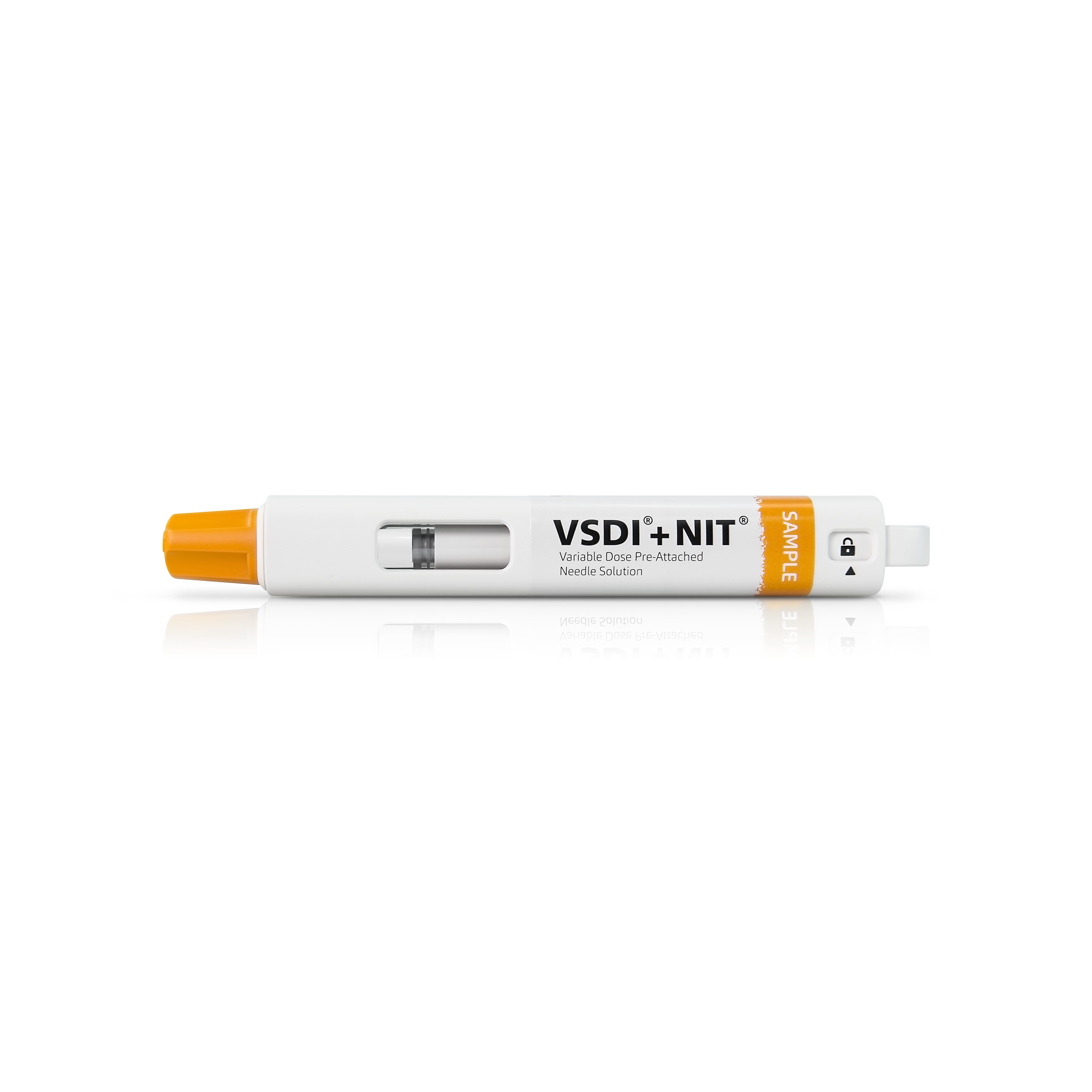
VSDI®+NIT® Autoinjector
Product Description
SHL Medical

-
CH
-
2020On CPHI since
-
2Certificates
-
5000+Employees
Company types
Categories
SHL Medical

-
CH
-
2020On CPHI since
-
2Certificates
-
5000+Employees
Company types
More Products from SHL Medical (9)
-
Product Molly® Modular Platform Autoinjector
Molly is a single-dose autoinjector technology developed to help pharma and biotech companies reduce initial investments and expedite development timelines. Built on a proven modular platform, Molly reduces risks during the development process while increasing flexibility in device design and production. A... -
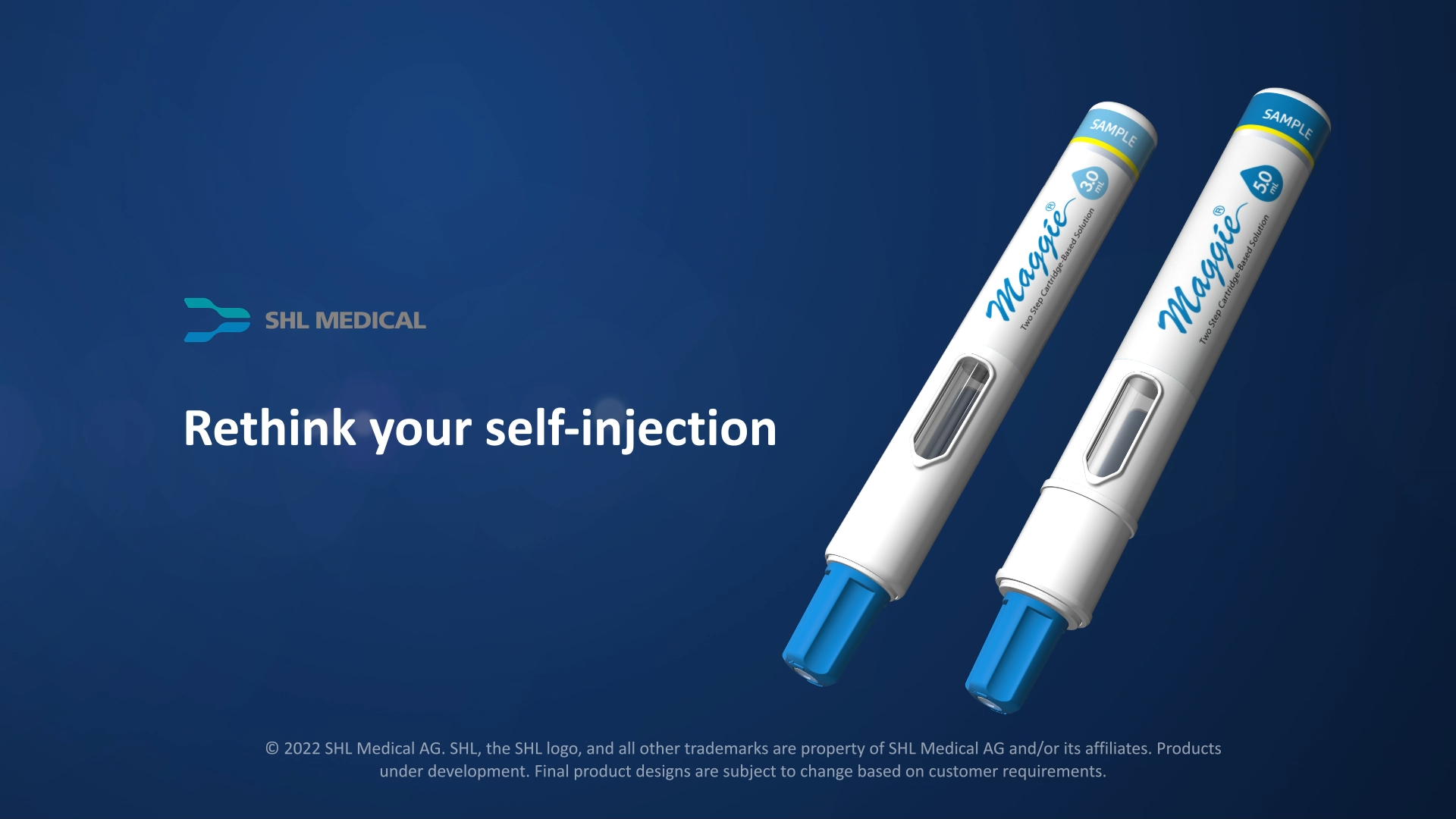
Product Maggie® 5.0 Autoinjector
Maggie® 5.0 is an innovative, large-volume, cartridge-based autoinjector built with SHL’s market-proven Needle Isolation Technology (NIT®). A two-step autoinjector, it was developed with a focus on opening a new pathway for handheld devices to accommodate a wider variety of formulation characteristics, suc... -
Product Maggie® Autoinjector
Maggie is a cartridge-based autoinjector that accommodates fill volumes of up to 3.0 mL. Built with SHL’s market-proven Needle Isolation Technology (NIT®), this 2-step autoinjector uses a pre-attached and sterile needle housed within the device cap. The needle automatically attaches to the cartridge upon c... -
Product Needle Isolation Technology (NIT®)
NIT® is a safety solution that eliminates the need for manual needle attachment in cartridge-based injections. It enables cartridges to be built into autoinjectors, supporting treatments beyond the 2.25 mL fill range and lyophilized drugs requiring dual-chamber containers.
Products built with NIT ... -
Product Autoinjector development expertise and capabilities
SHL Medical’s commitment to innovation has driven our leadership in the drug delivery industry. We offer vertically integrated expertise and capabilities to ensure the production of our self-injection systems are delivered with unparalleled speed, quality, and reliability.
Learn more about SHL... -
Product InsulCheck® DOSE Pen Injector Add-On
InsulCheck DOSE by Innovation Zed is a reusable add-on device that automatically transfers injection time and dialed dose units to a mobile application. Built with a customizable sleeve and firmware over-the-air (FOTA) capability, InsulCheck dose can accommodate any pen injector and continuously imp... -
Product SDI MIX®+NIT® Autoinjector
SDI MIX+NIT is a dual-chamber disposable autoinjector built with SHL’s NIT (Needle Isolation Technology), which allows cartridge-based injectors to be built into easy-to-use autoinjectors. To accommodate drug formulations that need to be reconstituted prior to patient administration, the device features a ... -
Product Elexy™ autoinjector
Elexy™ is a versatile, electromechanical autoinjector that features a reusable power unit compatible with drug cassettes containing a pre-filled syringe or a cartridge. Its versatility enables a wider choice of intended injection profiles, including drug volume, drug viscosity, and injection speed. In ... -
Product Molly® Connected Cap Autoinjector
The Molly® Connected Cap is a compact, retrofittable autoinjector add-on that records and transmits data on patients’ use of the device. Upon cap removal from the Molly device, the Connected Cap becomes active, allowing timestamped data to be relayed either through a smartphone app or smart data transmissi...
SHL Medical resources (22)
-
News SHL Medical releases the 2023 sustainability report
SHL Medical is pleased to announce the release of its 2023 sustainability report, which details the company’s ongoing efforts and accomplishments in sustainability and innovation. -
Video Make Every Injection Count with Molly® Connected Cap
Ready to embark on a journey where connectivity meets autoinjectors? Dive into the world of SHL Medical’s Molly® Connected Cap and experience the future of healthcare. Driven by its experience with commercializing over three dozen autoinjectors worldwide, including 17 combination product projects from the market-proven Molly modular platform technology, SHL Medical developed the Molly Connected Cap autoinjector – an agile solution that supports remote adherence monitoring for personalized care.
Explore the webpage to learn how Molly Connected Cap can help transform patients’ treatment journey: https://bitly.ws/32ZBu -
News SHL Medical and AARDEX Group forge strategic alliance to interface medication adherence software with connected self-injection solutions
SHL Medical and AARDEX Group announced their strategic partnership aimed at delivering an end-to-end solution for pharma customers seeking to demonstrate patient adherence within clinical trials. -
Video Molly® Modular Platform Autoinjector
SHL embarked on a journey of transformation with the Molly® autoinjector. After successfully supporting the development of more than a dozen combination products globally, we decide to take the innovation up a notch.
THIS is the next generation of Molly and Molly 2.25. Built upon a modular platform technology, Molly now offers flexibility beyond a conventional platform model that spans across the development processes.
Get an in-depth understanding of how Molly works to address the varying requirements of the industry and learn more about SHL's offerings at our website.
Visit the Molly website for more information: https://bit.ly/3FkJtzm -
News Aptar Digital Health and SHL Medical enter strategic partnership to optimize the patient experience with self-injectable therapies
SHL Medical and AARDEX Group announced their strategic partnership aimed at delivering an end-to-end solution for pharma customers seeking to demonstrate patient adherence within clinical trials. -
Video Maggie®: Rethink Your Self-Injection
The constant evolution of drug formulations across various disease areas opens a wider avenue for subcutaneous drug delivery. SHL Medical's Maggie® is a cartridge-based autoinjector that requires just two steps to operate. Built with the market-proven Needle Isolation Technology (NIT®), the device can accommodate varying formulations with volumes up to 3 mL. Following that, SHL Medical is excited to introduce Maggie 5.0 − the newest member of our NIT device family, developed to potentially accommodate injection volumes previously not considered by traditional autoinjectors. -
News Committed to a sustainable future: SHL Medical’s Sustainability Report 2022
SHL Medical announced today the release of its Sustainability Report for 2022. The report provides a comprehensive overview of the company’s efforts to promote sustainability and outlines its commitment to a sustainable future. -
Brochure Your drug delivery partner with proven track record
From design to mass production, SHL Medical's end-to-end device development infrastructure ensures the lasting value of self-injection systems that we design and develop with our customers. Our comprehensive in-house manufacturing capabilities and expertise include every core competency and value-added services across the product lifecycle. 30 years of pioneering the drug delivery industryOver 50 combination products launchedTop 25 bio/pharmaceutical companies in partnership Over 2,000 granted patents in current portfolio -
News SHL Medical acquires 100% of the shares in Swiss company LCA Automation
SHL Medical announces the acquisition of LCA Automation AG, a Swiss innovative automation solutions provider. The acquisition is SHL Medical’s response to the growing market demand for drug delivery solutions and will support its manufacturing operations globally, especially the upcoming Swiss manufacturing site in Zug. -
Technical Data SHL Medical Products
Working with leading biotechnology and pharmaceutical companies, we develop advanced drug delivery systems, including disposable and reusable injection devices and systems with fixed or variable dosing, high dose accuracy, and the ability to accommodate high volumes and high viscosities.
-
News SHL Medical and MoonLake Immunotherapeutics collaborate to develop sonelokimab autoinjector
SHL Medical, a world-leading provider of advanced drug delivery solutions, announced that it has signed a collaboration agreement with MoonLake Immunotherapeutics, a clinical-stage biotechnology company focused on creating next-level therapies for inflammatory diseases, to develop an autoinjector for clinical and potential commercial supply of MoonLake’s Nanobody® sonelokimab based on SHL Medical’s Molly® autoinjector technology. -
Video Final Assembly, Labelling and Packaging
SHL’s CMO services were established to create added value for our partners following the successful design and development of their device, resulting in a fully integrated service from device design to commercialization.
-
News SHL Medical further strengthens vertical capabilities with the acquisition of US manufacturer Superior Tooling Inc.
Continuing its ambitious growth journey, SHL Medical has acquired Superior Tooling Inc., a US-based manufacturing company specializing in plastic injection molds. The integration of Superior Tooling will strengthen the SHL’s inhouse manufacturing capabilities, particularly for its upcoming US manufacturing site in North Charleston, SC, scheduled to begin operations in mid-2024. -
Video The Makings of an Autoinjector
SHL’s commitment to our partners as well as the patients they serve stems from a culture deeply rooted in our corporate vision − that of delivering quality and leadership through innovative solutions that empower people.
-
News SHL Medical acquires Swiss toolmaking company SMC Mould Innovation
SHL Medical, a leading provider of drug delivery solutions worldwide, furthers its expansion strategy by acquiring SMC Mould Innovation, a Swiss manufacturer specialising in high-performance injection molding tools, based in Hallau. -
Video The SHL Experience
Through an ensemble of rich multimedia content introducing our expertise and capabilities, visitors will be able to see our machine systems and experts at work on developing the devices that we are so proud of.
-
News ten23 health® and SHL Medical announce a strategic partnership agreement for integrated services
SHL Medical, a world-leading provider of drug delivery devices, and ten23 health®, a leading Swiss Contract Development and Manufacturing Organization (CDMO) for sterile drugs, announced strategic partnership to provide a streamlined offering for pharmaceutical and biotech companies that require sterile drug/device combination products. -
Podcast Podcast: Leveraging data science for sustainable autoinjector development
Sustainability is becoming increasingly important in the pharma and biopharma industries, with many companies working to incorporate more eco-friendly materials, minimise waste and improve social impact. One such company is SHL Medical, a producer of advanced delivery devices, who are utilising data science to advance their sustainability goals. In this podcast, we discuss the relationship between data and sustainability and hear from SHL how their data-driven approach has produced ‘win-win scenarios’ for the company, its customers and the planet. -
News SHL Medical partners with SteriPack Contract Manufacturing to set up final assembly service
SHL Medical, a world-leading solutions provider of advanced drug delivery systems and SteriPack Group, renowned global supplier of secondary packing and final assembly services to the pharmaceutical industry, enter a non-exclusive strategic partnership. -
Brochure Molly Connected Cap Autoinjector Datasheet
The Molly® Connected Cap is a compact, retrofittable autoinjector add-on that records and transmits data on patients’ use of the device. -
Video A Drug-Agnostic Electromechanical Device Platform to Facilitate the Flexible Development of Combination Products
In this presentation, we introduce our most recent development – a versatile, reusable, and connected electromechanical solution that upholds the concept of drug agnosticism, platform flexibility, ease-of-use, as well as device programmability. By leveraging technological application expertise, we demonstrate how an electromechanical, versatile device platform can flexibly host different primary containers and formulations. Here, we also demonstrate how adding programmable firmware into the device circumvents formulation uncertainties across the pre-clinical, clinical, and commercial development of the drug counterpart. Finally, we show how the conserved element of the device results into an autoinjector solution that flexibly addresses various primary containers, therapy areas, regional healthcare landscapes, as well as patient groups. We also discuss an optional function for tracking injection-related data, opening pathways to gather detailed insights into the patient experience, and ultimately opening possibilities to connect such device usage data with other digital health solutions. -
Webinar The Integration of Digital With Mechanical: Explorations of Connectivity and Data Generation via Our Innovation Partnership Program for the Molly Autoinjector
Abstract: Digital health solutions create myriad possibilities in terms of enabling “beyond the pill” health outcomes. However, the path to streamlining a strategic approach between pharma and its development partners toward creating smart combination products remains a challenge. With a general consensus that developers must accurately determine in advance what users want, many companies wait upon the success of early players as a determinant to foray into digital health. This philosophy, however, disregards the heterogeneity of combination products in development along with the diversity of clinical or patient-reported outcomes measures. Here, we present a dissection of SHL’s Innovation Partnership program along with our connected device offering – both of which allows us to flexibly collaborate with any pharmaceutical partner in the development of connected therapeutics.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance










































-file145532.png)