All News
All news
-
News Thermo to invest $150 million to expand Pharma Services capabilities
Expansion projects will increase capacity to meet growing customer demand for biologics. -
News Acquisition of niche API portfolio ensures quality and continuous supply
A fully redundant supply of digoxin API from C2 Pharma will be available via two CMO manufacturing sources at Nobilus in Poland and Laurus Labs in India. -
News Sizeable growth potential for global drug delivery and packaging in 2019
Patient-centricity, eco-friendly and smart packaging trends marked as the biggest growth drivers and innovations in 2019. -
News FDA approves nasal drug delivery device for breakthrough treatment of depression
Aptar Pharma’s Bidose liquid system is designed for local or systemic delivery of drugs. -
News Robinson Brothers feature in The Parliamentary Review 2019
Robinson Brothers features alongside the Prime Minister, Rt Hon Theresa May MP, and other UK organisations who wish to use The Parliamentary Review as a platform to share knowledge, insight and experience with other companies. The Parliamentary Revi... -
News Specialist API CDMO acquired by GHO Capital
Future plan is to extend Sterling’s international presence to meet increasing demand from customers worldwide. -
News Bringing next generation single-use sensor technologies to the life science market
Pall and Broadley-James’ combined expertise proves complementary in addressing critical customer pain points in modern bioprocessing. -
News Simagchem Corp.Joins Society of Chemical Manufacturers and Affiliates
Simagchem Corp.announced today its membership to the Society of Chemical Manufacturers and Affiliates, the leading international trade association representing the batch, custom and specialty chemical industry. -
News Nordson EFD Introduces Dial-A-Dose and Posi-Dose Animal Health Dosing Syringes
EFD provides advanced primary packaging and controlled drug delivery systems for animal health products worldwide. -
News New Plant to Open in Blarney Ireland
ILC Dover, a global leader in flexible containment solutions for the pharmaceutical and biopharmaceutical industry is expanding its Irish production footprint by opening a location in Blarney, County Cork, Ireland. -
News Sharp enhanced clinical IRT solution with latest investment
Sharp has announced a $650,000 investment into enhancing its Interactive Response Technology (IRT) solution. Sharp’s IRT solution is used to manage patient interactions and drug supplies during clinical trials and perform a range of functions for spons... -
News Sharp completes relocation to $23 million clinical services facility in Bethlehem, PA
Sharp – a part of UDG Healthcare plc, a global leader in contract packaging and clinical supply services, has completed its relocation to its $23m state-of-the-art facility in Bethlehem, PA. -
News Spray+Lock Presentation
Spray+Lock is our NEW Child-Resitant Spray Packaging which consists of bottles and closures. -
News GENTEC OPENS NEW OFFICE IN MIAMI (USA)
Gentec Pharmaceutical Group is pleased to announce the opening of its new office in Miami, Florida, in January this year. -
News Excite Pharma Services announces Retiree Program
We are excited to announce that Excite Pharma Services continues to change the industry! -
News BIOINTAXIS - A NEW COMPANY DEDICATED TO THE INVESTIGATION OF RARE DISEASES
Originally a foundation at the Institut d’Investigació en Ciències de la salut Germans Trias i Pujol (IGTP), Biointaxis has been created for the investigation of rare diseases and especially of Friedrich's Ataxia. -
News Third Sartorius Research Xchange Forum 2019 focuses on CAR-T cell therapy
Event for experts from academia and industry, including talks, live demo sessions, and panel discussion. -
News Injecting innovation into micro-injection molded solutions
End-to-end industrial production of innovative medical technology products. -
News Sichuan HuaFaMei Enterprise attends CPHI Japan and North American
Sichuan HuaFaMei Enterprise Co.,LTD. will attend CPHI JAPAN and CPHI NORTH AMERICA. Booth No. of CPHI Japan is 1B-11.
Booth No. of CPHI Japan is 912. Warmly Welcome to visit us. -
News Envigo launches PATHWAY to enable FIH clinical trials
PATHWAY manages the complexity of the entire safety assessment process on behalf of the company’s pharmaceutical and biotechnology customers. -
News Overcoming lyophilization challenges at Interphex 2019
Line of Sight approach to freeze-drying enables organizations to bring pharmaceutical products safely and quickly to market. -
News Asymchem is awarded as “2018 Best Partner of China Pharmaceutical Enterprises in Transformation and Upgrading”
The 2019 China Pharma New Year Forecast was successfully held in Beijing by the Healthcare Executive and China Healthcare Industry Investment Forum, supported by the China Pharmaceutical Enterprises Association on January 19. -
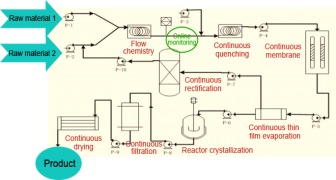
News Asymchem Takes New Step on Application of Continuous Reaction
Asymchem has become a global leader in the pharmaceutical CDMO industry. Asymchem has been committed to the development and commercial application of green pharmaceutical technologies for many years. -
News CPC Scientific Inc. and UNC Lineberger Comprehensive Cancer Center Announce Neoantigen Peptide Collaboration to Manufacture Neoantigen Peptides for the PANDA (Personalized and Adaptive) Cancer Vaccine Program.
CPC Scientific Inc., a leading global CDMO specializing in synthetic peptide manufacturing, announced today that they will manufacture GMP-grade neoantigen peptides for investigators at UNC Lineberger Comprehesive Cancer Center (LCCC) for th... -
News Sentinel Process Systems Named Exclusive North American Distributor for UNIQSIS
Uniqsis appoints Sentinel Process Systems Inc as exclusive US partner. -
News Purolite opens an Agarose Resin Manufacturing Facility with 100,000L-CAPACITY in Wales, UK
In October, Purolite and Purolite Life Sciences opens one of the largest manufacturing facilities in the world for the production of agarose-based chromatographic resins. -
News Push Tab® awarded at Pharmapack
Huhtamaki’s Push Tab® has been selected as the Best Innovation in the Solid Dosage Packaging category in 2019 Pharmapack Awards. The annual awards recognize new innovative products that are set to shape the future of the pharmaceutical industry. -
News Novel systems for membrane chromatography
Optimally run membrane chromatography processes will provide higher productivity, smaller-scale operations and increased robustness. -
News Lilly to introduce lower-priced insulin
Authorized generic version of Humalog will be available in US pharmacies at 50% lower list price. -
News Assist in Drug Development of Oligonucleotides
Utilizing our experience of synthesizing various nucleic acid related compounds over 60 years, Yamasa can collaborate in development of new monomers used in drug development of oligonucleotides. -
News SEQENS R&D services offer
Seqens offers complete and fully integrated R&D services to support our customers’ projects from initial stage to commercial manufacturing in total compliance with the highest quality, safety and environmental standards. -
News Biogen acquisition boosts its ophthalmology pipeline
Nightstar Therapeutics has two potentially first-in-class mid- to late-stage clinical assets as well as preclinical programs. -
News Copper-Mediated N-Arylations of Hydantoins
MINAKEM RECHERCHE has collaborated with the research group of Prof. Gwilherm Evano, from the ULB to develop a flexible approach towards N-arylated hydantoins. We are very pleased to announce that the publication has been approved in the Journal of... -
News Servier Launches InnoPreP™
Servier, an independent international pharmaceutical company, today announced the launch of InnoPreP™ – a new preparative chromatography service offered under its contract development and manufacturing business. -
News Uniqsis expands LED photoreactor range
The availability of three new alternative switchable LED configurations provides chemists with the unique ability to optimally undertake almost any photochemistry reaction. -
News Systech Releases Expanded Cloud Platform to Secure Global Supply Chains
Systech, a global leader in supply chain security and brand protection, today announced the latest release of its cloud-based traceability and authentication solutions, which ensure that products are authentic, safe and connected throughout the supply ... -
News Crowdfund and discover promising therapeutics via Molecule
Connecting scientists, patients, and industry to advance drug development in a collaborative open market. -
News Japan solid dose market predicted to bloom in 2019
Executives predict Japan to be the second fastest growing mature market for solid dose drugs in 2019, and the second fastest overall country for biologicals growth. -
News Recipharm achieves new ISO 45001 certification to support sustainability efforts
ISO 45001 developed to improve employee safety, reduce workplace risk and create better working conditions globally. -
News 'Tamper-evident' benefits are evident
2019 will mark Uniplast's debut at this year's edition of CPHI Worlwide in Frankfurt, where it will showcase its expertise in plastic packaging. -
News SGS Appoints Biologics Manager at Facility in Illinois
SGS, a leading bio/pharmaceutical analytical and bioanalytical contract solutions provider, today announced the appointment of Dr. Haris Jamil as Biologics Manager of its recently expanded biopharmaceutical testing facility in Lincolnshire, Illinois, U... -
News Pharma in South East Asia booming as CPHI opens event for the first time in Thailand
Healthcare and pharmaceutical reforms, designed to increase access to high-value medicines, driving growth. -
News CPC and UNC Lineberger Comprehensive Cancer Center announce Neoantigen Peptide collaboration
CPC, a leading global CRO/CDMO specializing in synthetic peptide manufacturing, have announced that they will manufacture GMP-grade neoantigen peptides for investigators at UNC Lineberger Comprehesive Cancer Center (LCCC) for the purpose of individuali... -
News Preservative-free multidose dispenser approved across Europe for glaucoma treatment
Aptar Pharma working closely with Santen Pharmaceutical to improve patient safety, achieve dosing accuracy and maintain product integrity. -
News Lonza Strengthens pharma, biotech and nutrition offerings
With the new structure, Lonza is positioned to leverage its innovation programs and technology platforms to create new solutions at the food-pharma convergence and in microbial control. -
News Spun bonded replacement for resin-bonded filters
A superior alternative filter solution for manufacturing processes using resin-bonded filter cartridges. -
News PMI BioPharma Solutions launches new facilities
Newly formed PMI BioPharma Solutions announces launch of research lab and manufacturing facilities tailored to the Pharmaceutical and Biopharmaceutical industry. -
News PMI BioPharma Solutions appoints CEO
PMI BioPharma Solutions announces the appointment of David Sunseri, PhD as CEO. -
News Exclusive technology and marketing collaboration for sublingual delivery
WuXi STA will have exclusive access to BioLingus's novel platform to stabilize and deliver sublingually drug targets including small molecules, peptides and proteins, that are currently administered to patients via injection. -
News Technical Presentation
Experts in deoxofluorination and Halex chemistry, Navin are able to run this specialized chemistry in bulk, using both SF4 and XtalFluor for deoxofluorination. -
News Skyepharma is developing its pharmaceutical packaging activities
SKYEPHARMA, a subsidiary of the VECTURA group, has chosen its St Quentin Fallavier site, in Lyon area, to develop its pharmaceutical packaging activities, with strategic investments in two new complete lines for bottle and blister packaging, includ... -
News Suspensions and their filling: Challenges in handling liquids filled with solids
Suspensions are dispersions of solid particles in a liquid. The solids correspond to the dispersed phase and the liquids to the dispersing agent. The production of suspensions enables the conversion of sparingly soluble active substances into a liq... -
News The market relies on individual, small batches in the production of cosmetics
Flexible dosing systems enable quick product changes in cosmetic filling Cosmetic production is undergoing a change. It is becoming increasingly important to fill small batches of various products in a short space of time. Previously, it... -
News Hemarina and Delpharm team up to manufacture HEMO2life®
Hemarina and Delpharm today announced the signature of a partnership involving the manufacture of HEMO2life®, an additive for organ transplantation solutions. -
News Another HUGE acheivement for Mastermelt Group!
Eco-Mastermelt (Singapore) are thrilled to announce our new ISO accreditations. -
News Murli Krishna Pharma Pvt at CPHI North America
We are pleased announce we are participating in CPHI North America 2019. -
News Therabel group strengthens partnership with Skyepharma Production
Skyepharma Production SAS, (“Skyepharma”) the contract development and manufacturing organization (CDMO) specialists in the development, manufacturing and packaging of classic and complex oral solid dosage forms, and Therabel, a privately-owned... -
News NEW PLANT INVESTMENT
2,800 square meters expansion of a plant in Sambuca Val di Pesa (Florence) and construction of a new automated warehouse. -
News Partnership expands to develop a new treatment for pachyonychia congenita
MedPharm and Palvella Therapeutics hope to develop the first approved treatment for PC in the US and Europe. -
News Contract Pharma Conference
The 18th Annual Contracting & Outsourcing Conference & Tabletop Exhibition will be held September 26-27, 2019 at Hyatt Regency New Brunswick, NJ, located in the heart of New Jersey’s pharmaceutical industry. -
News Sirio Pharma receives sports nutrition production licence approval
The approval enables Sirio to add special sports nutrition ingredients into products and covers six functional classes for ‘protein supplementary’, ‘speed & power’, ‘endurance’, ‘energy control’, ‘energy supplementary’, and ‘post-exercise recovery’. -
News All Recipharm facilities ready for EU serialisation, regardless of Brexit
The CDMO invested EUR 35 million into its operations and launched a 3-year programme to provide a compliant serialisation solution for its customers. -
News Chemspace and LabNetwork collaborate to extend access to chemical databases
Collaboration provides drug discovery researchers access to European distribution and local support for library of in-stock and on-demand compounds. -
News Bayer and Orion (Fermion) experiences in development of API process for Darolutamide
The compound was first synthesized and developed by Orion, who partnered it with Bayer for Phase III clinical development. Bayer holds the global marketing rights, while Orion and its subsidiary Fermion handle all the CMC and manufacturing activities f... -
News SGS to conduct its first malaria human challenge trial in Belgium
The advanced candidate compound undergoing the trial has demonstrated potential to target malaria parasites before they have a chance to establish the symptoms of malaria. -
News Andrew Alliance and Sartorius collaborate to provide software-connected pipettes for life science research
Researchers to benefit from an innovative software-connected pipetting system, bringing improved reproducibility and traceability of experiments to life-science laboratories. -
News Post-EU FMD deadline - 'business as usual'?
Partner connectivity will be an immediate and ongoing challenge beyond the deadline and several pharma companies are likely to break agreements with current suppliers through breach of contract clauses. -
News BASF opens Application Technology Laboratory for personal care in Nigeria
Local competence in application and formulation technology for sub-Saharan African personal care market. -
News FDA approves first Nanobody-based medicine for adults with aTTP
Cablivi is the first US approval for Sanofi's new rare blood disorders franchise. -
News Brexit - the EU FMD's painful problem
Teething problems are no stranger to the implementation of new regulations, but with the EU FMD, Brexit came like a set of wisdom teeth – late, painful and problematic. -
News Alveofact® - Avoidance of plastic waste
Avoidance of plastic waste - New packaging for Alveofact® -
News Claudio Puglisi, COO of PQE Group, chosen for PDA Executive Committee
PDA Italy Chapter has chosen the new Chief Operating Officer of PQE Group, Claudio Puglisi, as a member of the executive committee for the role of Chapter Liaison and External Relationship. -
News Pharmapack Europe 2019 Award Winners
Awards showcased a wide range of industry-changing discoveries in drug delivery, packaging and automation. -
News New series of advanced freeze dryers
The LyoConstellation series allows the development of highly aggressive cycles and the mapping of the furthest reaches of theoretical design space for a formulation. -
News Femimens and Femicycle - new products in the gynecological portfolio.
Femimens and Femicycle are the new products in Ananta Medicare Ltd's gynecological portfolio. -
News Hitachi Chemical to acquire apceth Biopharma
Acquisition will strengthen Hitachi's presence in the second-largest cell and gene therapy market in the world. -
News Recycled Plastic Tube - from bottles and ocean plastics
Our recycled tubes are made from a mixture containing 75% of these recycled, food-compliant plastics. -
News FDA approves first generic Advair Diskus
Mylan's Wixela Inhub is the first FDA-approved therapeutically equivalent generic of Advair Diskus (fluticasone propionate and salmeterol inhalation powder) for certain patients with asthma or COPD. -
News Sartorius joins the community of the National Institute for Innovation in Manufacturing Biopharmaceuticals
Company hopes to contribute substantially to further optimizing and accelerating the current manufacturing processes in the biopharmaceutical industry. -
News Particle Sciences partners with Encube Ethicals to develop novel vaginal rings
The partnership will focus on rings for the administration of existing approved drugs, improving drug effectiveness and patient compliance. -
News Manufacturing consultancy up 150% as life sciences prepare for a digital future
Increasing appetite for digitised manufacturing and using data to improve GMP manufacturing. -
News Santen’s Tampere manufacturing facility to be acquired by NextPharma
Santen SA and NextPharma today announce that Santen’s Tampere manufacturing facility and related operations will be acquired by NextPharma, a leading European pharmaceutical Contract Development and Manufacturing Organisation (CDMO). The acquisition is... -
News UK and Russia sign MoU on regulatory co-operation on medicines
Medicines regulators of the UK and Russian set out key principles to support their collaboration on improving public health. -
News SGS expands testing capabilities at its Glasgow facility
Clients will benefit from a fully comprehensive range of validated biosafety methods to support cell bank and viral vaccine manufacturing and lot release of drug product. -
News BASF and B2B Cosmetics partner to enable individualized personal care products
B2B Cosmetics will contribute their Emuage technology, which has received the “Innovation Award” at the trade show Consumer Electronics Show (CES) in Las Vegas. -
News Pharmapack report predicts diversification of innovation leading to a rise in licensing and partnering
New report highlights Germany, France and Switzerland as tier-one nations for ‘drug delivery innovation’, and warns the challenge will be to scale-up and approve promising prototypes. -
News Wasdell Group granted HPRA licence at new EU headquarters
The new Irish facility will allow the company to continue to act as a gateway to Europe for its British and North American customers. -
News ChargePoint Technology expands smart monitoring technology into North America
Company's VERIFI smart monitoring hub allows production teams to manage and maintain their containment solution which, in turn, helps assure operator safety and sterility assurance related performance during pharmaceutical manufacturing processes. -
News TC BioPharm creates allogeneic cell banks for CAR-T cancer therapy products
The first completed bio-banks will be used to develop more cost-effective, safe and efficacious cancer treatments. -
News New API screening program strengthens Particle Sciences' nanomilling offering
Advanced formulation techniques such as nanomilling may provide an excellent route to improved bioavailability and enhanced therapeutic effect. -
News Complete series of products to treat extreme dry and scaly skin
Peters Krizman AG introduces a product for the treatment of extreme dry and scaly skin, in case of neurodermatitis, eczema and psoriasis and to restore the disturbed barrier function of the skin. -
News Alvotech completes US $300 million financing deal
Fuels growth and biosimilar development in a fast-growing market. -
News Inhalation Advancements
Chrissy Bell, Global Business Leader for Inhalation and Connected Health, at 3M speaks to Innovations in Pharmaceutical Technology about how the end user's experience can be enhanced through recent drug delivery technologies and how the challenges face... -
News Smarter Technology
Technological breakthroughs are changing the scope of drug delivery. In particular, inhalation technology has experienced pivotal growth in recent years. By identifying new ways to accurately and efficiently deliver drugs to the lungs, the industry is ... -
News 3M, the company that never stops innovating
3M’s inventions have improved daily life for hundreds of millions of people all over the world since 1902. Silvia Perez, President and General Manager of the Drug Delivery Systems (DDS) Division of 3M, highlights some of her company’s recent innovation... -
News Could Devices Help Pharma Improve Outcomes?
Chrissy Bell, Global Business Leader – Inhalation and Connected Health, 3M Drug Delivery Systems shares how advances in technology, digital health, and data protection could help patients better manage their drug regimens. -
News Sartorius introduces BIOSTAT RM TX single-use bioreactor
Flexsafe RM TX bags enable reliable process performance for optimal cell growth. -
News Fiera Milano to host CPHI Worldwide 2020
The exhibition district will see the world's largest pharma industry event. -
News Product registered in US market
Bliss GVS Pharma Ltd. have received the authorisation for commercialisation of Bisacodyl suppository in the US market. -
News FDA approves first generic version of Sabril tablets
Approval of generic vigabatrin tablets granted to Teva Pharmaceuticals USA. -
News New software platform improves freeze drying process control
The LyoS 2.0 control system software is fully configurable and fully validatable for regulated pharmaceutical and biotechnology applications. -
News BASF’s new active ingredients target skin conditions’ root causes
New active ingredients coming from the research of BASF’s innovation platforms on epigenetics, microbiome and extraction. -
News Borealis and Borouge launch new regulated Bormed™ healthcare solution for medical and diagnostic devices at Pharmapack 2019
Borealis and Borouge, leading providers of innovative, value-creating plastics solutions, announce the introduction of Borealis Bormed™ BJ868MO, a high flow, heterophasic polypropylene copolymer used for the production of medical and diagnostic devices... -
News MedPharm expands to de-risk customers' development programs
The $4-million investment will be used to expand topical dosage formulation development services and innovative performance testing on topical and transdermal pharmaceutical products. -
News Univercells introduces breakthrough vaccine manufacturing platform
The automated NevoLine bioproduction system that facilitates safer, faster and closed bioprocessing in a much smaller footprint. -
News AFA BLOG
Asociacion Forum Auditorias has a blog in which they cover the trending topics in the industry. -
News Allergan to establish R&D presence in Cambridge, Massachusetts
The new, strategic presence will allow the company to more easily interact and engage with venture firms and start ups in the area. -
News Expedeon signs supply and license agreement for is Lightning-Link technology
The technology will enable Cell Guidance Systems to manufacture TRIFic detection assays on-demand with highly reproducible and scalable results. -
News Lilly launches new robotic laboratory
Web-based platform provides broad access to the first-ever cloud laboratory to integrate automated biology and chemistry research capabilities in a closed-loop system. -
News Lilly to bag Loxo Oncology for $8 billion
Acquisition will broaden the scope of Lilly's oncology portfolio into precision medicines. -
News BMS and Celgene merge to create premier innovative biopharma company
Significantly expands Phase III assets with six expected near-term product launches, representing greater than $15 billion in revenue potential. -
News Sharing knowledge to improve scientific exchange in UK drug discovery
ELRIG UK and British Pharmacological Society initiate a 2-year strategic alliance. -
News Novartis offers to acquire CellforCure
Proposed acquisition will expand manufacturing capacity for innovative cell and gene therapies. -
News BMS receives offer from Taisho to purchase UPSA
The potential transaction would position BMS to further refine its portfolio to focus on transformational medicines for patients facing serious diseases. -
News An increased risk of a hard or ‘no deal Brexit?
A ‘no deal’ Brexit will change and burden how all industries that move materials and goods across borders, including the pharmaceutical and medical device sectors, do business. -
News Recipharm to potentially end operations in Ashton-under-Lyne facility
The operation has not been profitable for several years; no likely prospect of it being able to deliver an acceptable return in the medium term. -
News Alcon acquires Tear Film Innovations
Tear Film's iLux Device offers innovative, customizable treatment for Meibomian Gland Dysfunction, a leading cause of dry eye. -
News PrestigeBio Pharmaceutical Celebrates Successful Completion of State-of-the-Art GMP Facility in South Korea
PrestigeBio Pharmaceuticals Co., Ltd. (PBK) has celebrated the successful completion of their first GMP facility located in Osong, South Korea at their official grand opening. -
News Prestige Biopharma partners with Cipla Ltd to market key cancer biosimilar
Prestige BioPharma (“Prestige”) announced today that it has reached a licensing agreement with Cipla Limited (“Cipla”) for its trastuzumab biosimilar (HD201) under which Cipla will have exclusive rights to distribute and market the drug in selected eme...
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance
.jpg)
.jpg)


.jpg)
%20(2).jpg)







.jpg)
%20(2).jpg)



%20(2).jpg)
.jpg)







.jpg)
.jpg)


.jpg)
.jpg)











.jpg)

.jpg)
%20(2).jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)



.jpg)

.jpg)

.jpg)
.jpg)

.jpg)
.jpg)

.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
%20(1).jpg)