight for the overall product development process to ensure key development milestones are successfully re...
Upcoming Events
-
CPHI North America 2024
Pennsylvania Convention Center, Philadelphia
07 May 2024 - 09 May 2024 -
CPHI & PMEC China 2024
Shanghai New International Expo Center
19 - 21 June 2024 -
CPHI South East Asia 2024
Queen Sirikit National Convention Center, Bangkok, Thailand
10 Jul 2024 - 12 Jul 2024
Pharmaceutical Industry Webinars
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance


































.png)


.png)



.jpg)