Organoid Biobank for Predicting Cancer Patient Response to Drugs
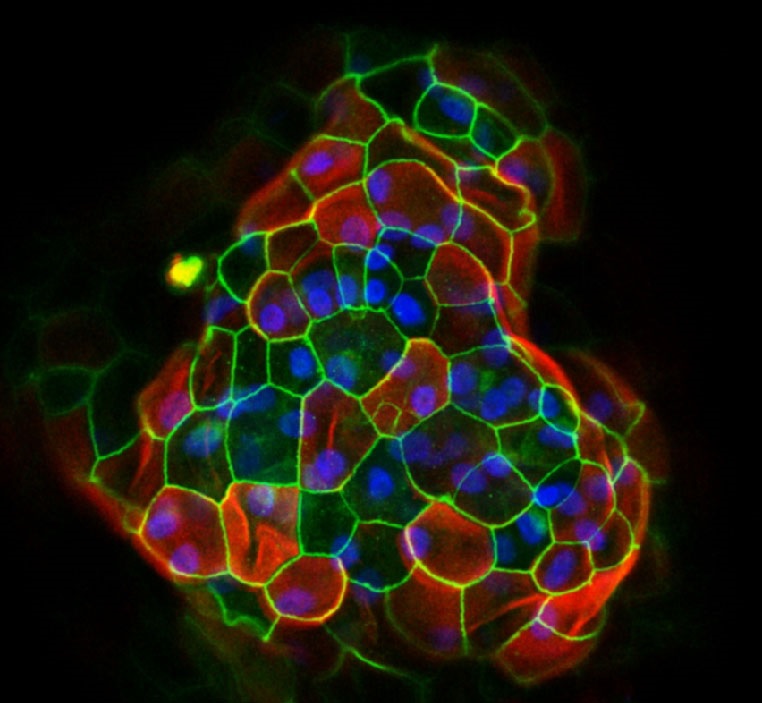
AMSBIO reports on a study, published in Cell, that demonstrates the power of organoids to capture, in three dimensions, the multiple mutations that occur in tumours. Organoids, small clusters of cells that accurately mimic the behaviour of human tissue, can be used to test cancer drugs and, eventually, to identify effective personalised treatments for patients.
In the described research samples from healthy tissue and cancerous tissue were taken from 20 patients with colorectal carcinoma and used to form the organoids. The organoids were then exposed to a range of colorectal cancer treatments as part of a high-throughput drug screen, which is testing hundreds of different drugs against cancer cell lines.
Until now, cancer drug screening has primarily been carried out using 2D cell lines grown in dishes, or in mouse models. Organoids, which more closely resemble human tumours than cell lines, and are less time and resource intensive than mouse models, offer researchers a middle ground between existing approaches. These exciting findings are the result of an international collaboration involving research teams in the UK, Netherlands and the US.
The collaborative research teams are now building a biobank of organoids that will help to illuminate the complex interactions between the multiple genomic alterations in tumours that determine which drugs work and which don't. Organoids could ultimately be used in the clinic to predict how a patient will respond to treatment. However, researchers say that more work to speed up and standardise the process of producing and testing organoids is needed before this is possible. In the short term, organoids are likely to speed up the process of developing new cancer treatments and reduce costs.
AMSBIO’s wide range of matrices and other solutions for 3D cell culture include natural hydrogels, recombinant matrices, artificial scaffolds and scaffold-free supports. Recently AMSBIO introduced two novel formulations of Basement Membrane Extract (BME2 and BME3) to address the heterogeneous nature of tissue and organ microenvironments that not only arise from organ specific stromal cells, growth factors, proteoglycans, and protein composition but also from the stiffness or tensile strength of the BME. Matrices from AMSBIO not only support cells and cell layers, but also play an essential role in tissue organization that affects cell adhesion, migration, proliferation, and differentiation. These aspects can be studied with tools from AMSBIO and the company’s experience in developing specialist matrices includes regulatory friendly solutions for regenerative medicine applications where customers are already in clinical trials.
Related News
-
News Patients vs Pharma – who will the Inflation Reduction Act affect the most?
The Inflation Reduction Act brought in by the Biden administration in 2022 aims to give better and more equitable access to healthcare in the USA. However, pharma companies are now concerned about the other potential costs of such legislation. -
News CPHI Podcast Series: What does the changing US Pharma market mean for industry and patients alike?
In this week's episode of the CPHI Podcast Series Lucy Chard, Digital Editor for CPHI Online is joined by James Manser to discuss the political and market changes in the US pharma field. -
News CPHI Barcelona Annual Report illuminates industry trends for 2024
The CPHI Annual Survey comes into it’s 7th year to report on the predicted trends for 2024. Over 250 pharma executives were asked 35 questions, with their answers informing the industry landscape for the next year, spanning all major pharma marke... -
News Which 10 drugs are open to price negotiation with Medicare in the USA?
The Centres for Medicare & Medicaid Services, under the Biden administration in the USA, has released a list of the 10 drugs that will be open to price negotiations as part of the new legislation under the Inflation Reduction Act (IRA). -
News EU Medical Devices Regulation causes unintended disappearances of medical devices for children, doctors state
Doctor groups and associations have appealed to the EU to correct the EU Medical Devices Regulation law that may cause unintended shortages of essential drug and medical devices for children and rare disease patients. -
News 10 Major Drug Approvals So Far in 2023
Last year, 37 novel drugs were approved by the FDA, this was a high number for such a category, and covered many fields including oncology, demonstrating how promising further research is, and how it is only continuing to build. To date, there are alre... -
News Detecting Alzheimer's disease with a simple lateral flow test
A novel rapid diagnostic test for early-stage Alzheimer's disease has been developed using a biomarker binder from Aptamer Group along with technology from Neuro-Bio, the neurodegenerative disease experts. -
News CPHI Podcast Series: outsourcing and manufacturing trends
Listen to the CPHI Podcast Series this June to hear Gil Roth of the PBOA speak with Digital Editor Lucy Chard about the biggest trends and topics to watch in pharma outsourcing and manufacturing at the minute.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







