Video
17 Sep 2020
Dabired - ready-to-fill Dabigatran pellets
MP4 42.1 MB
Content provided by our supplier
Dr. Reddy's Laboratories SA

-
CH
-
2015On CPHI since
-
3Certificates
-
5000+Employees

Company types
Primary activities
Other Content from Dr. Reddy's Laboratories SA (8)
-
News Pharma Explained: Active Pharmaceutical Ingredients
What are APIs? As the name suggests, Active Pharmaceutical Ingredients (APIs) are the heart of any dosage form, whether they are oral, injectable or inhaled, and are absolutely essential to the development of a formulated product. -
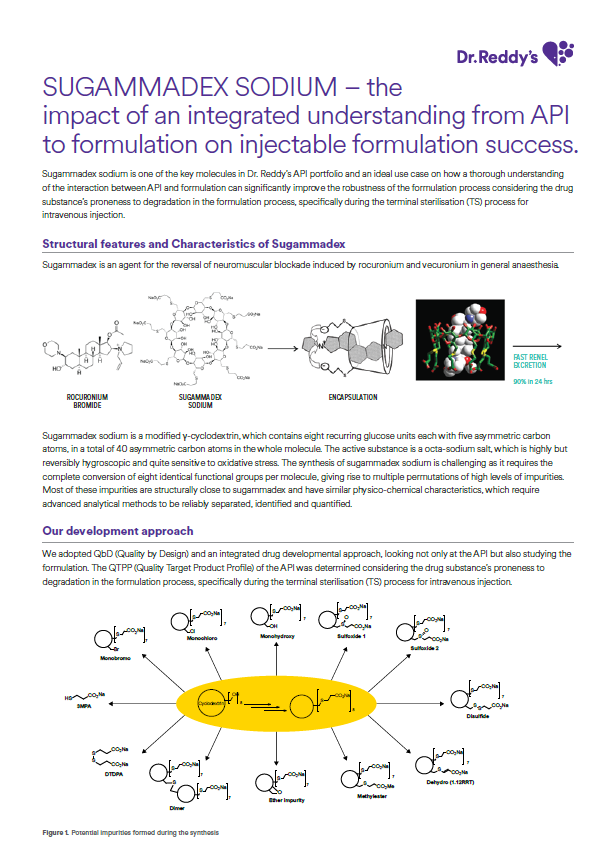
Brochure Sugammadex Sodium API article
Sugammadex sodium is one of the key molecules in Dr. Reddy’s API portfolio and an ideal use case on how a thorough understanding of the interaction between API and formulation can significantly improve the robustness of the formulation process considering the drug substance’s proneness to degradation in the formulation process, specifically during the terminal sterilization (TS) process for intravenous injection. -
News Live Webinar: Strengthening a sustainable API supply chain performance.
At Dr. Reddy’s, we’re in the business of ensuring good health for both people and the planet. Our experts will share insights on our sustainability approach and on how it helps B2B customers support their sustainable supply chain performance while sourcing APIs. Please register here: -
Brochure Dutasteride
• We offer polymorphic crystalline form 2.• A novel synthetic process developed for crystalline form 2.• Quality by design (QBD) based API development for a consistent quality profile -
News Dr. Reddy’s partners with FUJIFILM and Global Response Aid for Avigan® (favipiravir), a potential treatment of COVID-19
Dr. Reddy's Laboratories Ltd. announces partnering with FUJIFILM Corporation through FUJIFILM Toyama Chemical Co. Ltd. and Global Response Aid for the development, manufacture and sales of Avigan® Tablets (generic name: favipiravir), a potential treatment of COVID-19. -
Brochure Pregabalin API
Crystalline form-I i.e., Innovator form.• The control strategy for the API impurity pro le has been designed through quality-by-design (QbD) based development.• Lactam impurity levels are not detected, thus able to achieve USP/ EP compliance after milling - opening a possibility for a wide range of delivery systems -
Webinar Strengthening Diversity, Equity, and Inclusion in Pharma
The pharma industry is aware that it needs to improve the representation of women and minority communities in its businesses. The benefits of diverse teams are clear, yet it isn’t an easy road. How can companies create an environment where people feel safe, thrive, and attract the best?
Approaching Diversity and Inclusion as part of a wider Sustainability / Environment, Social, and Governance (ESG) performance is an essential indicator of pharma companies' commitment to driving I&D for a more diverse and successful workplace, which ultimately also ensures to better serve patients around the world. -
Video Innovation Across Borders: How Pharma and innovators are fighting back against COVID-19 and ways forward
A global pandemic demands a fast response. The research-based pharmaceutical industry is working upstream at extraordinary speed to develop a safe and effective vaccine for COVID19. Pharmaceutical companies around the globe are joining forces and speeding up the process by conducting phases and development in parallel and forging new partnerships and collaboration. The pharmaceutical and life science industry has faced the need to leverage science and technology, developed pre and during the pandemic. Firms are coming up with new ways to maintain production despite disrupted supply chains, or they are mobilizing to design new products as demand for existing products collapses. This discussion will examine how the COVID-19 pandemic is driving pharma and life science companies to collaborate, innovate and prepare for an uncertain future. This webinar was originally aired as part of the CPHI Festival of Pharma.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance