Nemera
About Nemera
Certifications
Categories

-
FR
-
2015On CPHI since
-
1Certificates
-
1000 - 4999Employees

Company types
Meet us at
CPHI Milan 2024
Fiera Milano, Italy
08 Oct 2024 - 10 Oct 2024
Pharmapack Europe 2025
Paris Expo, Porte de Versailles - Hall 7.2 | Paris, France
22 Jan 2025 - 23 Jan 2025
Products from Nemera (15)
-
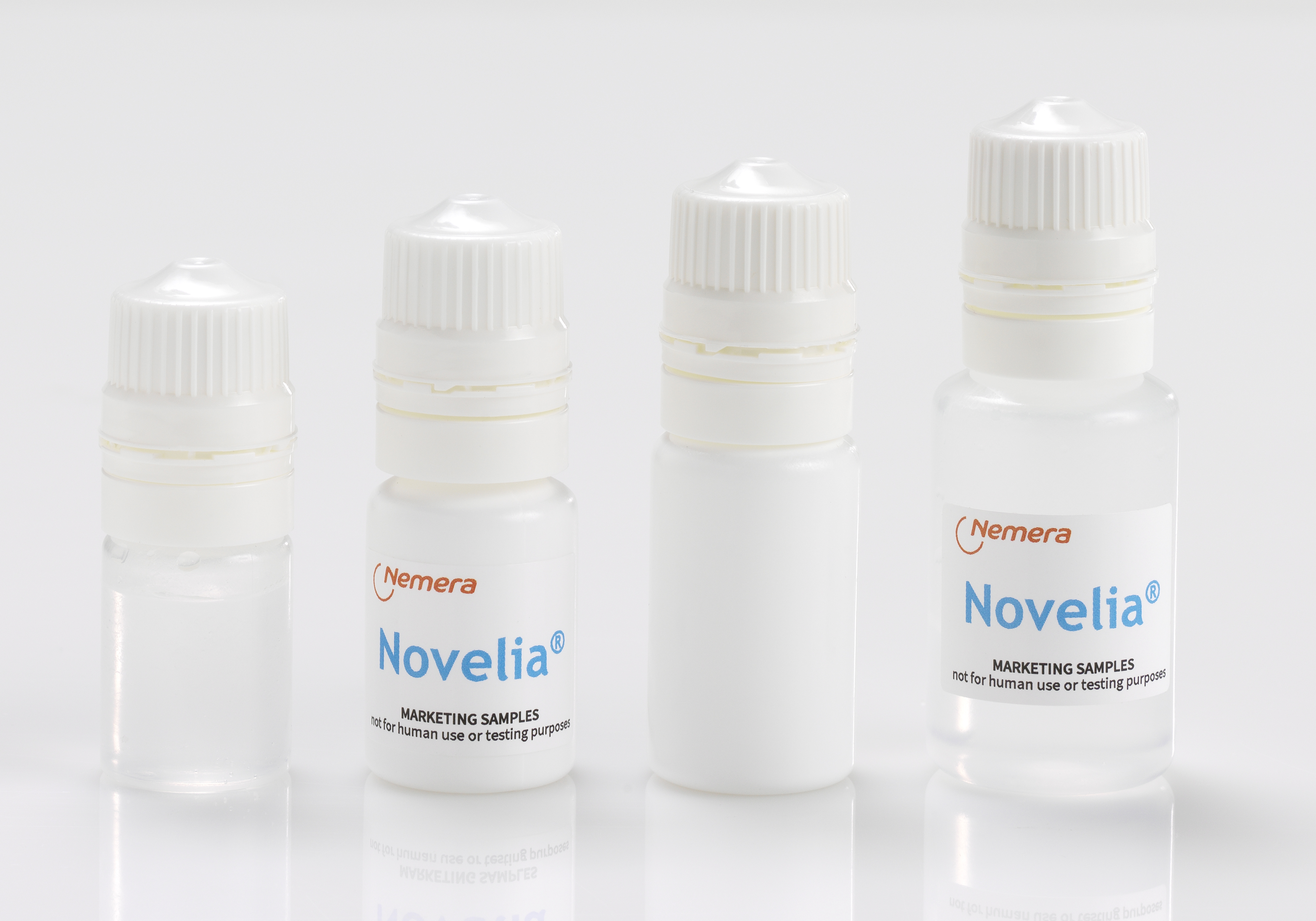
Product Novelia®: Nemera’s preservative-free multidose eyedropper
Novelia®: Nemera’s preservative-free multidose eyedropper, is used across 4 continents
Preservatives have been shown to be harmful to the eye and can lead patients to discontinue or skip treatment to avoid irritation and discomfort.
The new preservative-free multidose ophthalmic packaging Nove... -
Product PUMPS SP27 / SP270+: quality and performance for ENT sprays
Nemera offers an extensive range of metered pumps and valves, compatible with a wide choice of actuators in different shapes and fitments for nasal, buccal and auricular delivery.
Here our different pumps:
SP27 - SP 37: Quality systems for unregulated markets
SP270+ - SP370+: Qual... -
Product Unispray
Unispray is a ready-to-use primeless nasal device with accurate single-metered liquid dose delivery. It offers one-handed activation with 360° functionality to face emergency and crisis.
Thanks to its ergonomic design, it is intuitive and easy-to-use.
-
Product Safe'n'Spray
Safe’n’Spray, the new smart nasal spray with locking features, dramatically improves the existing way of administrating specific pain treatments, avoiding the risk of overdosing.
-
Product DPIs / PMDIs : expert in development & manufacturing of respiratory drug delivery devices
Nemera has a recognized expertise in the development and manufacturing of respiratory drug delivery devices, such as Dry Powder Inhalers (DPIs), pressurized Metered Dose Inhalers (pMDIs) and dose counters.
More than ten million asthmatics rely everyday on devices manufactured by Nemera.
-
Product DPI / PMDI
From early-stage concept development to manufacturing we are truly a holistic partner in inhalation.
With a long-standing know-how in inhalation delivery, we are today the leader in the Dry Powder Inhalers (DPIs) manufacturing business.
Our expertise covers Dry Powder Inhalers (reservoir, b... -
Product Safe'n'Sound®
Safe’n’Sound® (1ml & 2.25ml) is a highly customizable platform of add-on passive safety devices for prefilled syringes. It helps in preventing needlestick injuries, providing user-friendly protection for healthcare professionals and patients or caregivers.
Ergonomically designed, Safe’n’Sound® is ... -
Product Symbioze
Symbioze is specifically tailored to administer complex drugs, such as biologic therapies, thanks to a highly engineered, reliable drug delivery system. It is suitable for various drug platforms and compatible with market-proven cartridges, as well as standard manufacturing processes. The cartridge is pr... -
Product Pen Vario
Our portfolio of disposable pens includes variable and fixed dose devices for automatic and manual drug administration, combining modern design with high intuitiveness of use for multidose therapies.
PENVARIO – Dedicated to manufacturing of disposable, variable-dose pen injectors
-
Product Reusable pens
Our reusable pens ensure the application of proven technological solutions with reliability, intuitiveness as well as ease-of-use.
-
Product Autoinjectors & pens
Proven track record in managing parenteral customized solution and large scale industrialization projects of Autoinjectors and Pens with top pharmaceutical companies.
Our offer:
- disposable & reusable pen injectors
- Taylor-made autoinjectors & pens
-
Product Implanters
Implants are fragile and insertion into the body requires caution. Nemera developed a portfolio of devices to deliver implants with integrated needlestick protection.
There is two types of implanters:
- Safety depot syringe with telescopic plunger rod
- Safety retro injector for ...
Nemera Resources (11)
-
News Nemera: Stronger than ever
More modern, our new brand identity truly embodies who we are, how we partner with our customers and ultimately the value we bring to millions of patients across the world through our drug delivery device solutions. -
Video Innovation in Connectivity Roundtable pt.1
The Innovation in Connectivity Roundtable originally aired as part of Pharmapack Europe 2021 Part 1 of this session includes three presentations: Patient Journey Driven Considerations for Integrating Digital Health for Combination Products The importance of utilizing applied ethnography and other primary research methods to understand the patient journey as the foundation for integrating digital health into a device/combination product development initiative. Considering patient needs in the context of a larger ecosystem including providers, payers, and regulators to develop a robust plan with respect to connectivity and digital health. Examples of how this understanding is applied across the combination product development journey through organic development, utilization of platform devices, or as a lifecycle management enchantments while optimizing the user experience resulting in safe, effective, and differentiated combination products. Mark Tunkel, Global Category Director for Insight Innovation, Nemera Electronic Product Information: A Case Study on Medicine Labelling for the Visually Impaired The Electronic Product Information Initiative for Medicines (ePI) aim to ease the accessibility of product information to everyone, including visually impaired patients. The GS1 Digital Link aim to provide a single barcode for identification, brand protection, and access to product information for patients. Our lecture will illustrate how the existing Datamatrix on packaging could be leveraged, today, by everyone, including visually impaired users, to access a tailored and relevant information. Laurent Tonnelier, CEO, mobiLead - xTag Lise Wagner, Accessibility Specialist and User Expert, OKEENEA Traceability of Medicines: from Finished Product to Unit Dose Delivery Challenges for B2B Supply Chain stakeholders Regulations: a global overview What about B2C Nathalie Wardé, CEO & Founder, D4P pharma Join the above speakers and moderator Lionel Jeannin, Group Head Oral Packaging & Delivery Systems, Novartis Pharma AG for the live panel discussion that followed in the Innovation in Connectivity Roundtable pt. 2 -
News Pharmapack Europe 2021 announces Awards winners!
Awards showcased a wide range of industry-changing innovations in packaging and drug delivery, with sustainability emerging as a key metric across all categories -
Video Innovation in Connectivity Roundtable pt. 2
The second part of the Innovation in Connectivity Roundtable brings together presenters from Nemera, D4P Pharma and xTag for a group discussion and live Q&A (45-minutes). Moderated by Lionel Jeannin, Group Head Oral Packaging & Delivery Systems, Novartis Pharma AG Participants: Mark Tunkel, Global Category Director for Insight Innovation, Nemera Laurent Tonnelier, CEO, mobiLead – xTag Lise Wagner, Accessibility Specialist and User Expert, OKEENEA Nathalie Wardé, CEO & Founder, D4P pharma This webinar originally aired as part of Pharmapack 2021 -
News Pharmapack 2021: Increasing connectivity is the key to patient centricity and simplified access to product information
During the live Innovation in Connectivity Roundtable at the Pharmapack 2021 online conference, experts discussed how the latest innovations in digital technology are shaping industry ambitions to put the patient journey at the heart of its drug device and delivery solutions by simplifying product information via increased connectivity. -
Video Welcome to the Digital Health & Connectivity & Patient-Centricity Track
This webinar will address: Key market trends in digital health and how they have accelerated in the past years Nemera’s vision of digital health and the implications on the whole drug/device development ecosystem – how connectivity is creating incredible opportunities for the industry but also raises some challenges: link to conferences in the track How does Nemera play in this landscape: presentation of the company, our capabilities and some examples of our connected devices. Webinar originally aired as part of Pharmapack Europe 2021 -
News Considerations for device selection in parenteral applications.
While patient centricity is the foundation for developing a robust device selection plan and completion of a successful combination product development initiative, developers need to remember there is a larger ecosystem that must be considered to ensure success.
-
Video Nemera: We Put Patients First
As a world-leading drug delivery device solutions provider, our purpose of putting patients first enables us to design and manufacture devices that maximize treatment efficacy. We are a holistic partner and help our customers succeed in the sprint to market of their combination products. From early device strategy to state-of-the-art manufacturing, we’re committed to the highest quality standards. Agile and open-minded, we work with our customers as colleagues. Together, we go the extra mile to fulfill our mission.
For more information, visit www.nemera.net -
News Nemera is excited to announce that its multidose eye dropper Novelia® has been approved in Brazil!
Nemera is excited to announce that its multidose eye dropper Novelia® has been approved by ANVISA for the preservative-free formulations CLILON [1] (Ketorolac 0,4% and 0,5%), a nonsteroidal anti-inflammatory drug (NSAID) that is used to treat moderately severe pain and inflammation, usually after surgery. -
Video NEMERA CEO SHARES 2021 AMBITION
Our CEO, Marc Hämel shares the lessons we learned from last year, extends his wishes, gratitude to the Nemera family and walks us through the top company goals that will shape and drive our story in 2021!
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance






















-file122583.png)

-file122681.png)

-file122584.png)




