CPHI North America 2024 – From the Floor
.png)
Welcome to Philly! CPHI North America once again graces the Philadelphia Convention Center, 7–9 May 2024.
As you would expect, we have a thrilling conference track at this year's event, as well as a few extra treats thrown in.
As an introduction to the show, we've taken an in-depth look at the four main tracks that will be featured in the conference theatres.
Track one: Sustainable Futures – features talks from Jim Massey, Zai Laboratories, Bridget Ferrari from Takeda, Rachel Harris of Astra Zeneca, and many more. This track is sponsored by Thermo Fisher Scientific.
Track two: Revolutionising Drug Manufacturing – where we hear from Jean-Marie Geoffroy and Pete Smith from Pfizer, Subramanya Nayak from AbbVie, and Hang Tran of ATS Life Sciences Group, among others.
Track three: Supply Chain Economy – see more influential speakers including Bikash Chatterjee, Pharmatech Associates, Joanne Chan of Pharmaceutical Research and Manufacturers of America (PhRMA), and Laura Bray from Angels for Change. The track is sponsored by the US Pharmacopeia.
Track four: Exploring Excipients – this trending track will features presentations from experts such as Nelio Drumond from Takeda, Björn-Hendrik Peters from Bristol Meyers Squibb, and Denise Reaves from Jurata Thin Film.
See more details about what will be discussed across these tracks in this article:
The CPHI North America agenda - see what's in store (cphi-online.com)
Further to this, we've also been speaking with the Track sponsors, to hear from them directly about why the work they're focusing on is so important and what the tracks have to offer.
CPHI are keen to make the sustainability conversation more prominent at each event. CPHI NA is no exception, with its conference track dedicated to the topic – Sustainable Futures. Thermo Fisher Scientific have sponsored this track, and are presenting some of the key sessions. In an interview with Matt Hancock, PhD, Director Business Management, API at Thermo Fisher Scientific, they discuss the role of sustainability in the North American pharmaceutical market, and go into more detail on sustainable API development from a CDMO perspective.
See the full interview here: On Track at CPHI NA 2024: Sustainable Futures with Thermo Fisher Scientific (cphi-online.com)
In recent years all players in the pharmaceutical industry have been turning their focus onto the supply chain, and how to make sure it's more efficient and more future-proof.
The US Pharmacopeia have taken big steps to ensure this for companies across the USA, and in this interview with Matthew Christian, USP’s Director, Supply Chain Insights, USP Supply Chain Centre, he discusses supply chain resilience, and the impact this has on individual patients.
Read the full interview online here: On Track at CPHI NA 2024: Supply Chain Economy with US Pharmacopeia (cphi-online.com)
We've also been able to hear from a few experts who will be presenting sessions at the show.
In a recent interview with Laura Bray, Chief Change Maker at Angels for Change, Bray unfolds the story of how Angels for Change came into being in 2020, and how since then, the organisation has helped to prevent shortages of hundred of thousands of medications in the USA. The evocative interview shows how the organisation manages to connect the affected patients to the drug manufacturers and distributors so directly, so that those patients that are impacted by drug shortages are not lost in menial figures. Bray will be hosting a presentation at CPHI NA as a part of the Supply Chain Economy track, absolutely not one to miss!
Angels for Change: Guardians of the pharmaceutical supply chain (cphi-online.com)
Start-Up Market
 For the first time at CPHI NA we are hosting a Start-Up Market. After the success of showcasing Start-Up companies at CPHI Barcelona and Pharmapack, the initiative has finally made it across the pond to North America where we are happy to exhibit some US-based companies, as well as more from Europe.
For the first time at CPHI NA we are hosting a Start-Up Market. After the success of showcasing Start-Up companies at CPHI Barcelona and Pharmapack, the initiative has finally made it across the pond to North America where we are happy to exhibit some US-based companies, as well as more from Europe.
The Start-Up companies attending the event this year have been listed in the following infographics, where you can hear from some of the CEOs from the Start-Ups themselves on why they are so excited to come to CPHI NA.
First look at the Start-Up companies featuring at CPHI North America 2024 (cphi-online.com)
Next up: five more Start-Up companies exhibiting at CPHI North America 2024 (cphi-online.com)
The final six: meet the last Start-Ups exhibiting at CPHI NA 2024 (cphi-online.com)
Diversi'tea'
Devoted to highlighting diversity and the voices of women in pharma across our events and the industry as a whole, we will be holding a diversity session on the third day of the event.
Several speakers, distinguised voices from across the industry, will be holding conversations through a range of topics. The opening speech will be focusing on the role of diversity in the shaping the future of pharma, how it's critical to embed diversity in the industry.
There will be a talk specifically on the role of women in pharma, following an ongoing theme CPHI upholds, and delving into the diverse experiences, challenges, and opportunities facing women in our industry.
There also has been much research surrounding the importance of acknowledging and understanding the impact of diverse populations in patients, to discuss this further, we have a session on diversity when it comes to patient-centric solutions.
In an interview with Alise Cortez, Chief Ignition Officer at Gusto, Now!, Dr Marielle Weintraub, Director of Scientific Strategy at Eurofins FCT, and Carletta Calahan, Patient Advocate, they delve into their daily efforts to ignite change within their workplaces and beyond.
See the full interview here: Women in Pharma: Diversi‘tea’ at CPHI North America (cphi-online.com)
Day 0 - 6th May
This day the team were all on site, exploring the show floor, this is always a fun day to see the whole team together and the exhibition come to life after months of work in the lead up.
I also started my food tour of Philly, and am very pleased about the proximity of Reading Terminal Market to the exhibition centre, with the famous Philly Cheese Steaks planned into the agenda for Thursday afternoon.
.png)
.png) In the evening, we attended a very interesting discussion and networking event hosted by EisnerAmper (with a fantastic view to boot). The group discussed the unique opportunities that Philadelphia provides for the pharmaceutical and healthcare industry, with the history of the region steeped in pharma genesis, speakers noting that life sciences are in the DNA of the community.
In the evening, we attended a very interesting discussion and networking event hosted by EisnerAmper (with a fantastic view to boot). The group discussed the unique opportunities that Philadelphia provides for the pharmaceutical and healthcare industry, with the history of the region steeped in pharma genesis, speakers noting that life sciences are in the DNA of the community.
The deep life sciences vein running through Philly is something that the new Mayor, Mayor Parker is keen to continue and to grow, providing further opportunities and attarcting more talent to the area. A representative of Mayor Parker stated: "Philadelphia is where history meets innovation".
Several experts discussed the investment landscape in the region, noting that the talent pool in Philadelphia was one of the best in North America, attracting a lot of investment from Angel Investors and Venure Capitals, which, in today's investor market, can give an edge to a company looking for funding to progress their products.
Day 1 – 7th May
Breakfast session with Adare Pharma Solutions.
Srini Shanmugam talks through the nuances of special populations, and why they need to be considered when developing certain medications. He explains the difference between acceptance and adherence, and how to acheive them persistently in these patient populations. 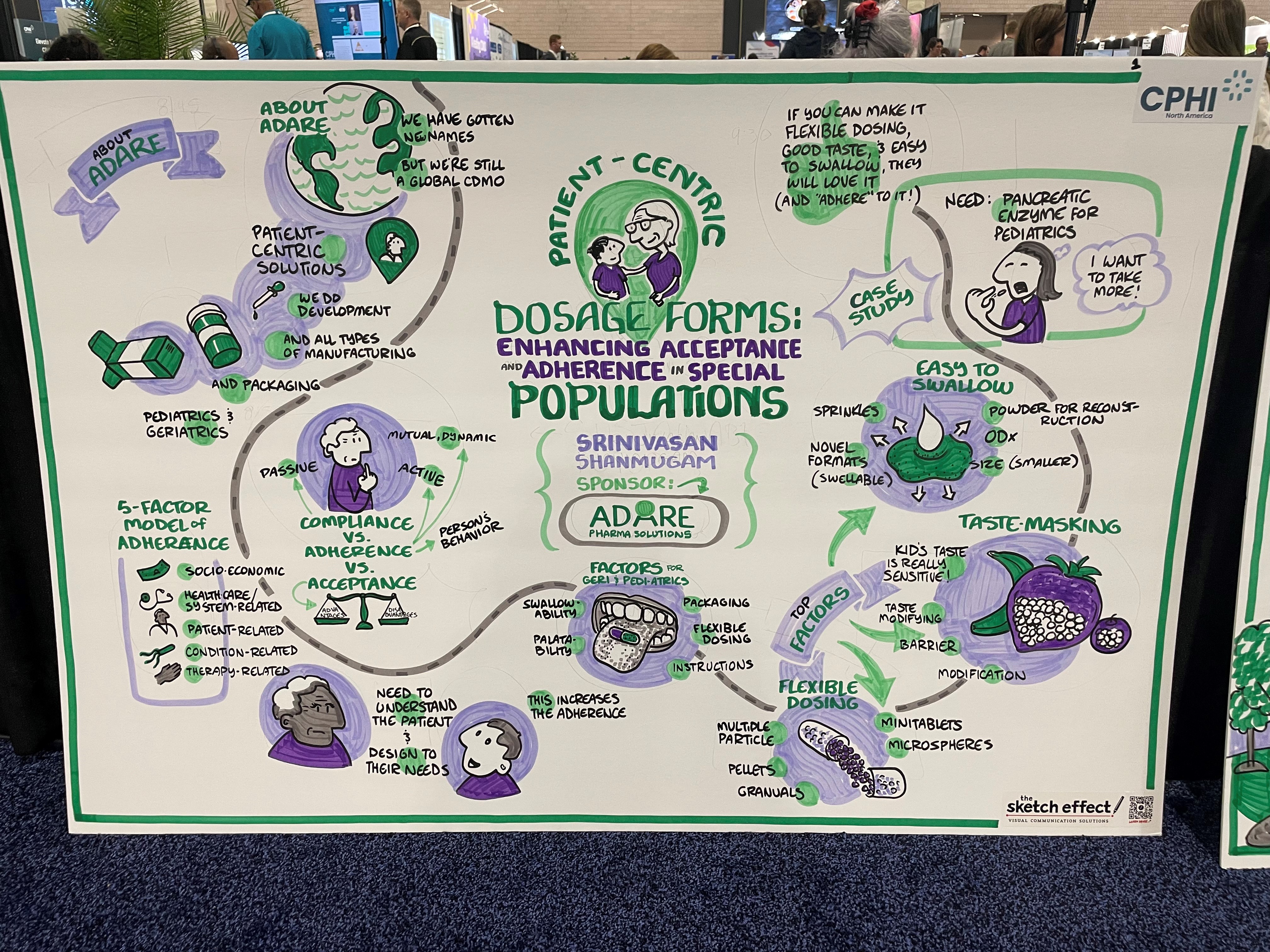
Shanmugan emphasizes the importance of identifying the specific need of the patient population, and then developing a product that addresses that need. Age is a very big factor when considering developing medications, paediactric populations have trouble with things such as swallowing medications, which needs to be taken into account. In geriatric populations, they often need very flexible dosing options. These are just some examples of the many needs of these populations, and if these are addressed, drug makers can go a long way to increasing acceptance and adherence in these special populations.
Track one in Conference Theatre one today focuses on sustainability. Matt Hancock from Thermo Fisher Scientific discusses green chemistry in his presentation 'Green Chemistry in CDMO world. Thermo Fisher Scientific's approach to sustainable API development and manufacturing'.
.png) Hancock discusses the use of solvents, which need to be classified, whether they are reccomended, or harzardous. But what we also need to know is are they single use? Can these solvents be effectively recycled? There are many aspects that come into this, regulation, the synthesis of the solvents, a classic option – can we reduce the number of solvents used? Broadly, a recycling strategy needs to be developed, so that the use of these materials can continue in a safe and more environmentally friendly way.
Hancock discusses the use of solvents, which need to be classified, whether they are reccomended, or harzardous. But what we also need to know is are they single use? Can these solvents be effectively recycled? There are many aspects that come into this, regulation, the synthesis of the solvents, a classic option – can we reduce the number of solvents used? Broadly, a recycling strategy needs to be developed, so that the use of these materials can continue in a safe and more environmentally friendly way.
The sustainability track covered more insights on greening the supply chain from Jason Wykoff, Schneider Electric, Bridget Ferrari, Takeda, and Devin Carsdale, Bristol Myers Squibb. The speakers discussed the path to decarbonising the supply chain in pharma industry in the USA.
They highlighted the advantages of models such as provided by the PSCI Maturity Models, and of programmes like the Energize programme from Schneider Electric.
The CDMO Roundtable – Outsourcing Trends
Chaired by Jim Miller, the panel was made up of Anil Kane, Thermo Fisher Scientific, Dave Powell from Grand River Aseptic Manufacturing, and Tim Quinn from Actylis.
The panel discussed the CDMO landscape, getting into the challenges surrounding regulations, and the potential of new technologies.
Kane discussed some products that are taking the market by storm at the minute, highlighting semaglutides and peptides, which can be delivered for a variety of indications including diabetes, Crohn's disease, and cancers to name a few. The panel discusses how oligonucleotides are being explored in the clinic for a variety of applications and are showing a lot of promise, and in turn attracting attention for investment.
Quinn added that certain skill sets are required when working on these products, attracting talent in niche areas, which adds value and broadens horizons for companies looking to explore these new areas. He also said this could lead to a lot more contract manufacturing for CDMOs in the USA.
Miller queried whether there was much interest in other forms of delivery or dosage forms, was this something companies are looking into at the minute?
Kane answers that there is always space for oral and injectable dosage forms, other forms have their benefits and certain molecules suit different methods, but oral and injectables are going to stay as the key elements.
Some things can be expanded, depending on assembling capabilities, autoinjectors are a prime example, companies can always buy more complex devices but it’s a big investment. It will take big pharma innovators to lead the way and make that investment.
Miller turns the panel to a conversation around the future, naturally concentrating on the role of artificial intelligence in drug development.
Kane: "It’s definitely a buzzword - the application of it is new, (even though AI itself isn’t new) we are applying it in new ways, predictive modelling tools to look at drug candidates, we use them a lot for scale-ups and in manufacturing we apply machine learning and use of data for predicting what should go right, what could go wrong?"
AI in the supply chain has almost unlimited potential for streamlining processes and improving manufacturing efficiency. By using inspective AI, automated inspection, leads to reduced time on manufacturing and reduces costs long term after the initial investment.
Quinn adds that it can be used in chemical route analysis, saving time in the synthesis pathway.
The panel concedes that AI has some controversial aspects, but that ultimately human intelligence leads the way, and AI is best used to assist.
The Role of Diversity in Shaping the Future – an inspiring panel discussing the challenges and advantages of a diverse work population in the pharma industry.
The Keynote from Demetria Johnson walked through the results of surveys of the industry, how people are seeing diversity, the importance of different cultures and different people. She highlights that many people think that the glass ceiling hasn't been broken to the level that we like to think, and this detrimentally affects people progressing, for example, Black and Hispanic women are more likely to feel imposter syndrome.
Johnson encourages companies to value early talent, and to prevent exclusion. Since COVID-19 there has been a mental shift, she states, and we know that what we need now is more talent, more partners who nuture this talent. A good mantra for partners is 'communication, consistency, and clarity', refering to continuously having open conversations and creating a safe space for all people in the work environment. Socioeconomic diversity, neurodivergency, and gender-related diversity all need to included in this conversation.
Johnson left us with a quote from Maya Angelou:
“I've learned that people will forget what you said, people will forget what you did, but people will never forget how you made them feel.”
The Diversity conversation moved on to a panel discussing the power of women in the pharmaceutical industry, moderated by Alise Cortez. The panel advocated speaking up and expressing how to treat people in the workplace, and teach others how you want to be treated.
.png) Dr Marielle Weintraub commented: "The first answer is not always final - finding a way to work how you want to work within seeming boundaries, in a subtle way asking for what you want," – having that shift the opinion of those around you to be able to get what you want, this is how we see progression born of having a range of opinions and people and backgrounds at the table.
Dr Marielle Weintraub commented: "The first answer is not always final - finding a way to work how you want to work within seeming boundaries, in a subtle way asking for what you want," – having that shift the opinion of those around you to be able to get what you want, this is how we see progression born of having a range of opinions and people and backgrounds at the table.
Cortez pointed out that women have exceptional emotional and relationship intelligence, which can be a real advantage in the workplace.
As the panel continued they spoke to the benfits of allyship in the workplace and those kinds of relationships, of championing other people.
"Every single one of us wants to matter, everyone wants to know that their time matters and that their work matters."
Tiffany says she loves to know that they're working towards, the pharma industry is very altruistic, which suits women in the industry, this has changed from a few years ago and is defintely a shift for the better.
Day 2 – 8th May
This morning we started off with a fantastically popular breakfast session with Veranova, 'Antibody-Drug Conjugates (ADCs): Navigating Four Pillars of Safety, Development, Supply Chain and Manufacturing Excellence' with speaker Dr Kishore Hotha, Veranova, Global Vice President, AR&D Operations. .png)
Laura Bray kicked off the presentations in the Supply Chain Economy track, bringing home the importance of keeping the patient at the centre of drug manufacturing and supply, to ensure that no individual goes without the life-saving medicine they need. See a more in-depth interview with Bray here: Angels for Change: Guardians of the pharmaceutical supply chain (cphi-online.com)
Joanne Chan, PhRMA, and John Murphy, Chief Policy Officer at BIO took to the stage to discuss drug pricing dynamics in the supply chain economy in the US.
They started off by going over some of the effects we've seen from the introduction of the Inflation Reduction Act in 2022. Seeing how pricing negotiations will affect the drug manufacturing and supply side, and the companies navigating this new challenge.
Bikash Chatterjee of Pharmatech Associates chaired the Supply Chain Economy panel Reshoring US Pharma: The Realities, which included Kevin Webb of the API Innovation Center, Rob York of Pacific Forum, Andrew Gonce of Mallinckrodt Specialty Generics, and Christina Smolke of Antheia.
 The conversation turned towards at-risk drug products given the current state of drug shortages, with Webb stating: "We realise as a nation that many of our drugs are at risk. Drug shortages are a matter of economics – fix the economics and you fix the drug shortage problem."
The conversation turned towards at-risk drug products given the current state of drug shortages, with Webb stating: "We realise as a nation that many of our drugs are at risk. Drug shortages are a matter of economics – fix the economics and you fix the drug shortage problem."
When Chatterjee asked about how to incentivise manufacturers in helping to find a solution, answers ranged from establishing a public/private model to invest long-term to investing in advanced technology with a 5–10 year commitment. "The manufacturer wants to make sure the customer exists on the back-end, the panel commented.
"Medicare and the government need to be there to buy these drugs when they come through the process and in the middle, we have to address some of the policy issues that have gotten us into this position in the first place."
York identified India as a friendly shore for the supply of generics, giving an example of shared perceptions on regional dynamics.
Smokle added: "Someone needs to lead and develop a strategic approach. Our government is good at responding to emergencies, but it’s not so good at developing a 10-year plan and following through on that. The government can set the vision and incentivise it, and the investment and private players will follow. There is a business case to be made there."
The panel also agreed that public and private partnerships are important to progress continuous manufacturing capabilities. Generics have idle manufacturing capacity as manufacturers have moved efforts off-shore. Repurposing these sites with advanced manufacturing technology could add 30 billion doses of products in the USA.
Consistency of standards were also an area of concern for the panel. While the Quality Maturity model has brought an understanding of the level of quality clients want and need, Chatterjee also suggested rewarding hospitals and manufacturers as a viable option for incentivisation. "For generic manufacturers, there's no new way to do it – companies need to redefine how we market their product," he said. "Elevate the companies that are doing well and complying to GMP, and reward that behaviour."
The Excipients Panel - Making Excipients Great Again
Chaired by Dr Bjorn-Hendrik Peters, the panel brought together David Schoneker of Black Diamond Regulatory Consulting, LLC, Nelio Drumond of Takeda, and Evonne Brennan of IMCD.
The panel began with the question of how to make excipients greater in this day and age. Answers ranged from getting them out of the commodity space. Schoneker began: "You don’t treat apis like a commodity, but your drug is made up of 85% of excipient, yet it’s treated like a commodity. We need to make the drug product itself greater – put more effort into it and take a little more risk. If we don’t change that paradigm, we’re only limiting ourselves."
.jpg) Peters disagreed that excipients are treated like commodities, commenting that it is more of a generalisation. Both Peters and Schoneker agreed that many companies treated excipients differently. Schoneker commented "Even an established excipient or a compound on the list but it’s not classified as safe to use, need more guidance. Clarification about how an agency thinks about novel excipients and the kind of data they expect to see out of excipient programmes would be of benefit. That is what we're trying with IPEC at the minute."
Peters disagreed that excipients are treated like commodities, commenting that it is more of a generalisation. Both Peters and Schoneker agreed that many companies treated excipients differently. Schoneker commented "Even an established excipient or a compound on the list but it’s not classified as safe to use, need more guidance. Clarification about how an agency thinks about novel excipients and the kind of data they expect to see out of excipient programmes would be of benefit. That is what we're trying with IPEC at the minute."
Drummond added that "Another consideration is looking at current excipients and seeing how can we solve some of the challenges there. Nitrites, for example, have a lot to do with excipients. We need to look at excipients having the right quality where they are not causing issues or trespassing limits that bring toxicity. You need to avoid impurities that bring toxicity to patients, and then you have to go back to the bench." Evonne also commented that "With excipients, understanding them, their challenges, and the regulatory pathways will help companies."
Drummond concluded with "Proactive communication is needed but these channels are difficult to navigate. The scientists don’t talk to the supplier, and those discussions are what you need. Otherwise it hurts the process."
Schoneker added: "More competition makes it seem like a commodity. Bringing it all together, excipients must be fit-for-purporse. We also need more communication, education, and understanding the excipients better.
The excipient supply chain is just the same as the mainstream supply chain – you need to make long-term commitments."
All of the above content, and more from the show is available online On Demand. You can catch up on any of the sessions you missed here.
That's all from our team! Make sure to tune in next year for CPHI North America 2025!
.png)
Related News
-
News Women in Pharma: Diversi‘tea’ at CPHI North America
CPHI North America will unite the pharmaceutical supply chain in Philadelphia from May 7–9, 2024 for 3 days of innovation and connections. As part of the content Agenda, our Diversity Track will bring the industry together to discuss the imperati... -
News More to celebrate at the CPHI Milan Pharma Awards 2024
The CPHI Milan Pharma Awards, which are now open for nominations until 18 May 2024, celebrate the innovators making a difference. This year, two new categories have been added to further honour the contributions of a diverse range of pharma professiona... -
News On Track at CPHI NA 2024: Sustainable Futures with Thermo Fisher Scientific
On the 7-9 May 2024 in Philadelphia, CPHI NA will be presenting an unmissable agenda, filled with content across 4 main tracks. In the following interview the Sustainable Futures Track sponsor, Thermo Fisher Scientific gives an overview of what to expe... -
News LEAP-ing into the future of pharma – the CPHI Middle East Vision
On March 6, 2024, the CPHI team hosted the Future of Pharma Forum at LEAP 2024, Saudi Arabia’s premier destination for tech leaders and innovators to meet and collaborate. -
News CPHI Middle East 2024 – What do we have to look forward to?
We’re bringing CPHI to the Middle East in December 2024. CPHI brings together the pharma community from all over the world, creating a platform for education, collaboration, and progress. -
News CPHI Pharma Awards 2023 – Packaging & Machinery Winner: Gasporox
After another year of impressive nominations for the CPHI Pharma Awards our winners were announced at CPHI Barcelona in October 2023. In this series of interviews, we speak to the people and teams behind the award-winning projects, concepts, and techno... -
News Unpacking what we learnt: Key Pharmapack takeaways with L.E.K. Consulting
After another successful Pharmapack show, we sat down with Karin von Kienlin, Senior Partner at L.E.K. Consulting, to discuss the biggest takeaways and surprises from 2 days of intense discussion and collaboration. -
News Key takeaways from Saudi Arabia's National Biotechnology Strategy
In January 2024, Saudi Arabia released its National Biotech Strategy, an ambitious plan to localise biomanufacturing, incentivise R&D and establish the Kingdom as the regional biotech leader by 2030, and an internationally recognised hub by 2040.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance

.png)



.png)
.png)
