CDMO Services
Product Description
ANGELINI PHARMA SPA

-
IT
-
2015On CPHI since
-
1000 - 4999Employees
Company types
Categories
ANGELINI PHARMA SPA

-
IT
-
2015On CPHI since
-
1000 - 4999Employees
Company types
More Products from ANGELINI PHARMA SPA (3)
-
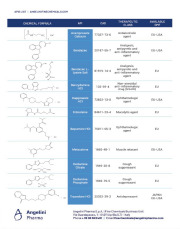
Product APIs List
Characterized by quality, cost-competitiveness and reliability with an uncompromising commitment to cGMP standards and HSE regulations, Angelini Fine Chemicals makes unique its generics. With DMFs filed worldwide, Angelini Fine Chemicals supplies its APIs to top ranking pharmaceutical industry in Europe... -
Product FlowChem Platform
Angelini Fine Chemicals is one of the first European CDMOs investing in flow chemistry and micro-reaction technology for development and manufacturing of APIs and HPAPIs. Angelini Fine Chemicals brings extensive experience in research and development of flow continuous process and offers extensive custo... -
Product HPAPI Platform
Responding to the rising number of custom HPAPIs demand, Angelini Fine Chemicals expands its HPAPI capacity. With over 50 years of CDMO experience, Angelini Fine Chemicals is at the forefront of chemical development and cGMP manufacturing of HPAPIs to serving brand pharmaceutica industry. The new Angel...
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance