BD Completes Study Investigating Performance of Glass Prefillable Syringes (PFS) in Deep Cold Storage
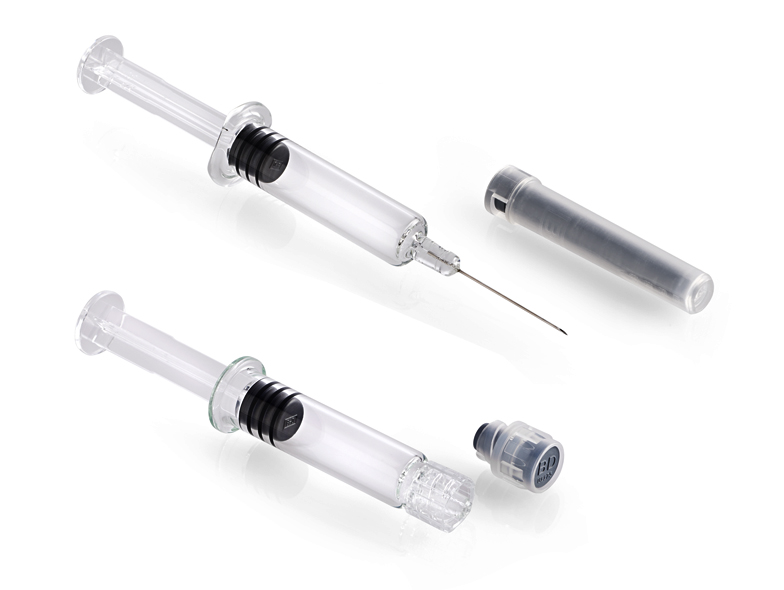
BD Hypak™ for Vaccines Glass Prefillable Syringes
Study reveals glass PFS can be stored at -20°C and -40°C without risk on PFS key product functions, establishing pathway to further assess the stability and performance of drug combination products with PFS in deep cold conditions.
Franklin Lakes, N.J., September 22, 2021 - BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, completed a preliminary study investigating the impact of deep cold storage (-20°C and -40°C) on glass PFS.
While traditional vaccine formulations are commonly stored up to about 2°C-8°C, new vaccine formulations – including those for mRNA – require much colder storage for over a long period of time to ensure drug-product shelf-life and potency [1][2]. Such low temperatures can induce changes in container dimensions, phase transition - liquid to solid and vice versa - and thermal shock. This could compromise the functional performance of the delivery system and Container Closure Integrity (CCI). BD began its investigation on the impact of deep cold storage on glass PFS as soon as information regarding the deep cold storage requirement for mRNA COVID-19 vaccines became available in 2020.
BD researchers conducted a comprehensive analysis of approximately 2,000 PFS after deep cold storage at -20°C and -40°C. Tests were conducted on several combinations of glass barrel coatings, formats - from 0.5 to 3 mL -, different tip and flange designs, and multiple elastomeric closures with various state-of-the-art methods, including Lighthouse Laser Headspace Analysis [3], at these temperatures.
The study showed that key product functions – PFS system level – were unaltered when stored at -20°C and -40°C. In addition, the count of subvisible particles and container closure integrity (CCI) remained unchanged when compared to those stored at room temperature.
“PFS are a known factor in addressing the complexity and costs of vaccine administration. They have been shown to significantly reduce time and labor associated with drug preparation compared to vial formats [4], and they support dose sparing [5]” said Bruno Baney, Vice President of R&D for Pharmaceutical Systems at BD. “This study confirms the promising opportunity we have to apply our leading scientific capabilities to develop innovative delivery solutions for our customers now and in the future. We’re encouraged by these results and look forward to partnering with pharmaceutical companies to further advance PFS in deep cold conditions.”
Following its completion of this study, BD anticipates that BD glass barrel PFS systems should be suitable for use when storage temperatures of -20°C and -40°C are required.
Comprehensive study results will be presented at the 2021 “Universe of Pre-Filled Syringes and Injection Devices” PDA conference on October 6th.
Detailed study results are available to BD customers/partners upon request.
BD is committed to a science-driven approach to innovating and testing delivery solutions for novel therapies developed by our pharmaceutical partners – particularly as the fight against COVID-19 continues. BD is also enhancing its manufacturing capacity and technology for PFS and advanced drug delivery systems with previously announced plans to invest $1.2 billion over four years.
About BD
BD is one of the largest global medical technology companies in the world and is advancing the world of health by improving medical discovery, diagnostics and the delivery of care. The company supports the heroes on the frontlines of health care by developing innovative technology, services and solutions that help advance both clinical therapy for patients and clinical process for health care providers. BD and its 70,000 employees have a passion and commitment to help enhance the safety and efficiency of clinicians' care delivery process, enable laboratory scientists to accurately detect disease and advance researchers' capabilities to develop the next generation of diagnostics and therapeutics. BD has a presence in virtually every country and partners with organizations around the world to address some of the most challenging global health issues. By working in close collaboration with customers, BD can help enhance outcomes, lower costs, increase efficiencies, improve safety and expand access to health care. For more information on BD, please visit bd.com or connect with us on LinkedIn at linkedin.com/company/bd1/ and Twitter @BDandCo.
For further information please contact:
Media
Trey Hollern
Director, Public Relations
+1 862.284.8629
[email protected]
Investors
Kristen M. Stewart, CFA
SVP, Strategy & Investor Relations
+1 201 847 5378
[email protected]
References
[1] Pardi, N. et al, mRNA vaccines — a new era in vaccinology, US National Library of Medicine, National Institutes of Health, 2018 Jan 12
[2] N. Rathore et al., Current Perspective on Stability of Protein Drug Products during Formulation, Fill, and Finish Operations Biotechnology Prog., pp. 504-514, 2008
[3] Lighthouse Headspace container closure integrity test methods are based on analytical measurements of gas ingress into a container through a leak, and the rapid nondestructive nature of the testing enables the generation of science-based statistical CCI data. Study results confirmed CCI remains unchanged as compared to when stored at room temperature (during the thermal cycle at -20°C and -40°C with no gas ingress).
[4] Pereira et al., 2010 , Expert Reviews.
[5] Makwana, 2011, Prefilled syringes: An innovation in parenteral packaging.

Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance