Merck brand relaunched
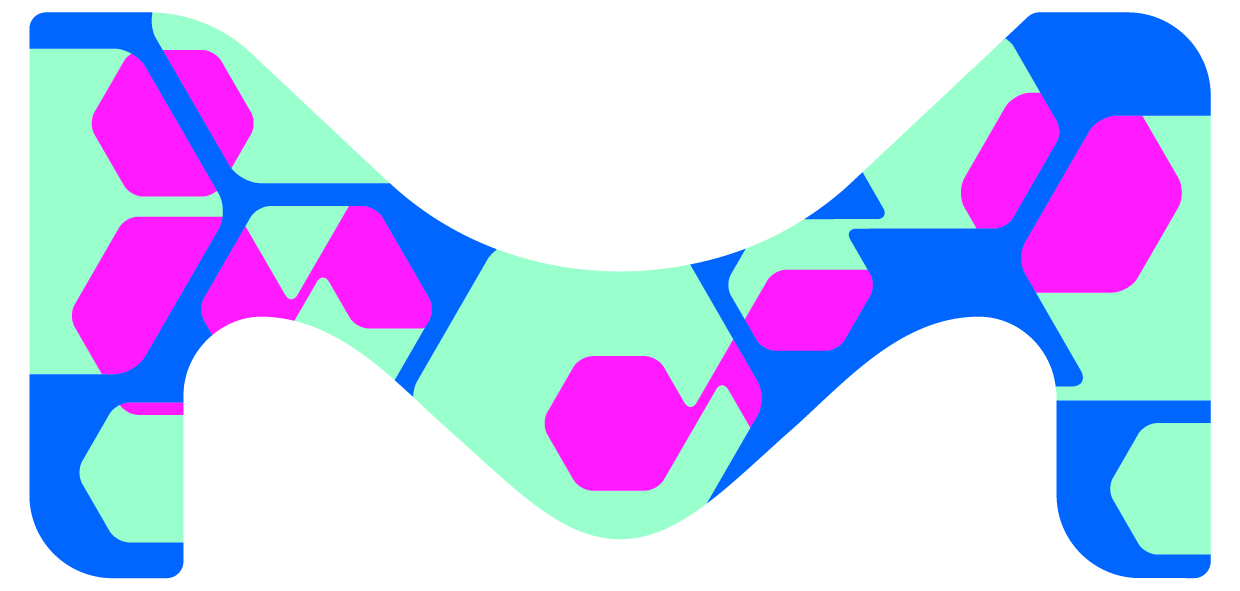
Merck Serono and Merck Millipore become Merck
Merck has announced the relaunch of its brand identity. The fundamental revision of the visual appearance, as well as the introduction of a new logo, reflect the transformation into a global science and technology company. At the same time, the brand architecture at business level has been simplified. Outside the US and Canada, the company will operate uniformly as Merck.
"Merck has fundamentally changed over the past 10 years," emphasized Karl-Ludwig Kley, Chairman of the Executive Board and CEO. "We have developed from a classic supplier of pharmaceuticals and chemicals into a global technology company. With our unique combination of highly specialized biopharmaceutical, life science and materials businesses, we are in a position today to offer solutions to support global megatrends such as health and digitization. The complete overhaul of our brand identity is to communicate this new direction vis-à-vis our customers, partners and applicants. We want to be recognizable and remain visible as Merck worldwide so as to strengthen our well-known brand name. For this we have deliberately rid ourselves of outdated features and will be focusing on a young and eye-catching image."
The investment in the Merck brand is part of the "Fit for 2018" strategic transformation and growth program, which includes the focus on innovative, technology-driven businesses as well as the modernization and expansion of global headquarters in Darmstadt, Germany. This also involves a more self-confident and at the same time clearer tone of voice, reflecting Merck's character and linking its pride in a nearly 350-year-old culture with scientific curiosity and a passion for research.
With the introduction of the new brand design, the previously independent divisional brands Merck Serono and Merck Millipore will be eliminated. In the future, Merck Serono will operate as the biopharmaceutical business of Merck, and Merck Millipore as the life science business of Merck. "Following the two major acquisitions, the Merck Serono and Merck Millipore brands helped us to position the duality of existing and acquired businesses in the marketplace. We succeeded in doing so. That's why we are returning to the brand that we have been known under for nearly 350 years," Kley added. Product brands such as Erbitux or MilliQ will not be affected by this change.
Merck holds the rights to the Merck name and brand globally. The only exceptions are the US and Canada. The company will therefore continue to operate in these two countries as EMD Serono in the biopharmaceutical business, as EMD Performance Materials in the high-tech materials business, as well as EMD Millipore in the life science business up until the planned acquisition of Sigma-Aldrich has been completed.
Merck's visual image is fundamentally new. It is based on the concept of a vibrant science and technology company, and is inspired by the colourful and multifaceted shapes seen under a microscope. The new Merck design features expressive colours and shapes with a scientific look-and-feel. These are also reflected by the new company logo. In comparison with the former logo, it makes a bold statement; its design is less complex and can be used in different colours.
"A comprehensive external and internal analysis showed that we must emotionalize our brand appearance to a much greater extent in order to be perceived as a vibrant technology company in the market and by applicants," said Walter Huber, Head of Group Communications.
In addition, the new corporate design is to create a strong visual link to the Merck businesses in the US and Canada. For this reason, the company is also introducing a striking, multicoloured "M" in addition to the Merck logo. It is to indicate the membership of businesses and products to the Merck Group, independent of company names or regions.
Related News
-
News Patients vs Pharma – who will the Inflation Reduction Act affect the most?
The Inflation Reduction Act brought in by the Biden administration in 2022 aims to give better and more equitable access to healthcare in the USA. However, pharma companies are now concerned about the other potential costs of such legislation. -
News CPHI Podcast Series: What does the changing US Pharma market mean for industry and patients alike?
In this week's episode of the CPHI Podcast Series Lucy Chard, Digital Editor for CPHI Online is joined by James Manser to discuss the political and market changes in the US pharma field. -
News CPHI Barcelona Annual Report illuminates industry trends for 2024
The CPHI Annual Survey comes into it’s 7th year to report on the predicted trends for 2024. Over 250 pharma executives were asked 35 questions, with their answers informing the industry landscape for the next year, spanning all major pharma marke... -
News Which 10 drugs are open to price negotiation with Medicare in the USA?
The Centres for Medicare & Medicaid Services, under the Biden administration in the USA, has released a list of the 10 drugs that will be open to price negotiations as part of the new legislation under the Inflation Reduction Act (IRA). -
News EU Medical Devices Regulation causes unintended disappearances of medical devices for children, doctors state
Doctor groups and associations have appealed to the EU to correct the EU Medical Devices Regulation law that may cause unintended shortages of essential drug and medical devices for children and rare disease patients. -
News 10 Major Drug Approvals So Far in 2023
Last year, 37 novel drugs were approved by the FDA, this was a high number for such a category, and covered many fields including oncology, demonstrating how promising further research is, and how it is only continuing to build. To date, there are alre... -
News Detecting Alzheimer's disease with a simple lateral flow test
A novel rapid diagnostic test for early-stage Alzheimer's disease has been developed using a biomarker binder from Aptamer Group along with technology from Neuro-Bio, the neurodegenerative disease experts. -
News CPHI Podcast Series: outsourcing and manufacturing trends
Listen to the CPHI Podcast Series this June to hear Gil Roth of the PBOA speak with Digital Editor Lucy Chard about the biggest trends and topics to watch in pharma outsourcing and manufacturing at the minute.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







