Quotient Sciences
About Quotient Sciences
Certifications
Categories

-
US
-
2019On CPHI since
-
4Certificates
-
1000 - 4999Employees
Company types
Meet us at
CPHI North America 2024
Pennsylvania Convention Center, Philadelphia
07 May 2024 - 09 May 2024
Products from Quotient Sciences (5)
-
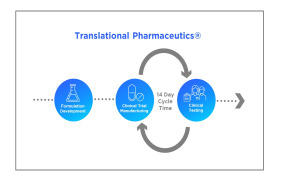
Product Translational Pharmaceutics
Translational Pharmaceutics®, accelerates development by integrating formulation development, real-time manufacturing and clinical testing. This integrated approach is proven to reduce timelines by more than 12 months and lower R&D costs by more than $100 million. Over the past decade ... -
Product Formulation Development
From preclinical and first-in-human (FIH) dosage forms to optimization of your drug product for late-stage development and market following clinical evaluation, we work with you to develop the most appropriate formulation based on the physicochemical and biopharmaceutics properties of your drug molecule, t... -
Product Drug Substance Synthesis and Manufacturing
Quotient Sciences provides drug substance synthesis and manufacturing capabilities from candidate selection through to commercial production. With tactical deployment of best-in-class technologies, we minimize chemistry costs and move your drug substance supply off the critical path. With more than 4... -
Product Clinical Trial Manufacturing
Quotient offers a clinical trial manufacturing, testing and certification service designed to meet your individual requirements for oral and inhaled dosage forms. Our innovative method of building integrated GMP and GCP programs provides you with a streamlined, flexible approach to drug product supply that... -
Product Poor Solubility & Bioavailability Enhancement
With nearly 30 years of experience, Quotient has established a broad suite of technologies and formulation approaches to address these complex solubility challenges. Our approach allows us to dramatically speed up the optimization of your drug products to improve oral bioavailability. For more information ...
Quotient Sciences Resources (2)
-
News Quotient Sciences expands service portfolio with Arcinova acquisition
The acquisition will extend the 12-month drug development timeline savings already delivered by Quotient’s flagship platform Translational Pharmaceutics -
Video Quotient Sciences Overview
Quotient Sciences is a drug development and manufacturing accelerator supporting customers across the entire development pathway from candidate selection to commercial launch. We deliver uniquely integrated programs and a range of tailored services that dramatically shorten drug development times. We’re driven by an unswerving belief that ideas need to become solutions, and molecules need to become cures, fast. Because humanity needs solutions, fast.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance