News
19 Jul 2013
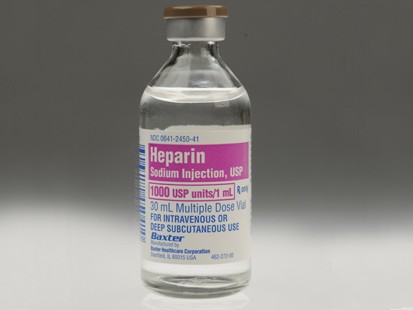
Rx-360's Summary of FDA 's Recently Published New Guidance for Industry, 'Heparin for Drug and Medical Device Use: Monitoring Crude Heparin for Quality'
Related News
-
News New WHO health emergency guidelines expect full transparency from Big Pharma
The WHO are proposing a new set of pandemic guidelines to set out how future global health crises should be handled. -
News Biden's $2 billion Biotech Boost
President Biden issues executive order to bolster U.S. biomanufacturing sector. -
News US FDA grants priority review for Eisai and Biogen's Alzheimer's drug
Decision by US drug regulator on lecanemab expected by early next year -
News Panaxia's medical cannabis facility in Malta gains EU-GMP standard
This second facility gives the company a significant power multiplier for export capacity and geographical reach -
News CPHI Discover: Remote excipient audits are on the increase, but we must do more to develop novel excipients
Ahead of the session, Value Added Excipients to Unlock the Potential of APIs, at CPHI Discover on Thursday 18th May – global pharma’s largest ever virtual gathering – we spoke with one of the speakers, Dr Iain Moor... -
News Pharma Explained: What is Quality Risk Management & cGMPs?
Need clarity on determining your QA from your QbD and your cGMPs? You’re not alone! In our new Pharma Explained series from CPHI, we bring you clear cut definitions from industry experts on a myriad of Pharma Terms, delivered in bitesize video presenta... -
News Former FDA Commissioner lays out roadmap for building out excess capacity to deal with pandemic situations
Dr Scott Gottlieb shares insights into COVID-19 vaccine development, the upcoming US election and pharma pricing at the CPHI Festival of Pharma -
News Successful MHRA regulatory inspection for Scottish CMO Symbiosis
Inspection conducted remotely using video-conferencing and an online private document sharing platform.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







