Pioneering Separations Company Quad Technologies Set To Revolutionize Cell Isolation Following Successful Investment
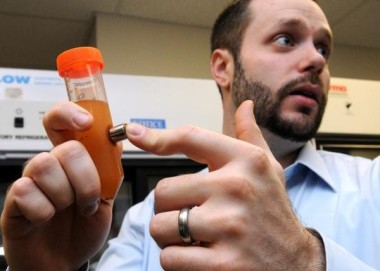
Quad Technologies Inc., developer of a novel separation technology, has announced its entry into the cell separation market. Focused on overcoming barriers associated with conventional cell purification techniques, the company boasts remarkable scientific expertise, a wealth of business experience and comprehensive financial backing. Led by visionary CEO Dr Sean Kevlahan, the company is currently building on successful manufacturing and feasibility studies of QuickGel, its lead patent-pending technology.
Quad Technologies is set to have initial major impact in the multimillion dollar market of cell separation, addressing the growing frustration amongst researchers with the low viability and efficiency of conventional techniques. These techniques can cause modifications to the cell structure and render it unviable for further development, and can have significant time and cost implications. Quad Technologies’ ground-breaking QuickGel is a dissolvable hydrogel technology that, for the first time, allows researchers to maintain the viability and phenotype of rare isolated cells. The company sees additional near term opportunities in protein purification and nucleic acid extraction.
“With our QuickGel technology in hand, Quad is poised for an exciting period of aggressive growth”, commented CEO, Sean Kevlahan who developed the technology while undertaking his PhD at Northeastern University. “Recent validation of our technology at world class beta test sites affords us great confidence in our go-to market strategy. We are looking forward to seeing the impact of our high value cell separation technology in areas such as stem cell research. Having already been widely published, the product is due to be made available later this year and we expect that it will enable major scientific breakthroughs to be achieved...”
Dr Jeffrey Chalmers, Director of the Ohio State University Medical School Cell Separation Core Facility and Professor of Chemical Engineering, who is a member of Quad Technologies’ scientific advisory board explained: “The initial testing of the QuickGel coated beads that we carried out for Quad Technologies clearly demonstrated the capabilities of the technology in overcoming previously unresolvable issues associated with conventional cell isolation techniques. QuickGel will enable developers of the new generation of therapeutics and diagnostics to achieve higher efficiency, cell viability and purity and I am extremely excited to be involved with the company at this time.”
Related News
-
News BioNTech to begin mRNA vaccine manufacturing in Rwanda by 2025
German biotechnology company BioNTech has stated their intentions to begin production at their mRNA vaccine factory in Rwanda by 2025, which will mark the first foreign mRNA vaccine manufacturing site on the continent of Africa. -
News Identifying Alzheimer’s Disease biomarker proteins with whole blood tests
A University of Manchester spin-out pharmaceutical company, PharmaKure, has reported successful study results for the quantification of Alzheimer’s Disease biomarker proteins with a whole blood test. -
News Bill & Melinda Gates Foundation to boost mRNA vaccine initiatives in Africa with USD $40m
To address vaccine inequality and accessibility issues, the Bill & Melinda Gates Foundation aims to deliver USD $40m to various biotech companies and vaccine manufacturers in support of mRNA vaccine development. -
News CPHI Podcast Series: Exploring neurological frontiers in Alzheimer's and beyond
The next episode of the CPHI Podcast Series delves into the science and background behind some recent developments in the field of Alzheimer's disease and neurological disorders. -
News Is patient centricity the future of pharmaceutical manufacturing?
In this interview with Sandra Sánchez y Oldenhage, President of PharmAdvice, she speaks to the importance of considering patients in the manufacturing stages of the pharmaceutical supply chain, and how it can redefine healthcare. -
News CPHI Podcast Series: How to leverage AI for Drug Discovery
Artificial intelligence is the topic of debate in the latest episode from the CPHI Podcast Series, where Digital Editor Lucy Chard speaks with Bill Whitford of DPS Group about the integration of AI in healthcare. -
News Pfizer forges ahead with blood cancer therapy after approval from FDA
Pfizer gains accelerated approval from the US FDA for their new bispecific antibody therapy for multiple myeloma, set to address an unmet need for patients. -
News Alzheimer's drug donanemab deemed effective in landmark clinical trial
Results from the TRAILBLAZER-ALZ 2 Randomised Clinical Trial into the use of donanemab to treat early symptoms of Alzheimer’s disease have been analysed.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance







