How will the New Administration Impact the US Pharma Market?

CPHI

-
2020On CPHI since

Other Content from CPHI (263)
-
News CPHI Podcast Series: the power of digital marketing in pharma
Digital marketing is a valuable tool for many industries, and the pharmaceutical and healthcare industry is no exception. The CPHI Podcast Series covers how marketing can be used by companies to increase their engagement and overcome challenges. -
Brochure Sustainability in Pharma - Infographic
CPHI spoke with pharma supply chain leaders to dive into the industry’s current status, challenges, and strategies when it comes to sustainability. Our experts comment on: what sustainability means to them what are the main sustainability challenges they face how they are actively working towards a more sustainable future The infographic also shows how we, at CPHI, are striving for a more sustainable future: running our events in as sustainable manner as possible encouraging sustainable ideas being conscious of the materials we use providing educational content on sustainability Download our free infographic to get the inside scoop on all things sustainability, direct from the expert’s leading our industry! -
News Press release: CPHI Sustainability Report 2024 – a guide to a greener industry
The CPHI Pharma portfolio is pleased to announce that their annual Sustainability Report has been published, with thought leaders outlining a guide to a greener pharmaceutical industry. -
News Seeking new opportunities: Novel drug approvals of 2024 infographic
In this informative infographic find out about the drugs that have been approved so far in 2024 by the US FDA. Covering a range of fields from infectious diseases to oncology, to drugs for diagnostics this year has seen a diverse range of approvals. We'll be seeing more therapeutics being developed across areas such as rare diseases as the year goes on. -
Brochure CPHI Annual Report 2022: Psychedelic Approvals, VC Driven Contract Services Boom and Next Generation Manufacturing.
The sixth CPHI Annual Survey will explore the perspectives of some 400 pharma executives, evaluating the likely trends in 2023 and the reputations of all major pharma markets. The analysis spans insights from some 35 questions and is a key bellwether of industry prospects in the year ahead. The second component of the CPHI Annual Report – now in its 10th edition – features the detailed analysis of 11 global experts who explore all facets of the industry today and look ahead to predict the major trends -
News Women in Pharma Anniversary: Celebrating Our Heroines of Pharma
Our Women in Pharma interview series is approaching its 1-year anniversary this month, and to celebrate, we are highlighting the Heroines of Pharma that our very own Women in Pharma admire. -
Whitepaper Trend Report - The True Cost of API Price Rises
As a direct result of various environmental and socio-political events in the last 2 years, the pharmaceutical industry is grappling with the rise in API pricing across the globe. This report will mainly consider factors in the recent hike in API prices and how these might be directly and indirectly addressed by manufacturers and CDMOs in order to meet their existing and future development goals. A separate section will review how external bodies such as governments of certain member states and regulatory bodies can aid in relieving the pressures of increased API pricing. Gathering insights from experts and manufacturers in the pharmaceutical industry, this report will offer a comprehensive report on the market trends, challenges and potential solutions to increased API pricing. .iiris-banner-imagecontainer { display: none !important; } -
News CPHI North America 2024 – From the Floor
Welcome to Philly! CPHI North America once again graces the Philadelphia Convention Center, 7–9 May 2024. -
Whitepaper CPHI Pharma Trends 2021 Report: out now!
2020 has been one of the most turbulent years in recent history and has cast a shroud of uncertainty over the future. While pharmaceutical supply chains have proved resilient to the unprecedented shock of COVID-19, many more questions will be asked of the industry over the coming months. Pharma’s response to the pandemic in terms of vaccines development and distribution of treatments has grabbed the majority of the headlines but the major issues facing the industry are by no means exclusively coronavirus related. In interviews with key industry experts, this report identifies the major and emerging trends that are sure to be prominent in 2021, as pharmaceutical manufacturing and outsourcing continue to respond not only to the challenges that the ongoing global crisis has presented, but also to evolving obstacles and hurdles that are typical in such a highly-regulated sector. -
News The final six: meet the last Start-Ups exhibiting at CPHI NA 2024
The last six Start-Up companies that have made the list to exhibit in a dedicated area in Philadelphia at CPHI North America. Learn more about the companies and what makes them unique in the latest infographic. -
Whitepaper CPHI Annual Report 2021: Golden Age for Discovery, Technology and Growth
The CPHI Annual Report is a comprehensive and critically important publication that analyses key trends and innovations forecast by our panel of world class experts, as well as the CPHI Pharma Industry Rankings (an annual executive survey of pharma professional). Now in its ninth year and launched at CPHI Worldwide in Milan, the report is an essential read for executives looking to get a head-start today on the shape of tomorrow’s industry.
The fifth CPHI Annual Survey explores the perspectives of some 370 industry executives from over 35 countries. Evaluating the likely trends in 2022 and the reputations of all major pharma markets across more than 10 metrics. The data provides an annual insight into the growth and development of the industry globally.
The second component of the CPHI Annual Report features the analysis of 10 global experts who explore all facets of the industry today and look ahead to predict the major trends of tomorrow.
This year’s expert submissions include contributions from ten experts spanning topics as diverse as CDMO future prospects for capacity and M&A, innovation in manufacturing, mammalian biomanufacturing, COVID-19 deals for contract manufacturers, biologics CDMOs in China, nutraceuticals, regulatory and excipients. -
News The 2024 Pharma Outsourcing Forecast: strategic partnerships across global landscapes
For the pharmaceutical industry, outsourcing drug development, manufacturing, and research operations to contract organisations is not just a passing trend – increasingly, the partnership between drug sponsor and drug vendor is one of vital importance. For those pharmaceutical companies and biotechs looking to focus on their core competencies, pharmaceutical outsourcing partnerships are essential to smooth and efficient operations in the pharmaceutical supply chain. In fact, more than 50% of pharmaceutical executives anticipate their outsourcing activities to increase in the next 5 years. -
Whitepaper Clinical Development Success Rates and Contributing Factors 2011-2020: Download the Report
Clinical Development Success Rates and Contributing Factors 2011-2020
Biologics have 9.1% likelihood of making it through the clinic to approval, compared to 5.7% for new molecular entities, according to this analysis of 10 years of Biomedtracker drug development data by BIO, Informa Pharma Intelligence and QLS, available for download here.
Biologic products advance through clinical development with a higher success rate than traditional small molecules, a new analysis of clinical development success from Informa Pharma Intelligence, the Biotechnology Industry Organization and the Quantitative Life Sciences shows.
“Biological complexity in drug modalities generally leads to higher LOA,” or likelihood of approval, the analysis concludes. New molecular entities – conventional small molecules – have a 5.7% chance of moving through all three phases of clinical trials to US Food and Drug Administration approval, while biologics had a 9.1% probability. Vaccines – with 10 times fewer candidates than other biologics – clocked in with a 9.7% chance of a Phase I candidate eventually gaining approval.
Informa and BIO analyzed a decade of data collected by Informa’s Biomedtracker database, identifying 12,728 clinical phase transitions, which mark a program’s movement out of a clinical phase, commonly by advancement or suspension. QLS Advisors applied machine learning to “uncover underlying drivers of success” in the 9,704 clinical drug development programs included in the study, “Clinical Development Success Rates and Contributing Factors, 2011-2020,” published 17 February 2021.
Overall, for all modalities, the LOA from Phase I was 7.9% for the 2011-2020 period. -
News CPHI Podcast Series: How manufacturing in Africa can improve patient access to medicine
The latest episode of the CPHI Podcast Series features Daniel Mora-Brito, a thought leader in pharmaceutical emerging markets, talking to Digital Editor Lucy Chard about the importance of the development of manufacturing in Africa. -
Whitepaper CPHI Annual Report 2020
The CPHI Annual Report is a comprehensive and critically important publication that analyses key trends and innovations forecast by our panel of world class experts, as well as the CPHI Pharma Industry Rankings (an annual executive survey of pharma professional). Now in its eight year and launched at the CPHI Festival of Pharma, the report is an essential read for executives looking to get a head-start today on the shape of tomorrow’s industry.
The rankings compile the answers from nearly 40 questions for 550 global executives, covering perceptions across the leading pharma economies for a range of indices including: ‘quality of API manufacturing’, ‘quality of FDF manufacturing’, ‘innovativeness’, ‘growth’ and ‘biologics manufacturing’ amongst many others.
This year’s expert submissions include contributions from ten experts spanning topics as diverse as CDMO capacity forecasts, biologics manufacturing, AI, and sustainability to macro supply chain geo-realignments, digital adoption and patient centricity. Running as a series of opinions and articles, the CPHI experts call upon their considerable commercial and technical acumen to prophesize the future direction, technologies, opportunities and threats in pharma.
The CPHI Annual Report 2020 includes 15 pages of survey data analysis – including the CPHI Pharma Index, executive supply chain concerns, COVID implications, and the national pharma rankings – 10 expert contributions, spanning 30,000 words and 74 pages of insights.
Still to come: not content with one edition, later this year, CPHI will also be issuing a second supplementary CPHI Annual Report 2020, which will include 4 further articles and executive survey results, looking ahead to 2021. -
News Next up: five more Start-Up companies exhibiting at CPHI North America 2024
The latest infographic introducing you to the next five Start-Up companies who will be gracing the exhibitor show floor in their dedicated Start-Up Market in Philadelphia for CPHI NA 2024. -
Whitepaper 2021 Sustainability in Pharma Packaging, Devices and Manufacturing Report
A starting point to address a big issue, hear from three pharma leaders on paving the way for sustainability in pharma.
In this short report, Informa presents a call to action from its sustainability lead, three contributions from CPHI sustainability experts, as well as a short review of the recent CPHI Sustainability Index. -
News The CPHI North America agenda - see what's in store
CPHI North America will once again be held in Philadelphia this May. Philadelphia has been home to CPHI North America for several years and continues to be a hub for the US pharmaceutical market. -
Whitepaper US Pharma Market 2022 and Beyond
Ahead of the return of CPHI North America, Informa Markets has commissioned an in-depth report into the US pharma market focusing on how the pandemic has affected public discourse and political priorities, and how it could alter the pharmaceutical supply paradigm in the medium term. Analysis from six leading US experts – following extensive interviews and data collection – provides an essential guide to near-term priorities, medium-term capacity constraints and longer-term shifts underway in the US pharmaceutical market supply chain. -
News LEAP-ing into the future of pharma – the CPHI Middle East Vision
On March 6, 2024, the CPHI team hosted the Future of Pharma Forum at LEAP 2024, Saudi Arabia’s premier destination for tech leaders and innovators to meet and collaborate. -
Whitepaper US Pharma Market Trends 2021
In interviews with key industry experts, this report identifies ten major and emerging trends that are sure to be prominent in 2021, as US pharmaceutical manufacturing and outsourcing continue to respond not only to the challenges that the ongoing global crisis has presented, but also to evolving obstacles and hurdles that are typical in such a highly-regulated sector. -
News Informa Markets International Women’s Day Panel Discussion
On March 7, 2024, the Informa Markets Amsterdam office hosted an International Women’s Day Breakfast and Panel Discussion to celebrate the women who drive the B2B events industry forward, including members of the CPHI team. -
Whitepaper CPHI Pharma Post-Pandemic Legacy Trends Report 2021: Out Now!
This special CPHI Report is split into two halves featuring an expert roundtable, global survey data analysis, and detailed expert contributions.
In Part one there is an in-depth write-up of the recent Post-Pandemic Roundtable, as well as analysis of the CPHI executive survey data on ‘opportunities in the latter half of 2021 and implications for pharma manufacturing’.
Part two includes three expert contributions on ‘the implications of QbD for excipients’, ‘Blockchain in Pharma Trends’, and ‘Crystal Ball Predictions for Pharma in 2025’. -
News CPHI Podcast Series: What to expect from novel excipients?
In this month's episode of the CPHI Podcast Series, Digital Editor Lucy Chard is joined by Nick DiFranco, the Global Market Manager for Novel Pharmaceutical Excipients at Lubrizol Life Science to discuss the ins and outs of excipients, how they are developed and the product trends in the market right now. -
News What did we learn from Pharmapack 2024? The green edition
Pharmapack Europe, held in Paris each year is a great opportunity to see the latest innovations and trends in pharmaceutical packaging and to hear from the companies and experts in the area on what aspects are most important to them in their products and businesses year on year. -
Video Keynote: Unleashing the Novartis AI & Analytics potential - Producing Good Data at the Source
In this session you hear from Johannes Galatsanos, Head of Data - Lean Digital Core ERP Transformation from Novartis. -
Video Transactional Strategies to Access Future Therapeutics and Technologies
What is the best strategy to access novel therapeutic approaches and technologies? Licensing vs M&A? How to build a stake in competitive future therapies and innovations How to beat competitors to your desired goals Exploiting innovation arising in non-medical fields and other industries -
News Navigating the Future: Challenges and Opportunities in Pharma Innovation and Investment – CPHI Barcelona 2023 Roundtable Report
In this comprehensive downloadable report, hear from a range of experts in finance and investment in the pharma industry on what investment trends will be shaping the future of the industry, in Catalonia, and the wider world. -
Video Delivering on the promise of cell and gene therapies: a patient-centric approach
Cell and gene therapies are at the forefront of personalised and precision medicines. By better understanding and harnessing the human body’s own biological/immunological processes to fight or even cure disease, these “living drugs” hold tremendous promise for millions of patients with high unmet medical needs. Delivering on that promise, however, requires navigating product and patient journeys that are unique from those that have long-dominated pharmaceutical development. From manufacturing and complex logistics to regulatory pathways and patient recruitment, multiple sectors, entities, and processes must be carefully and consistently aligned to patient-critical pathways.
In this session, attendees will gain insights into the complexities of the CGT ecosystem and the challenges that must be overcome to successfully move these therapeutics from the laboratory to the patient. The presentation will outline best practices, tools, and integrated solutions for streamlining CGT development and discuss key areas of focus for improving the journey of patients enrolled in CGT clinical trials and beyond.
Key takeaways will include: An understanding of the scale of complexity of products and patient journeys for cell and gene therapies Best practices for aligning clinical and commercial processes and workflows to patient-critical pathways Integrated approaches and solutions for accelerating the journey from laboratory to patient -
News CPHI Podcast Series: Start-ups take centre stage at CPHI Barcelona
The first episode of the CPHI Podcast Series since we attended CPHI Barcelona in October covers the Start-up market at the event, with expert Matthew Wise joining Editor Lucy Chard to discuss the event. -
Video The State of Advanced Genetic Therapies in 2022
The pipeline for advanced genetic therapies has grown exponentially since 2014, and now totals over 3,500 different developmental candidates. This session provides a comprehensive global snapshot of this R&D landscape, spanning gene, cell, and RNA-based drug modalities. This includes analysis of drug pipelines and active clinical trials for prominent classes such as gene transfer, gene editing, genetically modified cell therapies, messenger RNA, and non-coding RNA. The common diseases targeted and sponsors leading these advances will also be reviewed, as well as important investment trends.
The presentation will help professionals involved in any aspect of developing and commercializing advanced therapeutics stay abreast of the latest developments. -
News CPHI Barcelona 2023 Pharma Awards: and the winners are...
The much anticipated Pharma Awards 2023 were held at CPHI Barcelona this week at the Fira Barcelona. -
Video mRNA based therapies & their regulatory pathway
mRNA has been called the software of life. It can help treat everything from cancer to cardiovascular diseases and can be used on high-scale as well as for niche indications. Covid-19 has put mRNA technology in the spotlights. The technology is therefore appealing to investors. However, how are these medicinal products regulated - are the vaccines differently regulated or are we taling about gene therapy altogether.
After this talk: you’ll understand why this topic is relevant in the development of mRNA based therapies you’ll feel empowered as to where to place vaccines, ‘ordinary’ medicinal products and ATMPs (gene therapy, somatic cell therapy) in the legal space -
News CPHI Barcelona 2023 – From the Floor
It's that time of year again - the biggest pharma event of the year comes to Spain this October for CPHI Barcelona 2023. Follow our Editors Vivian Xie and Lucy Chard as they worked their way through the conference, bringing highlights from content sessions, panel discussions, and much more from Fira Barcelona! -
Video Microbiome Therapeutics Market and its Potential for New Therapies
The human microbiome enlists all the microorganisms and their related metabolites/products identified in and on the human body. Technological advancement has enabled the assessment of the pleiotropic effects of the human gut microbiome in health and diseases. With the extensive role of microbes in human health, they have an enormous potential to be used as therapeutics for disease management and acts as the potential source to achieve the objective of personalized therapy by overcoming key issues like interpersonal variability and stability in every type of environment.
This presentation aims to address the current knowledge and landscape of microbiome therapeutics, provides examples of existing health-and-disease applications and discusses the potential future directions of microbiome modulations. -
News Infographic: The Industry Disruptor Watchlist
CCD Partners in conjunction with CPHI Barcelona have created a Watchlist for some of the Start-Ups and independent small businesses attending CPHI Barcelona this year. The Watchlist covers a snapshot of 21 Start-Up companies and includes details about the businesses and expert insights as to how these companies are set to shake up the future of pharma. -
Video Enabling a Digital Culture Through Integrated Business Processes
The acceleration of digital transformation across industries has been a prominent theme in the wake of COVID-19. In the pharmaceutical industry, the pandemic highlighted the need for agility and resiliency within the supply chain, fueling an increased urgency for the adoption of Pharma 4.0 technologies that bring together the workforce, customer, equipment, supplier, and environment to help businesses gain greater organizational clarity and process reliability.
Digitally enabled systems that improve performance through integrated business process, manufacturing automation, and supply chain integration are essential for responding to industry and environmental challenges and ensuring that safe, high-quality products get to patients when and where they need them.
This presentation will share our perspective for achieving digital transformation and digital culture enablement, focusing on: Innovative solutions for meeting market demands and managing increasing complexity through integrated business process Opportunities for attaining new levels of operational excellence through proven technologies to drive digital transformation and Pharma 4.0 journey Key considerations for transforming the traditional supply chain model into a digitally integrated value chain network -
News Pharmapack Survey and Annual Report 2023
The annual report from Pharmapack 2023 covering the drug delivery market trends across Europe and the US, and how Pharmapack acts as a bridge between the global markets. -
Video Movement Towards Decentralized Trials
A review of how big pharma has reacted to the trend: who is going virtual with their trials? The usage data of virtual trial elements pre and post pandemic Predictions for the future -
News Trend Report: The True Cost of API Price Rises
Download our Trend Report to discover what the current landscape of APIs and their pricing means for the pharmaceutical industry in the future. -
Video Digital Health Value Pools and Digital Health Reimbursement
Have you wondered what value digital health brings to health systems?
This session will cover the ~25 main digital health value pools on health system level and show what value digital health solutions provide. In addition, digital health reimbursement is addressed, with a particular focus on the new digital therapeutics' reimbursement process in Germany (the so-called “DiGA process”). -
News Knowledge is power when it comes to managing ESG risks – CPHI Frankfurt preview
For more on managing exposure to ESG risks, register to attend CPHI Frankfurt this November -
Video Data of this World United – What it Takes to Enable Health Data Collaboration Across Organizations and Industries
Digitalization in biopharma is more complex than just replacing paper documents with electronic counterparts or hiring data scientist In the context of bioprocessing, the industry needs a better way to transition quickly from lab to clinical to commercial manufacturing Speed to clinic requires training of personnel in simulated GMP context, just as pilots are trained on simulators More importantly, cross-training will be required to enable common technical conversations through the training of IT professionals and other support roles Just as every employee is now trained on using computers, they will be expected to be data savvy – self-service dashboards, design of experiments To achieve its strategic goals, NIBRT continues to partner with other leaders in the space through collaboration with Siemens and BCG -
News Pharma needs ‘mindset change’ to unlock benefits of digitalisation – CPHI Frankfurt preview
For more information about digitalisation in pharma, register to attend CPHI Frankfurt 2022. -
Video Sustainability Strategies in the Life Sciences Industry
This short session answers the most pressuring strategic questions with regards to sustainability: Why does it matter? Where to focus? How is it measured? What is the current industry status? How to improve?
Did you enjoy this session? If so, you might like to visit our Connect to Frankfurt platform, where you can browse our collection of 30+ on-demand webinars and learn about our upcoming CPHI Frankfurt event, taking place 1-3 November in Frankfurt, Germany.
Bringing together the global supply chain under one roof, CPHI Frankfurt puts you at the heart of pharma. Can’t attend in person? You can access many of the event offerings online! Browse our exhibitor list, arrange meetings, view on-site content and network – all from home.
Click here to register
-
News ‘Incredible potential’ of cannabis products in healthcare - CPHI Frankfurt preview
CPHI caught up with Monica Vialpando to discuss developments in the cannabis space and preview her CPHI Frankfurt panel discussion -
Video Building Future Pharmaceutical Talent: Students Guide
This session will help you understand real-world life as a student entering the Pharma industry post-pandemic. We will learn more about the role a CDMO plays in the pharmaceutical industry and the importance of hiring university and school early careers from the lens of a business leader. Finally, we will hear from an industry leader in talent acquisition on those tips and tricks that every student should be doing to secure their first job tomorrow!
Did you enjoy this session? If so, you might like to visit our Connect to Frankfurt platform, where you can browse our collection of 30+ on-demand webinars and learn about our upcoming CPHI Frankfurt event, taking place 1-3 November in Frankfurt, Germany.
Bringing together the global supply chain under one roof, CPHI Frankfurt puts you at the heart of pharma. Can’t attend in person? You can access many of the event offerings online! Browse our exhibitor list, arrange meetings, view on-site content and network – all from home.
Click here to register
-
News Establishing a digital marketing strategy - a visual guide
Are you looking to generate qualified business leads year round, boost your brand exposure and stand out from the competition? Establishing a compelling digital marketing strategy will help you achieve all the above, and more. -
Video The Ukrainian Pharmaceutical Industry: Challenges and Opportunities Ahead
Introduction-Pharmaceutical Industry Review Magazine- Svitlana Havrylenko, Editor in Chief, Pharmaceutical Industry Review Magazine Ministry of Health - Oleksandr Komarida, First Deputy, Minister of Health of Ukraine Ukrainian Pharma Market overview for the 1st half of Y2022. Market in war turbulence Iryna Gorlova, General Manager, SMD «If You're Going Through hell, Keep Going». Pharmaceutical Business in the Face of Real Force Majeure Anatoliy Reder, General Director, INTERCHEM SLC “Resilience in the Time of War: How Sustainable Pharmaceutical Business Can Survive Deepest Crisis and Demonstrate Tendencies for the Future Growth” - Volodymyr Kostiuk, CEO, Farmak JSC "Lekhim" Group of Pharmaceutical Companies - Mykhailo Renskyi, Director of Development Department, "Lekhim" Group of Companies
Did you enjoy this session? If so, you might like to visit our Connect to Frankfurt platform, where you can browse our collection of 30+ on-demand webinars and learn about our upcoming CPHI Frankfurt event, taking place 1-3 November in Frankfurt, Germany.
Bringing together the global supply chain under one roof, CPHI Frankfurt puts you at the heart of pharma. Can’t attend in person? You can access many of the event offerings online! Browse our exhibitor list, arrange meetings, view on-site content and network – all from home.
Click here to register
-
News On-demand medicine manufacturing secures supply 'whether on the battlefield or hospital' - CPHI Frankfurt preview
CPHI caught up with Eugene Choi to discuss point-of-care medicine manufacturing ahead of his CPHI Frankfurt presentation -
Video FebriSol: The Low Cost Adherence Intervention for People and Profit
According to the WHO, “Increasing the effectiveness of adherence interventions may have a far greater impact on the health of the population than any improvement in specific medical treatments.”
Unfortunately, many of the available interventions are cumbersome, inaccessible and or too expensive to be applied, even at scale. In my talk I will present FebriSol, an innovative and cost-effective packaging intervention that helps patients remember to take their chronic medication.
The design, journey and challenges I faced in the development of FebriSol; Treatment adherence as a shared value business model; My vision for the future of the product.
Click here to register
-
News Connect to Frankfurt - Five reasons to tune in
Get a jump-start on your CPHI Frankfurt experience with exclusive access to our online event platform. -
Video "Your Order has Been Dispatched”: Opportunities in Moving Patient Care Closer to the Home
In the throes of the COVID-19 pandemic, global healthcare systems increased their focus on providing medicines outside of a hospital setting, whether that be in outpatient clinics, mobile clinics, pharmacies or at-home care. As a result, pharma companies have a unique opportunity to seize on this momentum by engaging with policymakers to diversify the locations a patient can receive care. This will allow flexibility within the healthcare system and give patients the best experience possible.
This session will explore the dynamics of how medicines are moving nearer to the home and highlight how pharma can both champion the movement and achieve success in the space.
Click here to register
-
News For supply chain security, ‘don’t live in the moment’ - CPHI Frankfurt preview
CPHI caught up with Jim Fries to discuss sustainable supply chains ahead of his CPHI Frankfurt panel discussion -
Video Innovation in Pharma and Life Science: This is Happening!
Technological innovation in Pharma and Life Science is happening now!
Metaverse, Artificial Intelligence, Data Analytics, Machine Learning, Digitalization: these topics are ranking high on the agenda of the business leaders who understand the competitive advantages that can be seized by moving now into the future of this sector.
Infomineo's presentation focuses on some of the most relevant steps taken by manufacturers and service providers who are leading the way in this technological evolution.
Click here to register
-
News UPDATED: COVID-19 Vaccine and Therapeutic Development Tracker
The latest coronavirus updates and developments impacting the global pharmaceutical supply chain. Includes the latest news surrounding the therapeutic developments, updates to vaccines and boosters, and the global pharmaceutical regulations that need to be met. -
Video Future Predictions on Drugs from Sales & Forecast
Future Predictions on Drugs from Sales & Forecast Looking ahead to see the shape and direction of the Pharma market. Using Sales and Forecasts to see which areas are predicted to lead or lag. Identifying key therapy areas and indications as well as molecule types that may come to dominate the market. Seeing which companies are most likely to reap the rewards of future sales and positioning
Click here to register
-
News Medinfar awarded first ever CPHI Verified Certificate
CPHI Verified certification improves supply chain transparency, providing a legal, regulatory and quality benchmark -
Video Bringing It Home: The New Generation of Clinical Trials
Bringing It Home: The New Generation of Clinical Trials
Click here to register
-
News China sees huge bounce in pharma rankings ahead of hybrid CPHI & P-MEC China
China is the pharma industry’s biggest gainer in the 2021 CPHI Annual Rankings, with digital matchmaking driving new waves of partnerships -
Video Transforming Pharmaceutical Supply Chains with Synthetic Biology
Antheia is a synthetic biology company developing rapid-response biomanufacturing processes for Active Pharmaceutical Ingredients and Key Starting Materials. In this session, Christina Smolke, PhD, CEO and co-founder of Antheia will dive into synthetic biology as a manufacturing technology and its potential to build more resilient and efficient pharmaceutical supply chains.
Join to learn more about Antheia’s platform approach, synthetic biology, and the future model for pharmaceutical manufacturing.
Click here to register
-
News CPHI Worldwide is back, and this time it’s hybrid
A new in-person and online edition of CPHI Worldwide, transforming your event experience.
-
Video DARC: exploratory biomarker for retinal disease
Novai is commercialising DARC, a combination biomarker consisting of Annexin A5 conjugated to a fluorophore (Dy776). ANX776 binds to stressed/apoptotic retinal cells which are visualisable as white spots using equipment routinely used by opticians, optometrists, and ophthalmologists. The images are then analysed to detect cellular level of disease activity.
When a therapeutic retards disease activity and reduces cell stress/death, DARC signal goes down in real time, providing prompt indication of drug effectiveness within days. DARC is a platform technology, currently being developed as a biomarker, with future potential as a glaucoma prognostic/diagnostic and a drug delivery theragnostic.
Click here to register
-
News Next stop, Milan - Welcome to Italy's life sciences hub
Milan is home to an internationally competitive pharmaceutical, biotechnology and functional foods hub, with strong growth prospects and a commitment to innovation across its entire supply chain. Italy is also home to one of the most advanced and efficient healthcare systems in Europe - making it an exciting destination for the 2021 CPHI Worldwide event. -
Video Innovating Stem Cell Medicine with the First Method for Routine Stem Cell Counting
Asymmetrex® markets the first and only technology for routine counting of therapeutic stem cells, like those in life-saving bone marrow transplants. The patented technology, “kinetic stem cell (KSC) counting,” tells physicians the dosage of stem cells in such treatments. KSC counting also improves biomanufacturing of stem cell and gene therapy products; improves stem cell science research; and reduces the cost of drug development by early detection of drug candidates that fail later because of stem cell toxicity.
Despite these many benefits, industry and market adoption of KSC counting has been challenged by entrenched non-quantitative practices and misinformation. Asymmetrex® will present three strategies deployed to overcome these barriers to its vision of establishing KSC counting as a new era for stem cell science and medicine: Demonstration of compelling medical, biomanufacturing, stem cell research, and pharmaceutical utility Establishing KSC counting as an industry standard for stem cell medicine Industry education campaigns
Click here to register
-
News CPHI Webinar: mRNA vaccines, Trends, Technologies and Supply Chain
The COVID-19 pandemic has meant scientists, together with pharmaceutical companies, have had to reinvent how to bring vaccines to market faster without impacting product quality, safety, or efficacy, according to experts speaking at a recent CPHI webinar. -
Video Improving Scope 3 Emission Data Quality to Drive the Decarbonization of Our Supply Chains
Recognizing that upstream Scope 3 greenhouse gas (GHG) emissions constitute a major share of a chemical or pharmaceutical company’s emissions, increasing emission data transparency and accuracy is crucial to drive emission reductions along the value chain.
In September 2022, Together for Sustainability (TfS) launched the Product Carbon Footprint (PCF) Guideline. It enables companies to calculate the PCFs of their chemical materials more precisely in a way that is harmonized across the industry, while remaining fully compliant with less specific international standards for GHG accounting. The PCF Guideline is open source.
Did you enjoy this session? If so, you might like to visit our Connect to Frankfurt platform, where you can browse our collection of 30+ on-demand webinars and learn about our upcoming CPHI Frankfurt event, taking place 1-3 November in Frankfurt, Germany.
Bringing together the global supply chain under one roof, CPHI Frankfurt puts you at the heart of pharma. Can’t attend in person? You can access many of the event offerings online! Browse our exhibitor list, arrange meetings, view on-site content and network – all from home.
Click here to register
-
News Introducing the all-new CPHI Online dashboard
Today, Informa Markets is pleased to announce the launch of the all-new CPHI Online Dashboard, which will allow CPHI clients to quickly and easily track awareness and interest in their company, products and content as well as view and download leads to start building new partnerships.
-
Video Keynote: European Regulatory Outlook
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme Adrian van den Hoven, Director General of Medicines for Europe, explores the European regulatory outlook for pharma Discussion points: The pharmaceutical industry as a key asset to the EU economy Pharmaceutical strategy for Europe Supporting a competitive & Innovative EU Pharmaceutical Industry Highlight initiatives on manufacturing competitiveness and regulatory efficiency Pharma driving sustainable post-pandemic growth Considerations for the future EU pharmaceutical strategy -
News CPHI Trend Report - CDMO Opportunities in the Chinese Market
Opportunities for contract development and manufacturing organisations (CDMOs) in China continue to grow but many questions remain over what the future of the sector will look like in the world's second largest pharmaceutical market. -
Video Optimising Patient Outcomes in Respiratory Medicine with Digital Healthcare Devices
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. When patients self-administer their medications, 60% do not take them as intended, and over half of all patients are non-adherent. The resulting cost of non-adherence and non-optimised therapy is steep, and everyone pays the price, from pharmaceutical companies and payers to HCPs and their patients. The cost of non-adherence alone is US$300 billion per year, with $188 billions of US pharma revenue lost. The rules around healthcare engagement are now being rewritten with connected ecosystems and the ability to link self-administered medications to disease management solutions. Technology has the potential to augment patient care, deliver value across the digital health ecosystem and deliver -
News US needs to look differently at public health preparedness, says former FDA commissioner Gottlieb
Speaking at CPHI North America, Dr Scott Gottlieb shares insights into how the US should prepare for future pandemics by building out vaccine capacities -
Video CPHI Pharma Awards
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. The CPHI Pharma Awards celebrates the thinkers and creators driving the industry forward through innovations, technological advancements and strategies across 10 distinct categories - from manufacturing excellence to sustainability. The Awards were open to all Pharmaceutical and Biopharmaceutical companies engaged in the design, development, manufacture, production and distribution of large and small molecule drug products. This year's winners were announced via a networking reception at the Fiera Milano on 9 November. This year's headline sponsor was Rentschler Biopharma: Your trusted partner for high-quality biopharmaceuticals -
News 5 Strategies for Engaging a Pharma Audience All Year Round
For pharma manufacturers and service providers, attending face-to-face events and exhibitions has always been a crucial tool for meeting existing clients, signing contracts, and gathering leads for new business. But with events unable to trade over the course of the pandemic, connecting with clients in an online environment and evolving to become more digitally astute has become a necessity. -
Video Milano Ecosystem for Life Sciences
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. Milano&Partners is the Milano official investment promotion agency, founded by the city and the local Chamber of Commerce. It supports the soft landing of foreign investments in the metropolitan area with a broad offer of free of charge services and promotes Milano's excellence worldwide. Milano Greater Region generates 33% of the Italian registered patents in the EU and supplies 5% of the world's active ingredients. Local Life Sciences production is worth €71 bln with 356,000 people employed. All the main national and EU trade bodies and regulators, including the EFSA (Parma) and EU JRC (Ispra), are based nearby the area. Milano ecosystem for Life Sciences session explores the macro data and performance of the region and introduce you to a few examples of excellence in the territory. -
News APIs, excipients and formulation: the bedrock of medical innovation
Our virtual CPHI Discover event (17th-28th May) is just around the corner, full to the brim with three main content tracks. Here, we explore the major trends in APIs, Ingredients & Formulation -
Video Device Design Strategy for A Connected Future
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. This session covers: Delivering Complex Technologies to End-Users Characteristics for Smaller, Smarter, Electronic, Connected Real-World Case Studies of Device Design and Development The Next Generation of Drug-Delivery Devices Medical Device vs Wellness Device - FDA Regulatory Requirements and Determining Category -
News Beyond the box: Device and packaging tech innovation vital for pharma
Our virtual CPHI Discover event (17th-28th May) is just around the corner, full to the brim with three main content tracks. Here, we explore the major trends in Drug Delivery and Packaging. -
Video Trends in Peptide Drug Discovery
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. This session explores: Characteristics of an ideal peptide drug and peptide drug target Lessons from earlier approaches that are still relevant today High-Throughput Discovery and Parallel Optimization of Peptide Therapeutics Emerging strategies broadening horizons for peptide drug discovery Discuss the Therapeutic Potential of Peptides Analyse the barriers that need to be addressed for them to reach their full potential and possible solutions -
News Drug R&D innovation reshaping manufacturing and the CDMO sector
Our virtual CPHI Discover event (17th-28th May) is just around the corner, full to the brim with three main content tracks. Here, we explore the major trends and issues surrounding Manufacturing and Outsourcing. -
Video Future Developments and Practices for mAbs and other Complex Molecules
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme and delivered in collaboration with BioProduction. The session explores: Utilising Data for Process Development and Optimisation Applying data-driven models in biopharmaceutical manufacturing to improve real-time decision-making Approaches for the Design and Selection of Single-Use Assemblies in a Clinical Drug Substance Manufacturing Facility Selection of single-use assemblies and materials requires the thoughtful assessment of process requirements, the material of construction compatibility, supply chain considerations, modularity vs customisation, and cost of goods implications. Harmonise assembly designs across multiple operations and reduce total SKU counts. -
News CPHI Trend Report: The Future of Indian API Manufacturing - Out Now!
Read this new CPHI Trend Report which, via interviews with key industry experts, explores the state of India's API manufacturing issue and how it is being impacted by the country's ambitious healthcare reform programme as well as efforts by the government to support domestic manufacturing and reduce supply dependence on rival China. -
Video Trends Shaping the Future of Cosmeceuticals
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. The session provides a snapshot of the current trends of the cosmeceuticals industry and provide an analysis of the market touching upon: Characterise opportunities and risks around cosmeceutical marketing, including regulatory considerations COVID impacts, implications of global trends (natural demand, sustainability) Possibilities posed by legislation in the US to modernise cosmetics regulations -
News Building supply chain resiliency to ensure access to medicines
With CPHI North America fast approaching, we ask Karen Flynn (President, Biologics & Chief Commercial Officer, at Catalent), Hamilton Lenox (Senior Vice President of Business Development, Sales & Marketing, at LGM Pharma), and Mike Kleppinger, Chief Commercial Officer, at AMRI) to weigh in on strategies the industry can use to build supply chain resiliency which ensure uninterrupted access to medicines -
Video Environmental Sustainability in Pharma
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. Top pharma companies are leading environmental sustainability initiatives in a sector that has been slow to react. Continued public and private cooperation is required to drive transparency, accountability, and action across the entire sector to meet international targets. The session touches upon some of the challenges and initiates and potential solutions taking place within the Environmental Sustainability in Pharma scenario Historical perspective and importance for healthcare Policy drivers and industry initiatives Commercially successful Green products Areas of unmet need Recommendations for the industry -
News US Pharma Market Trends 2021 - Download Report Now!
2020 will go down in history as a pivotal year for the US pharmaceutical manufacturing sector. Firstly, the emergence of the global coronavirus pandemic provided a seismic shock to the industry and forced major rethinking on several lines. And secondly, a bitter and hard-fought Presidential election saw Joe Biden defeat Donald Trump to usher in a complete change of approach towards pandemic management and healthcare delivery. -
Video Addressing Viability and Commercial Challenges of Manufacturing Large Biologics
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme and delivered in collaboration with BioProduction. The session explores: The Economics of Seed Train Intensification A Technical End-to-End Project Review to Ensure Commercial Viability of Biologic Products Developability to Ensure Streamlined Manufacturing Predictive manufacturability testing prior to development in order to ensure the selection of compatible molecules -
News CPHI Japan open for business!
Informa Markets' first in-person trade show of the year welcomes attendees in Tokyo as face-to-face interaction recommences among pharma community -
Video India - Pharma Innovation Landscape
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme. India, with a population of almost 1.4 billion, is becoming ever more vocal about access to good quality healthcare, which will have a huge impact on the fortunes of the country’s pharmaceutical sector going forward. While India is the largest global producer of generic drugs, there is also a rising interest in drug discovery and development for innovator products, and this session will examine India's potential to become celebrated as an innovator market. Discussion points: Opportunities and challenges in the Indian pharma market Key innovations and growth drivers Forthcoming challenges and changing skill-sets for the Indian pharmaceutical industry Potential breakthroughs in next-generation innovator products The future of Indian pharma -
Video Pharma 4.0, Automation & Assembly Solutions for Smart Manufacturing
This session was originally broadcast as part of the CPHI Worldwide 2021 digital content programme The next generation of pharma manufacturing is already here, and 'Pharma 4.0' represents an exciting opportunity to optimise engineering processes with a more streamlined, digital approach. This session explores the latest in automation, connectivity and digitisation: Current Market Trends Pharma 4.0 and digital transformation Benefits of modular manufacturing in pharma Automation and digitization for device manufacturing -
News CPHI Online Report - Pharma Market Trends 2021
Twelve pharma market trends we anticipate across 2021 for the drug development, manufacturing and outsourcing sectors. -
Video API Trend Outlook 2022
This session was originally broadcast live on 25th October as part of the CPHI Worldwide content programme, it is now available to view on-demand. API sourcing and supply chains have been key areas of focus over the past 18 months, with the pandemic raising questions of where APIs should be manufactured. As the global demand for quality and increasingly novel APIs continues to grow, what does the future look like? This panel discussion brings together leading market experts to discuss the lay of the land: Market Outlook for APIs Key driving factors & challenges in the global API market Emerging product trends & market opportunities Impact of COVID-19 on the global API industry What does 2022 hold for the API Market? -
News CPHI & P-MEC China 2020 adapts to customer needs with hybrid pharma event
Virtual Expo Connect to empower access for international pharma to the world’s largest pharma ingredients market -
Video China: Evolving Dynamics of the CDMO Landscape
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 An overview of global CDMO markets – positioning, strengths and weaknesses of the Chinese CDMO industry. The Evolution of the Chinese CDMO industry, from CMO to CDMO, from starting materials, intermediates to API, drug substance to drug products, generic drugs to NCE/NME – what innovations are on the horizon? The challenges and opportunities of the Chinese CDMO industry – Growth dynamics, the competitive landscape and regulatory barriers. Building a successful CDMO amidst fierce competition, what are the critical attributes of a successful CMC team? How to strategically build technology platforms and capabilities which meet global compliance standards, what are the key requirements for facilities and systems? Langhua Pharma’s own strategy towards becoming a leading CDMO in China, what does the future look like? -
Video Titanium Dioxide – will food ingredient concerns spill over into pharma sector?
In Europe, questions have arisen about the use of nanoparticle titanium dioxide (TiO2) which has led to concern regarding TiO2 as an ingredient in foods, additives, and — by extension — as a color in film coating formulations for vitamin and dietary supplements, and potentially, pharmaceutical products. In this podcast, David Schoneker, President of Black Diamond Regulatory Consulting and Vice Chair for Science and Regulatory Policy, IPEC will discuss the current status and assess the potential market impact for the pharmaceutical industry. The discussion reviews the current legal situation, the position of the different regulatory bodies, the science-based facts influencing the industry response and provides insight into the possible implications on the use of TiO2 in pharmaceutical products, as well as food supplements. This podcast provides listeners with a clear understanding of the situation relating to the use of TiO2, based on facts and science. Key Learning Objectives: What’s all the noise about TiO2, a widely used material across the food and pharmaceutical industries? Navigating through the current market uncertainty surrounding use of TiO2 Gaining clarity and understand viable alternatives to meet film coating needs This session was originally broadcast live as part of CPHI North America 2021 -
Video M&A Climate & Current Market Dynamics
With over $1 trillion in M&A deals in the previous eight years, this trend in activity shows no signs of slowing with private equity firms capitalizing on the highly fragmented CDMO space and a predicted surge in activity in the biologics and cell & gene as more treatments are approved. This webinar will address the benefits of private equity ownership, in triggering new growth, enabling greater strategic focus, increasing prioritization on M&A and better utilization of internal funds for organic growth initiatives. Discussion points: Update on current M&A landscape How to optimally position a company for first-time external investment Life under PE: strategic support to the company’s management team, The investor view: financing organic growth This session was originally broadcast live as part of CPHI North America 2021 -
Video Innovation in the Race to Develop COVID-19 Vaccines
Vaccines are considered to be one of the greatest public health achievements of the last century, so when the COVID-19 pandemic brought the world to a stop, all energies were focused on bringing a vaccine to market. With multiple vaccines now approved and being manufactured in record time, we celebrate this accelerated pace of development in the field. Exploring the different vaccine technologies to understand their mode of action, benefits, risks, and potential real-life impact on protection. The challenges in manufacturing enough doses to vaccinate the world and the potential impact the WTO’s IP waiver proposal will have on future innovation. Discussion Points: Vaccine Science: Two Centuries of Continuous Research, Improvements, and Achievements Future Potential of mRNA Where are the facilities equipped for these new molecule types? What problems are arising and how is the industry working to fix them? Vaccinating the world, a look at WTO’s proposed IP waiver and what it could mean for future innovation This session was originally broadcast live as part of CPHI North America 2021 -
Video Carving Out a Career in Pharmacovigilance
While pharmacovigilance was originally used to describe the drug safety monitoring system in place once pharmaceutical products had made it to market, the definition has expanded somewhat to reflect not only its role as a monitor of changing risks and benefits in both post- and pre-marketing but also that of its use as an interventionist risk management tool. This expanded field has given rise to a breadth of career opportunities for scientists interested in joining it. In this podcast, Jan Petracek, Director at the Institute of Pharmacovigilance, talks about his work at the International Society of Pharmacovigilance (ISoP), where he is leading a Special Interest Group to develop a global Career Framework for Pharmacovigilance Professionals. This session was originally broadcast live as part of CPHI North America 2021 -
Video CDMO Deal Making in a Pandemic and Beyond
This panel discussion is led by Global Business Reports, alongside contributors to their 2021 US Biopharmaceuticals Report. With all eyes on science and pharma for solutions and treatments for COVID-19, 2020 was a colossal year for the industry. Thanks to unprecedented levels of collaboration, vaccines and therapeutics have been approved at record speeds. Amidst a global pandemic, CDMOs were active in making acquisitions and investments. Our expert panellists discuss those deals, the challenges closing them and reflect on their relation to some of the broader themes and future trends for the US Pharmaceutical market. In Partnership with GBR This session was originally broadcast live as part of CPHI North America 2021 -
Video How Diversity of Leadership can Incite Change for Patients from all Backgrounds
This panel session will discuss the importance of intersectional diversity across all levels of leadership in life sciences, to help ensure that under-served patients groups are fairly represented in all areas, from drug development to employee healthcare. Discussion Points: Lack of diversity in clinical stages The correlation of the effects of racism, poverty, and social instability of health and health outcomes. The employer's role in healthcare quality and health care costs and the future of healthcare plans post COVID-19 This session was originally broadcast live as part of CPHI North America 2021 -
Video What role will Pharma play in the Healthcare System of 2022?
In this Fireside chat with Dr David Nash of Jefferson College of Population Health, we discuss the inequities of the current US healthcare system highlighted by the pandemic and the role pharma plays in the future of population health. Discussion Points How might Pharma get ahead of the price transparency curve by promoting public availability of prices? What role can Pharma play in improving some of the SDOH that limit our ability to improve the health of the population? How can we really harness patient-centred outcomes and Real World Evidence so that patient committees will actually pay attention? In the healthcare system of the future, what expanded role might Pharma play to actually promote health? This session was originally broadcast live as part of CPHI North America 2021 -
Video The Women in Leadership Forum: Navigating the Road to Investment for Female Founders
The Women in Leadership Forum is a place for women across the pharma supply chain to come together to connect, network and share ideas for successfully working and succeeding in the industry. This year attendees will hear inspirational stories from our panel and learn about various methods and routes to securing funding for female founders in pharma. -
Video The Real Cost of Bringing Manufacturing Home
We apologise for the background music for the first two minutes of broadcast. Many years before the pandemic the industry has highlighted the importance of a globalized supply chain and in particular the heavy reliance on Asian suppliers. The situation fueled calls from political leaders and lawmakers for U.S. manufacturers to start producing critical supplies domestically. But what will be the cost of bringing manufacturing home, and who will the burden lie on? Discussion Points: What will the “Made in America” pledge deliver? Can AI/Digitalization of facilities reduce costs? Addressing the skills and facilities gap Near-shoring – reducing dependence on Asian suppliers. -
Video Building Supply Chain Resiliency through Responsible Sourcing
When the world went into lockdown in early 2020, manufacturing of critical ingredients came to a standstill, and companies were forced to re-evaluate their supply chains. Forward to 2021, and companies are still dealing with port closures and shipments of expensive ingredients sat in a backlog, many are faced with the decision of either waiting weeks for deliveries or the faster, yet more expensive domestic production to ensure the continuity of supply of essential medicines. Discussion Points: Creating a geographically robust supply chain is critical for reducing drug shortages, Ensuring essential medicines security through public-private partnerships Risk management and the pivot to regional manufacturing hubs In Partnership with PSCI This session was originally broadcast live as part of CPHI North America 2021 -
Video Biosimilar Waves - Future of Biosimilar Approvals and Development.
Discussion Points: Patent expiry opportunities 2021-2026 Catching up with Europe This session was originally broadcast live as part of CPHI North America 2021 -
Video De-risking Drug Development with Digital
Moving away from traditional, costly, time-consuming and often unsuccessful R&D methods, innovative companies are looking to AI and machine learning to reduce costs and improve success rates in drug development. This session will explore the different types of AI currently being used by Pharma companies, projects that identify novel pathways, targets and biomarkers and discuss the challenges of biometric data security, and how companies can work alongside existing regulations. Discussion Points What are the different types of Artificial Intelligence and use cases? A look at current regulations and how to make them work for you Federated learning in AI development How biocatalysts are reshaping the synthesis of chemical entities. The power of 3D molecular modeling In Partnership with AAPS This session was originally broadcast live as part of CPHI North America 2021 -
Video CPHI MEA: The Manufacturing Roadmap for Africa
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 Africa is an exciting emerging market with significant investment potential. While pharmaceuticals are currently manufactured across South Africa, Kenya, Nigeria, Algeria, Morocco and Egypt, the continent still relies on imports for over 70% of pharmaceutical and medical consumables. Discussion points: Current outlook for African pharma manufacturing What are the obstacles to growth? How can the right infrastructure for domestic manufacturing be nurtured? Facilitating R&D and innovation -
Video Drug Delivery Start-Ups to Watch in 2021 and Beyond
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 As the pharmaceutical industry keeps on growing, and medical technology keeps on advancing, drug delivery devices are increasingly facing challenges to keep up to date and adapt accordingly. Drug delivery device manufacturers are not only increasingly looking to other industries but also to start-ups to identify novel technologies and new innovative products that are not only patient-friendly and cost-effective but also innovative and functional. Considering the growth of market demand and technological advancements. It is an exciting time to be a start-up working in the drug delivery landscape. This session will feature start-ups providing novel technologies and innovative solutions to current challenges in the drug delivery technology landscape. And it will tackle some of the main trends and challenges ahead for this industry. Emerging start-ups on the Drug delivery landscape The role of Drug Delivery tech start-ups in improving patient access and driving innovation A look into the future of drug delivery: regulation, capital raising, and investments -
Video Track & Trace Solutions for Direct-to-Patient Delivery
This session was originally broadcast as part of the CPHI Webinar Series and aired during CPHI Discover - 17-28 May 2021 Building a comprehensive track-and-trace system for medicines, which is aligned with the European Falsified Medicines Directive and to ensure compliance is a must in today’s environment. Pharmacies, hospitals, HCPs, caregivers, and patients now expect a completely secure and traceable delivery process, which requires buy-in from pharmaceutical companies and distributors involved in the supply chain of Investigational Medicinal Products (IMP), SKU’s and unique doses. This webinar provides an overview of the key trends and developments to improve the security of drug delivery direct-2-patient in 2021. Key discussion points: A global overview of regulations on traceability of medical devices and medicines Technical challenges when tracking medicinal products along the product life cycle, from clinical trials to post-marketing authorization What does the future look like? The potential of delivering Direct-to-Patient unique doses -
Video Global Vitamin Market Trends
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 Vitamin markets have seen growth spikes in the past year, both globally and in China - the largest manufacturing region for vitamin products.Sales of conventional products such as Vitamin C and D have risen in the midst of the COVID-19 pandemic, as consumers look to boost immunity. This session will address key trends and developments in this sector in the past year, and examine new products such as CoQ10 and NMN, which are creating fresh market opportunities. This webinar will comprise of two presentations: China Vitamins Market: what’s new and what’s to come? Ellie Adams, Managing Director, QIVA CoQ10 Market in Japan and New Water-Soluble Product Trend Yukihiro Miyazaki, Managing Director, Jupiter Bioscience Japan -
Video API Manufacturing Trend Outlook
This webinar originally aired as part of the CPHI Webinar Series and repurposed for CPHI Discover - 17-28 May 2021 The global market for the outlook and manufacture of APIs is shifting as more complex or potent ingredients evolve which present specific handling and manufacturing challenges. What are the latest technological approaches for these products, and how can quality, safety and cost be managed and improved? A key impact of the COVID-19 pandemic has been to question the reliance and dependence on Asia as the global leader of API manufacturing. Do we expect to see specific regions implement reshoring of manufacturing efforts? How will global supply chains be impacted? Market Outlook for APIs Can we expect a reshoring of API manufacturing? Will hubs shift from Asia? Continuous API manufacturing –for cost, quality and safety benefits Manufacturing considerations for complex formulations and high potency APIs, what are the latest approaches? This session was originally broadcast as part of the CPHI Webinar Series. -
Video Driving Digital Transformation in Pharma
This session originally aired as part of the CPHI Webinar Series and was repurposed for CPHI Discover - 17-28 May 2021 Increased adoption of digital technologies can offer a multitude of benefits for pharma companies –from data optimization and analysis to increased productivity in R&D, to sourcing candidates for clinical trials. How can pharma companies take a more active approach to implement digital innovation, to help cope with new challenges in the healthcare industry? One approach to speeding up adoption is to engage with start-ups and established technology companies who can bring different expertise and a more agile approach. This webinar will look explore the relationship between Pharma companies and start-ups, as the key to unlocking digital transformation. What can digitalization do for Pharma companies? What are the benefits? Has the Covid-19 pandemic accelerated digital innovation in the pharma industry? Partnering for success –how engaging with start-ups and tech companies can help acquire expertise and accelerate development A review of successful solutions, initiatives and Start-Ups helping to solve healthcare challenges What does the future look like? Can we expect to see increased digital transformation? -
Video Formulating Plant-Based Ingredients for Cosmeceuticals
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 The Cosmeceuticals market is projected to grow 8.9% in the period 2020-2026, driven primarily by conscious consumers who are looking for increased potency in products which offer better efficacy than standard OTC solutions. Beyond this, consumers are also increasingly concerned about potentially harmful chemicals in cosmetics, leaning instead towards natural and plant-based solutions. This session will address cosmeceutical market dynamics and identify opportunities for growth. Discussion points: What are the key growth drivers for the Cosmeceuticals market? Formulating plant-based ingredients - challenges and opportunities Responding to the demand for plant-based ingredients Regulatory considerations for natural ingredients and cosmetics Developing sustainable solutions to differentiate your business for the conscious consumer -
Video Biomanufacturing Trends
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 Innovative biotherapeutics and novel technologies are currently being developed and commercialised at record levels, providing practical treatments for patients with cancers and autoimmune diseases across the world. In 2019 the FDA approved 48 novel drugs, several of which represent advanced, first-in-class therapies. It comes as no surprise then to hear that venture capital funding into US-based biotech firms was $3.5 billion+ for each of the last eight quarters leading into 2020, according to Pitchbook data. Today, in the fight against the novel COVID-19 virus all eyes are on the global scientific community who have come together to work on and produce various treatments and vaccines at record speed, and we are reminded just how important scientific innovation is. Join us for the webinar Trends in Biomanufacturing and hear from industry experts to discuss the following: Product pipeline - what is the latest overview? Which products post key manufacturing challenges? Securing and sustaining the right talent pool to support growth Innovation in manufacturing techniques - bioanalytical capabilities, cell line development, aseptic manufacturing, downstream/upstream Continuous biomanufacturing - significant support for this in the small molecule space, what -
Video Economics and Ecology: The Impact of Water Reduction in Biotechnology
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 Throughout the last decade, the importance of sustainable manufacturing increased rapidly in the biopharmaceutical industry.The process mass intensity (PMI) is a metric, which mainly displays the water consumption in a process. Based on continuous antibody production, we analyzed the correlation between the reduced cost of goods and a reduced PMI to align economic and ecological benefits. -
Video Increasing efficiencies in bioprocess development and manufacturing through digital process development
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 The application of modelling tools to better understand and control bioprocess development and manufacturing became a hot topic within the last few years. But what do we actually need? Is it just about data and powerful modelling algorithms or might a shift in focus towards “why” rather than “how”, speed things further up? Discussion points: The overall goal Experimental design concepts Different modelling approaches for different requirements Solutions to speed up both up- and downstream development Model transfer along scales Model usage for real time monitoring and control -
Video Beyond Brexit: The Future of British Pharma
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 The United Kingdom left the European Union on December 31st, 2020. Before this, the EMA had already relocated its European HQ from London to Amsterdam, and there was considerable uncertainty on how the European Pharma Market would be impacted. This session will provide an analysis of the challenges presented by Brexit, and the impact on British Pharma. What is the impact of the UK leaving the EU on Pharma to date? What has changed? Security of supply for medical and pharmaceutical products Recent policy discussions on increasing the resilience of global supply chains How are manufacturers going to ensure continuity of supply in a post-Brexit world? How will innovation be impacted? What are the long-term implications of Brexit? -
Video Application of advanced proteomic and mass spectrometry technologies to enhance the efficacy of production of biopharmaceuticals
This webinar originally aired as part of CPHI Discover - 17-28 May 2021 Proteomics and mass spectrometry are key technology to the understanding of biological systems. We are using proteomics to characterise recombinant mammalian cell lines (Chinese hamster ovary) in order to gain insights into improving the productivity of biopharmaceuticals. Discussion points: Overview of advanced proteomic and mass spectrometry technologies How they can be utilised to enhance the efficacy of production of biotherapeutics Potential application of proteomics and mass spectrometry in other industries to enhance research capabilities and to potentially improve production capabilities -
Video CPHI Webinar Series: Latest Approaches for Large Volume Drug Delivery
Innovation in the design and development of large volume drug delivery devices can solve the challenge of accommodating the increasing trend towards patient self-administration and home care while at the same time ensuring that drugs that require intravenous administration are delivered correctly and safely. This was just one of the messages that came out of a recent CPHI Webinar, Latest Approaches for Large Volume Drug Delivery, sponsored by Cambridge Design Partnership. -
Video The Evolution of Pharma R&D Models
This webinar discusses the evolution of R&D models, the outsourcing landscape and how to de-risk your development and maximise ROI. -
Video Pharmapack Webinar – Track & Trace: Challenges and opportunities for direct-to-patient delivery: Watch Now
This recently recorded CPHI Webinar in association with Pharmapack delves into the vitally important world of traceability of medicines and what the packaging industry is doing to address the issues of medication errors, security and drug wastage. Click the link above to access the on-demand session.
Providing a regulatory outlook, Nathalie Wardé, Chief Executive Officer, D4P pharma, said that with 50% of medication errors coming from dispensing and administration, improving traceability of medicines for direct-to-patient delivery is a must.
“Traceability is a global trend in healthcare B2B,” she said “Over the last seven to ten years, it has started to be a real concern for all the health authorities mainly to fight against counterfeit (or ‘falsified’) medicines or medical devices and to facilitate the recall process.”
Arguing the case for why global standards are needed across healthcare and providing an introduction to GS1 and Gs2 standards, Tania Snioch, Director Healthcare Operations, GS1 Global Office said that “never has a global, secure and efficient supply chain been more important than today,” citing the current pandemic situation.
“We’ve learned in these difficult times that global standards have a role to play; we’re all looking for an efficient supply chain that operates across countries and across continents and to have that, we need to be able to talk to each other in a language everybody understands,” she added. “ We need to be sure we’re talking about exactly the same products and it needs to be unambiguous; a global standard is the way to achieve that.”
Providing insights into the management of clinical supply chains, Pierre Fernandez-Barbereau, R&D Clinical Supply Chain Operations, Industrial Development, Sanofi, said that two key objectives are to ensure alignment between clinical demand and to design a clinical package in order to protect the dose and ensure the ‘blinding’ of the medicinal product.
“But the third objective, which is also the most important one, is to package on the production line and distribute throughout the world to clinical sites and patients, ensuring quality and traceability of these medicinal products,” he said. -
Podcast CPHI Podcast Series: Sustainable Strategies in the Pharmaceutical Packaging Industry
Sustainability in pharma is now gaining the recognition it deserves, quickly becoming one of the biggest priorities for pharma companies across the spectrum. Upgrading this Packaging in pharma comes in all shapes and sizes – whether we are talking about replacing single use plastics in manufacturing, or waste management at the end of the commercial chain. Today we will be talking about sustainability in the pharmaceutical packaging industry, how it can be more ingrained and the impact it will have, with an expert from a leading European packaging company, Adelphe. Félicie Pachot is the Health Marketing Manager at Adelphe and she goes into the model Adelphe uses to support sustainable practices through recycling, reduction of waste, and reuse. -
Podcast CPHI Podcast Series: The importance of Pharma 4.0 to innovation leads in India
In this latest episode from the CPHI Podcast Series, Digital Editor Lucy Chard speaks to Sanjay Sharma, Global Head of Manufacturing, from Dr Reddy’s. Sanjay has over 26 years of experience in roles encompassing sales, the supply chain, and technical operations. Pharma 4.0 is the upgrading of the pharma industry to a more digitalised state, including automation of processes and greater system integration. This has a wealth of advantages, such as leading to greater compliance and quality control in the manufacturing process. Pharma 4.0 also leads to increasing the sustainability of many aspects of the pharmaceutical chain. Dr Reddy’s is a leading pharmaceutical company and is at the forefront of adopting industry 4.0 into their everyday systems. Dr Reddy’s largest manufacturing facility in Bachupally, Hyderabad, India, recently joined the Global Lighthouse Network of the World Economic Forum, a network of over 100 manufacturing companies committed to developing and leading the progression into Industry 4.0 technologies. This commitment from Dr Reddy’s will help to reduce manufacturing costs, production lead time, and energy consumption, as well as increasing the quality assurance throughout the manufacturing process. -
Podcast CPHI Podcast Series: Orally Dissolving Tablets
In this month’s CPHI podcast, sponsored by Galien Pharma, we focus on the technology behind orally dissolving tablets. The orally dissolving tablet is a solid dosage form that is sometimes viewed as an unsung hero compared to other patient-centric drug delivery systems. ODTs are becoming increasingly important in the pharmaceutical market for both prescription and OTC medications not least because they can significantly improve patient compliance. However, there are various challenges in the formulation process and there is still much room in the space for manufacturing technology innovation. To find out more, tune into this month's podcast with our guest Franklin Delaye, Strategic & Transformation Director at Galien Pharma. -
Podcast CPHI Podcast Series: Driving innovation with pharmaceutical startups
The pharmaceutical industry is brimming with innovation – from biologics such as cell and gene therapies to COVID-19 treatments and precision oncology drugs. And as pharmaceuticals grow more advanced, so too must the drug delivery and packaging solutions that get them safely from the manufacturer to the patient. This is a competitive arena and among all the companies operating in this space, startups are generally seen as the drivers of innovation, often taking high-risk approaches and doing business with greater agility. In this episode, CPHI is joined by Gareth Pearce, founder of Pacifi Ltd, a startup that modifies glass vials with laser technology to create DuoVIAL, an innovative lyophilized drug delivery solution. Gareth takes us through his company’s journey, explains his long-term vision and speaks more broadly about the role events can play in a startup’s path to success. Listen to the full episode now. -
Podcast CPHI Podcast Series: Identifying and monitoring animal health and diseases to prevent the next pandemic
An outbreak of zoonotic disease can have devastating economic and social consequences, as evidenced by the COVID-19 pandemic and increasing concern over monkeypox. Defined as a disease that has transitioned from a vertebrate animal to humans, zoonoses account for about 60% of known infectious diseases and 75% of new or emerging infectious diseases. So, what can be done to limit the spread of these diseases and prevent the next pandemic? In the latest episode in the CPHI Podcast Series, we hear from two companies – Chipsafer and SoundTalks - whose products use data analytics and AI to identify diseases in animals at the earliest indication. Chipsafer is a patented platform that detects and tracks anomalies in livestock behaviour, giving farmers earlier warning when disease breaks out in the herd. Joining us to break down how the platform works is Chipsafer’s founder and CEO, Victoria Alonsoperez. Similarly, SoundTalks is a company that provides real-time health monitoring in pig herds, using microphones and sound analytics to detect possible outbreaks of respiratory disease. SoundTalks’ founder Dries Berckmans joins us to discuss. Listen to the full episode now. -
Podcast CPHI Podcast Series: The importance of novel excipients for innovative drug development
Excipients are essential to the formulation of drug products, but their importance in supporting innovative drug development is often underestimated. According to the International Pharmaceutical Excipients Council (IPEC), drugs that have yet to be developed may need more excipients than are currently available to us, and many new treatments are unlikely to succeed without novel excipients. In short – innovation in drug development requires innovation in excipient manufacturing.
However, the lack of an independent regulatory pathway for novel excipients has historically created barriers to their use and discouraged innovation in the excipients space. In this episode Nigel Langley, Chair of IPEC – Americas and Global Technology Director at BASF Corporation, joins us to discuss developments in the novel excipients arena, as well as the pilot review program launched by the FDA last year. -
Podcast CPHI Podcast Series: The Supplements Market – From Slump to Surge
The supplements market is on the rise, with products like vitamins, herbal drinks, and protein powders found in more household cupboards than ever before.
While supplements can be an excellent way to cover deficiencies not addressed by food intake, the wide range of products available in health food shops, pharmacies and supermarkets can be overwhelming for consumers. And while market growth is expected to continue, products such as herbal supplements and CBD continue to face regulatory issues and scrutiny over the soundness of their health claims.
To discuss the latest trends in the supplements space, we’re joined this month by David Ridley, Senior Editor at HBW Insight and host of Over the Counter podcast. He tells us which products surged in popularity during the pandemic, discusses the rise of self-care, and gives an overview of the regulatory environment in Europe. -
Podcast Pharmapack Podcast Series: Senior patient-friendly drug device design - Part Three
Partnering with one of northern Europe’s oldest academic universities, Lund University in Sweden, Pharmapack has launched this three-part podcast series, delivering the latest insights on medication packaging innovation and the gathering momentum towards patient‐centric design. In the third and final episode of this three-part series, we talk to Dr Alex Cole, Head of Strategic Marketing, CPI, and Giana Carli Lorenzini, postdoctoral research fellow, Packaging Logistics at Lund University, about how companies should approach the design of senior-friendly self-administered drug devices by putting the patient and their physical and cognitive limitations at the heart of their programmes, and also how to keep older patients involved and engaged at every phase of the development process. -
Podcast CPHI Podcast Series: Pharma 4.0 and the Digitalisation of Manufacturing
In this episode we delve into the issue of Pharma 4.0 and what steps the industry is taking to transition pharma manufacturing to a much more digitalised model. The pharma industry is constantly seeking new ways of streamlining its manufacturing processes and digitalisation lies at the core of this transition. It is seen as the springboard to a brave new world of pharma manufacturing, driven by smart facilities of the future, big data, artificial intelligence, data integrity, and end-to end integration to name but a few choice descriptors. We speak to Thomas Halfmann (below), Global Head Production IT, NNIT, who sheds light on the numerous opportunities for manufacturing presented by Pharma 4.0 and what impact the current pandemic has had on the digital transition. -
Podcast CPHI Podcast Series: Decentralised Clinical Trial Design - Collecting Real World Data in a Remote Setting
In this podcast interview with Fabien Du Fraisse and Deepu Joseph at Quanticate, we delve into the trend of decentralising clinical trials Not only does the transfer of clinical trials more and more away from investigator sites and into volunteers’ homes offer a more patient-centric approach and reduce the burden on caregivers, it also offers up great opportunities for the industry to capture more data. And as the world adjusts to the reality of a prolonged global pandemic and the difficulties of asking volunteers to make physical visits to sites, never has decentralising clinical trials become such a pertinent issue. -
Podcast Special Podcast: CPHI Outlook Roundtable - Listen Now
In this special roundtable podcast exploring potential opportunities in a post-vaccinated world, we are joined by a panel of three world class experts – Bikash Chatterjee, CEO of Pharmatech Associates; Parrish Galliher, Managing Director, BioProcess Technology Group, a division of BDO USA and Valdas Jurkauskas, PhD, VP Technical Operations at Black Diamond Therapeutics. The panel discusses insights into what new discoveries and technologies might offer the best potential, the approvals we might see in the next few years, and what might be the long-term positive COVID legacies – from speed of innovation and clinical trials, to proprietary information exchange and the global supply chain. To download a copy of the CPHI Post Pandemic Legacy Trends Report, click here: https://www.cphi-online.com/this-special-cphi-report-is-split-into-two-halves-file113613.html -
Podcast Pharmapack Podcast Series: Patient-Centric Packaging Design & Innovation - Part Two
Partnering with one of northern Europe’s oldest academic universities, Lund University in Sweden, Pharmapack has launched this three-part podcast series, delivering the latest insights on medication packaging innovation and the gathering momentum towards patient‐centric design. Podcast Two: How do you apply patient-centric design to clinical packaging? In the second of this exclusive three-part series, we talk to Sascha Sonnenberg, Global Head Business Development, Sharp Clinical Services, and Giana Carli Lorenzini, postdoctoral research fellow, Packaging Logistics at Lund University, who discuss the challenges of introducing better functionality and usability to clinical packaging amid tight constraints and how the latest innovations in this area are driving patient adherence. -
Podcast CPHI Podcast Series: Pharmaceuticals in the Environment and Antimicrobial Resistance
Pharmaceuticals discarded in the environment pose a risk to fish and other wildlife, for example by affecting their ability to reproduce, by altering their behaviour in ways jeopardising their survival, or through direct toxic effects. Increased awareness has prompted further investigation, as well as calls and proposals for action to reduce emissions to the environment, in particular to water but also to soil. Incorrectly disposed medicines may also contribute to the serious problem of antimicrobial resistance (AMR). In this podcast we talk to Dan Caldwell, PhD, Toxicology Fellow, Johnson & Johnson and Steve Brooks, Advisor to the AMR Industry Alliance about what progress the industry is making in terms of addressing the multifaceted challenges that the release of pharmaceuticals poses to the environment, looking at all key points within the pharmaceutical lifecycle. -
Podcast Podcast: CPHI Discover – A look ahead to three days of insightful content: Listen Now!
Our online pharma event CPHI Discover is almost upon us! While we’ll be delving into the typical themes of ingredients, manufacturing, supply chain logistics and technology, we’re also going to be examining some newer trends which are impacting on pharma manufacturing and distribution – such as highly politicized issues such as the change of administration in the US and Brexit and of course, the sector’s drive towards sustainability. In this podcast, Informa Markets’ Head of Content, Tara Dougal and Pharma Editor, Gareth Carpenter run the rule over a packed three-day agenda running from 18-20 May of webinars, podcasts and learning labs. -
Podcast CPHI Podcast Series: M&A strategies in the CDMO sector
This month we discuss what is driving M&A strategies in the pharma space, paying particular attention to how investors regard the potential for the growing but fragmented CDMO sector The contract development and manufacturing organisation (CDMO) market is projected to grow to just short of $158 billion by 2025 as a CAGR of 6.9%, amid increasing appetite from biopharma and pharma companies to outsource their manufacturing. In many cases, M&A gives companies the opportunity to expand their reach beyond certain geographies or beef up their service offerings with additional capabilities in the development and manufacturing chain. In this month’s podcast, we speak to Kevin Bottomley, partner at global corporate advisory firm focused on healthcare and life science companies, Results Healthcare, about what is driving M&A strategies in the pharma space and whether investors see high potential for the highly fragmented CDMO sector. -
Podcast Pharmapack Podcast: Patient Partnerships for Medical Devices
Healthcare provision is constantly evolving. Whereas in the past, decision making on treatments for patients was very much the domain of physicians and doctors, patients now feel more empowered to be involved in the decision making process. The healthcare industry is fully aware of this change and sees a constructive partnership and dialogue with patients as the best way of achieving better health outcomes. In this podcast, we discuss with Nicholas Brooke, founder and executive director of The Synergist and founder of Patient Focused Medicines Development how the concept of patient engagement has developed and what tools are becoming available for meaningful patient engagement in the drug and medical devices development process. -
Podcast CPHI Podcast Series: The Cannabinoid Opportunity for Cosmetics & Pharma
Cannabinoids have caught the attention of Pharma and Cosmetic companies alike in recent years, celebrated for their anti-inflammatory, anti-oxidant and anti-bacterial properties in cosmetic products, and neurological and chronic pain benefits on the pharma side. There is also rising consumer demand for cannabinoids, exacerbated by a shift towards ‘natural’ and toxin free products, which present an interesting opportunity for ingredient manufacturers and producers. In this podcast we speak to Dr. Monica Vialpando, founder and CEO of Via Innovations about the promising applications of cannabinoids, the innovative approaches to product development and formulation, and examining the regulatory and legal considerations of bringing these products to market and whether there is an opportunity for pharma companies to diversify into the cosmetics market. Within this discussion, Dr. Vialpando: 1) Describes what cannabinoids are and why pharma and cosmetics companies are currently investing heavily in their research 2) Shares some key learnings from her transition from a pharmaceutical background into the cannabis industry busts some myths about the origins of cannabinoids 3) Highlights key areas in pharmaceuticals and cosmetics where cannabinoid research is most likely to reap dividends explains where the current research focus on cannabinoids is and how can this be applied to the pharmaceutical and cosmetics industries 4) Provides advice on the key considerations you have to make when designing and branding cannabinoid/CBD-based products -
Podcast CPHI Podcast: The distinction of drugs vs cosmetics from a regulatory perspective
As more and more companies operating in the pharmaceutical space are moving into cosmetics, the temptation is to think that life will be much easier from a regulatory and compliance perspective. However, while it is true that pharmaceuticals are more heavily regulated, this does not mean that cosmetics developers can rest on their laurels. The subtle lines between making marketing claims about cosmetics that are deemed allowable and those crossing the line into drug products can often be blurred. The wrong claims can cause a cosmetic product to be misbranded attracting unwanted attention from the US Food and Drug Administration. In this podcast with John Bailey and Catherine Bailey from EAS Consulting, we take a look at the various common compliance issues that can arise when jumping the boundary from drugs to cosmetics. -
Podcast CPHI Podcast Series: The opportunities and challenges of providing fill and finish services for mRNA platforms
mRNA (Messenger ribonucleuc acid) technology has been thrust into the limelight in recent months, with the platform proving to be the springboard for two approved vaccines against the SARS-CoV-2 virus. The complex science around mRNA is very different from that of small molecule traditional pharmaceuticals or traditional biologics such as monoclonal antibodies. This month we speak to Christy Eatmon, Global SME, Sterile Drug Products, Pharma Services Group at Thermo Fisher Scientific about how CDMOs are providing their manufacturing and fill finish expertise to bring mRNA vaccines to market in as timely a fashion as possible. -
Podcast CPHI Podcast Series: Annual Report 2021 Roundtable
In this special edition podcast, Informa Markets pharma editor Gareth Carpenter speaks to Bikash Chatterjee, CEO at Pharmatech Associates and Dan Stanton, Editor and Founder at BioProcess International about some of the findings in the recently published CPHI Annual Report 2021, including latest innovations in continuous manufacturing and what is driving growth in the mammalian biomanufacturing sector. -
Podcast CPHI Podcast Series: Securing the Biopharma Workforce of the Future
The biopharmaceutical industry has seen incredible growth in the past decade, and shows no sign of slowing as the need for specialised treatments in oncology, immunology and vaccines continue to grow. To support innovation, the biopharma industry needs a suitably skilled workforce, and therein lies one significant challenge facing the market at present. Emerging technologies and new capabilities require a specialized skill-set, and finding the right candidates in a competitive market can be complex. In this month's episode, CPHI speaks to Killian O'Driscoll, Director of Projects at the National Institute of Bioprocessing Research & Training (NIBRT) on some of the key challenges in the biopharma landscape, and why finding the right talent is such a priority for the sector. Listen now to learn more about the biopharma skills-gap, and what can be done to ensure innovation continues at pace. -
Podcast CPHI Podcast Series: Connecting the user to your product – Smart packaging for smart devices
Smart devices are revolutionising how we live our lives and changing the shape of the industries adopting them. One such industry is healthcare, where the benefits of context-aware devices and real-time data sharing are having tangible effects on patient outcomes. Many smart devices, like wearable monitors, allow doctors to oversee a patient’s health status remotely, facilitating better decision-making and shifting healthcare management from hospitals to homes. But with such rapid technological advancement comes a new challenge: user adoption. In this month’s episode, CPHI speaks to Alex Driver, Senior Industrial Design Consultant at Team Consulting, about breaking down the barriers to smart device access. He shares some of the pain points faced by new smart device owners and outlines strategies for making these accessible to even the most digitally-averse users. We discuss the importance of packaging, the “unboxing” effect and how intelligent design can induce behaviour change. Listen now to learn more about the about the rise of smart devices, and the strategies facilitating onboarding and adoption. -
Podcast CPHI Podcast Series: Success in healthcare M&A, taking a people-first approach
Many companies are turning to M&A to optimise their portfolios and gain access to new technologies, fuelled by an abundance of capital in the sector. These deals can have transformative effects on the companies involved; ideally promoting synergy, diversifying product offerings and improving economies of scale. -
Podcast Podcast: Hot Melt Extrusion- Formulation Approaches and Technologies
This podcast originally aired as part of CPHI Discover - 17-28 May 2021 While hot melt extrusion was developed in the 1930s and initially used in the plastics and food sector, the pharmaceutical industry has now firmly embraced this technology platform in its manufacturing as a way of solving formulation challenges, particularly those around solubility enhancement. Advantages of the approach include the ability to use a wide range of excipients and the ease with which process analytical technology can be incorporated. In this podcast, we explore how HME has developed as a proven technology for bioavailability enhancement of poorly soluble active pharmaceutical ingredients, and how it stacks up against other formulation options in terms of cost and scalability in order to deliver high quality, commercialised pharmaceutical products. -
Podcast Podcast: Spray Drying- Formulation Approaches and Technologies
This podcast originally aired as part of CPHI Discover - 17-28 May 2021 Spray drying is used in various pharmaceutical applications and has become an increasingly popular technique in the production of formulations of poorly soluble APIs and inhalable dry powder formulations. This remarkable technology involves spraying a liquid formulation onto a surface which is then allowed to dry to produce a powder in one single step. In pharmaceutical manufacturing, spray drying is suited to the production of peptides, proteins or poorly water-soluble APIs, particularly antibiotics, because it allows them to be co-processed with solubility enhancement excipients. Another advantage is that the approach reduces the number of unit operations, potentially lowering manufacturing costs. This podcast explores how spray drying can overcome the problem of low aqueous solubility that all too often stall clinical testing programmes. -
Podcast Podcast: Co-Crystals - Formulation Approaches and Technologies
This podcast originally aired as part of CPHI Discover - 17-28 May 2021 Co-crystals are crystalline materials composed of two or more different molecules, typically active pharmaceutical ingredients and co-crystal formers in the same crystal lattice. Tailoring of co-crystals can provide opportunities to not only enhance bioavailability and stability but also the processability of APIs during drug manufacturing. In 2018, the US Food and Drug Administration published guidance clarifying the regulatory classification of pharmaceutical co-crystal solid-state forms for companies planning to submit new drug applications. This move has generated a lot of interest in the co-crystal field. In this podcast, we take a look at how the use of co-crystals have evolved and what are the best approaches to screening and selecting co-crystals to identify the most viable ones to help find suitable development candidates. -
Webinar Panel: Major bottlenecks – Skilled Workforce, Product Quality, Manufacturing Capacity, Cost and Speed to Market
Join our panel discussion, where out experts will conduct a deep dive through the challenges in drug manufacturing. Key discussion points include: Bottlenecks in skilled workforce and what can be done to overcome this Assessing the challenges in both product and manufacturing quality and identifying solutions to this Reviewing the costs and why it’s a driver to the speed to market. This session was broadcast as part of the CPHI North America show. -
Webinar In Pharma Services, Where Have All the Entrepreneurs Gone?
Merger, acquisition, and consolidation activity in CRO/CDMO world has intensified to the point where many of my colleagues are beginning to wonder how big the global players can actually become. While many of the mid-sized players have disappeared, a huge number of small pharma service organizations are thriving; creating a giant divide between the small and large players. This also creates a tremendous opportunity for the small pharma service provider to fill the void that’s been created. Today, these entrepreneurial companies are doing incredible science and providing a highly-tailored, fast, and flexible business model to the pharma and biopharma industry. This presentation will take a close look at key market statistics within the smaller pharma services demographic here in the US, as well as a distillation of our findings from discussions with over 200 business owners over the past 18 months. Key questions to be addressed: What are the keys to your current success? What are the barriers to future growth? How has the pandemic impacted your businesses? What are the key risks and opportunities facing the market? This session was broadcast as part of the CPHI North America show. -
Webinar Connecting the User to your Product – Smart Packaging for Smart Devices
Originally broadcast as part of the CPHI Monthly Podcasts Series Smart devices and their companion apps are transforming the patient experience and moving healthcare management from hospitals to homes. However, this digital shift can prove challenging for some users. In the latest instalment of our podcast series we speak to Alex Driver, Senior Industrial Design Consultant at Team Consulting, about the creative design solutions increasing accessibility to smart devices. How important is packaging when it comes to user perception? What makes for a good unboxing? And how can manufacturers simplify smart device setup? This session was broadcast as part of the Pharmapack show. -
Webinar Sustainable Talent Acquisition: Where has All the Talent Gone?
Global drug sales have now hit $1 Trillion for the first time; the R&D pipeline has doubled in the last 10 years, CDMO market is expanding at a consistent 10% YOY. The pharma industry faces countless opportunities; however, there is a problem. Business leaders are asking where has all the talent gone?
This session will explore real-world insights into why the industry is facing a talent crisis. He will share those critical factors any pharmaceutical business should be thinking about when preparing their organization to sustainably grow, including some practical solutions, no matter what your size, to hire more effectively. -
Webinar CPHI Frankfurt22-Manufacturing Excellence - Panel Discussion
This leadership-powered panel discussion will touch upon some of the actions, challenges and the way ahead for producers of fine chemicals to achieve manufacturing excellence through sustainable competitiveness, growth and the promotion of responsible global environmental performance. -
Webinar Accelerating Innovation and Development of Future Biomanufacturing Processes
BioPhorum has brought together biomanufacturers, supply partners, engineering partners, and regional innovation hubs to build a consensus strategy roadmap, encompassing considerations for all aspects of development from raw material to product, for the future of the industry. The roadmap will be presented alongside recent developments in priority areas agreed by the industry.
These developments facilitate progress for the industry through identifying technology targets and modeling solutions. Current work includes proof of concept demonstrators and business case modeling, plug-and-play hardware, smaller buffer preparation footprints, and driving the move for more standard facility design to accelerate capacity builds. -
Webinar Are local manufacturing and tech transfer still a challenge in the MEA region?
The demand for localization of pharma products in the countries of the MEA region is booming ! Are the know how and the local infrastructures ready? Is there a clear understanding of the processes? What is the best business model and partnership when it comes to tech transfer? Case Study: Reshaping of KSA pharma business & industry from Importation to Localization -
Webinar On-Demand Manufacturing for Medicine Supply Chain Resiliency & Patient-Ready Medicine Delivery
On Demand Pharmaceuticals is creating technology platforms that enable point-of-care medicine manufacturing whether on the battlefield or hospital. By building modular automated systems that can produce raw materials to finished drug product, we are strengthening medicine supply chain resiliency and transforming how patients access medicines. This vertically integrated approach offers agility, flexibility, and a rapid response capability that will mitigate drug shortages while providing quality medicines when patients need them -
Webinar Advanced Digital Customer Segmentation–Elevating Customer Understanding Leveraging AI
For the past few years, the traditional one-size-fits-all go-to-market strategy, predominantly based on physical channels, has failed to support drug launches and meet business objectives. Inadequate or incomplete understanding of customer needs has been identified as one of the top-3 root causes.
To optimise patient outcome, biopharma companies need to target and engage effectively with key stakeholders, differentiating the approach based on distinct behaviours and preferences. Our advanced segmentation approach enables to define data-driven HCP segments by leveraging AI and machine learning algorithms. In particular, we display a detailed use case that describes how we have helped biopharma companies leverage data and AI to build differentiated and actionable HCP segments.
Our recommendations detail next best actions to optimize customer engagement and assess the implications on company’s overall level of push marketing actions. Essentially, it will enable to reach the right customers, through the right channel with the right frequency. -
Webinar What if a New Generation of LBEs Could Lift Advanced Pharmaceutical Drug Development with Poorly Soluble APIs to its Next Level by providing a Built-in Fit-for-Purpose Feature for Advanced Processes such as Hot Melt Extrusion and 3D-Printing?
More than 75 % of new chemical entities are poorly soluble and require bioavailability-enhancing excipients. The application of excipients in advanced pharmaceutical processes, such as hot melt extrusion and 3D-printing, aligned with advanced approaches towards personalized medicine, requires special features. Lipid-based excipients (LBEs) are “natural contenders” for these tasks. However, until now, it has not been possible to fully exploit the potential of LBEs.
The main challenge is the unstable solid state of current LBEs, e.g. polymorphism, leading to instability of the final dosage form. Compared to polymers, the low melting temperature of lipids, combined with highly crystalline arrangement, limits their application in advanced pharmaceutical processes such as hot melt extrusion and 3D-printing. -
Webinar iGAL 2.0: an Accurate Metric for a Greener Future in Drug Manufacturing – a Supply Chain Perspective.
With this session you will learn about best practices in Green Chemistry metrics for evaluating the impact of waste in pharmaceutical drug substance manufacturing, and their limitations, in the context of API lifecycle assessment. You will also be informed about the new iGAL 2.0 metric as a valuable extension to the existing waste metrics, allowing evaluation of relative process greenness and the sustainable innovation impact by scientists. Finally, you will learn about opportunities to effectively integrate the new metric into Pharma-CDMO partnerships for the benefit of their business and the planet.
The presentation is based on these recent publications: Improved iGAL 2.0 Metric Empowers Pharmaceutical Scientists to Make Meaningful Contributions to United Nations Sustainable Development Goal 12 | https://doi.org/10.1021/acssuschemeng.1c01940 A Green Chemistry Continuum for a Robust and Sustainable Active Pharmaceutical Ingredient Supply Chain | https://pubs.acs.org/doi/10.1021/acssuschemeng.9b02842 -
Webinar Sustainability, Quality and Supply Chain Integrity: A New View
Understanding and channeling ourselves to think about the intersection of sustainability, quality, and supply chain integrity within pharmaceuticals. This relationship has become significant as we enter the post pandemic era in which the pharmaceutical industry strives to not only secure its supply chain and material quality, but in parallel ensure that its sustainable to protect around shortages. This presentation discusses opportunities on how the three sectors align and how through best practices in one area the other two are improved. -
Webinar Keynote: Julien Tremblin, General Manager Europe, Terracycle
Julien Tremblin, European GM of TerraCycle, an innovator in sustainability solutions, shares how companies large and small can drive change, in and out of the pharmaceutical industry and how retailers, brands and consumers are reacting, pushing and adopting as they transition from linear to circular solutions. -
Webinar ESG Risks in Pharma and Biotech – How to Manage your Exposure
Pharmaceutical and Biotechnology companies are exposed to more than their fair share of ESG risks, that, if not addressed properly, can have significant financial and reputational impacts in the long run. To measure a company’s level of exposure with industry-specific material ESG risks and how well a company is managing them, Sustainalytics’ multi-dimensional ESG methodology assesses at least eight different Material ESG Issues (MEIs). Among them, Sustainalytics’ ESG Risk Rating includes Product Governance, Access to Basic Services and Carbon – Own Operations. But what other concepts and indicators should corporate pharmaceutical and biotech professionals consider in their ESG strategies?
Understanding your company’s total ESG risk exposure relies on accurate, independent information, such as reporting, policies and programs to evaluate and improve corporate sustainability. Learn more about how the world’s leading companies leverage Sustainalytics’ ESG Risk Ratings to identify and understand the financially material ESG issues (MEIs) that can affect an organization’s long-term performance. -
Webinar Advanced Digital Customer Segmentation – Elevating Customer Understanding leveraging AI
For the past few years, the traditional one-size-fits-all go-to-market strategy, predominantly based on customer interactions through physical channels, has failed to support drug launches and meet business objectives. Inadequate or incomplete understanding of customer needs has been identified as one of the top-3 root causes. To optimise patient outcome, biopharma companies need to target and engage effectively with key stakeholders, differentiating the interaction based on distinct behaviours and preferences.
In this short webinar, we outline why our advanced digital segmentation approach leveraging AI is the right solution for these challenges and also discuss why it was the right moment for Merck to partner with Deloitte to leapfrog towards data-driven HCP segments. In addition, we talk through what impact can be generated by engaging the right customers, through the right channel with the right frequency and how companies can get started immediately to make the right steps in this direction.
Alessandro de Luca, Group CIO, Head of Information Technology, Merck Group Thomas Bernhardt, Director Life Sciences and Healthcare North and South Europe, Monitor Deloitte Federico Careddu, Manager Life Sciences and Healthcare, Monitor Deloitte Switzerland
Did you enjoy this session? If so, you might like to visit our Connect to Frankfurt platform, where you can browse our collection of 30+ on-demand webinars and learn about our upcoming CPHI Frankfurt event, taking place 1-3 November in Frankfurt, Germany.
Bringing together the global supply chain under one roof, CPHI Frankfurt puts you at the heart of pharma. Can’t attend in person? You can access many of the event offerings online! Browse our exhibitor list, arrange meetings, view on-site content and network – all from home.
Click here to register
-
Webinar Panel: Make America Pharma Again — Is Domestic Production here To Stay In a Post-Pandemic World?
In this webinar is originally broadcast as part of CPHI North America, with speakers Robert Bloder, Chief Business Officer, Ascendia Pharma, Chris Senanayake, CEO & CSO, TCG GreenChem, Inc. and John Pennett, Partner-in-Charge of the National Technology and Life Sciences Group, EisnerAmper. The thread line: The pandemic demonstrated the need for critical life sciences products to be manufactured domestically given supply chain disruptions and reactionary policies from historic suppliers like China and India. At the same time, billions poured into the industry as the US led the race towards developing a vaccine for Covid-19. Two years later, the groundwork has been largely established in terms of increased manufacturing capacity and general industry consensus that domestic production reduces risk, impacting both pharma and biopharma companies as well as CMOs and CDMOs who operate on American soil. But as Covid-generated issues like supply chain delays are gradually elevated, is domestic production here to stay? If so, how will manufacturers keep prices competitive? The panel will dive into current domestic R&D and production capacity, particularly of APIs, and how this influences the CMO sector. -
Webinar Revealing the Marketed and Pipeline Drug Trends –What are the Outsourcing Opportunities?
A presentation that discusses novel drug approval trends and how outsourcing propensity is affected by different factors. This session was broadcast as part of the CPHI North America show. -
Webinar More Women in the Room
In this webinar, originally broadcast as part of CPHI North America, Marrisa Fayer, Ceo and Founder HerHealthEQ discusses in this session: • Where women are needed more (hint, it's everywhere) • Why having women involved in all aspects of the business makes better business • How to get more women involved • How women's involvement will improve outcomes for your customers • What does this mean for women globally • How this affect better women's health outcomes -
Webinar Pharmaceutical Cannabis Global Marketplace
In this webinar, originally broadcast as part of CPHI NA, Michael Sassano, CEO and Chairman, Somai Pharmaceuticals discusses: • Global Pharmaceutical Cannabis market and the history of recent growth • Current trends in the Medical Cannabis market versus the Canadian and American experience • Herbal Medicinal Cannabis ACM versus AIM Cannabinoid pharmaceutical trials • Building or conforming to GMP standards for pharmaceutical cannabinoid applications • Single cannabis API versus multiple API combinations - Delivery devices for Cannabinoid products and best practices for bio-availability This session was broadcast as part of the CPHI North America show. -
Webinar Pharma Marketing Strategy Workshop
Join us for our 1 hour workshop, where industry experts will discuss the latest B2B marketing strategies in Pharma and what can be done to help evolve and create an omnichannel between CDMO’s and Biotech. This session was broadcast as part of the CPHI North America show. -
Webinar Are Pricing Reforms on the Horizon?
Recent Congressional proposals have been targeted at drug pricing reform, including opportunities to allow Medicare to negotiate prices for certain drug products, penalizing prices that rise faster than the rate of inflation, and capping out-of-pocket costs for beneficiaries in the Medicare Part D program. This session will explore where the proposals currently stand, what types of drugs are likely to be impacted, and the likelihood of passage of these proposals. Additionally, the session will explore actions that could be taken on drug pricing through the Administration's authority under the Center for Medicare and Medicaid Innovation (CMMI) -
Webinar Ripple Effects of Expediting Vaccine Development
In this webinar, originally broadcast as part of CPHI North America 2022, Frances Zipp, President and CEO, Lachmann Consultants Services discusses lessons learned and best practices for these relationships that can be applied to both accelerated approvals as well as “normal” drug supply during non-pandemic times. During normal times, sponsors are often challenged with qualifying outsourced manufacturing services for drugs that receive expedited market entry. However, during the COVID-19 pandemic, multiple products have received EUAs in order to bring them to the market at an even more accelerated timeline in order to address the unprecedented public health crisis of COVID-19. In her talk, Frances Zipp will discuss the quality perspective on these manufacturing relationships and will discuss the checks and balances that need to be in place between sponsors and their outsourcing partners in order to ensure a continuous supply of medicines. -
Webinar Start Ups Pitch Session
In this webinar, originally broadcast as part of the Pharmapack Europe show, this session is the opportunity to discover new ideas from companies that are at an early-stage and want to know if their product may match the market's expectations. A patented idea can become the next big thing in pharma packaging! Speakers: Gareth Pearce, Founder Director, Pacifi Ltd. Csilla Rimóczi, Head of ECL Business Line, CODEX Ltd. Tristan Salomon, CEO, Axom Francesco Mainieri, Aghetto Thomas Kern, Managing Partner Vinc ,Strategic Life Sciences Transactions Vincent Galand, Expert in Tech for Pharma & Business Development | Busines Angel (Biotech / Medtech), 321funded -
Webinar Bioprocessing of Hypo-immune iPSC: Toward Cost-effective, High-quality Allogeneic Cell Therapies
Discuss strategies and methodologies to develop scalable, robust, and cost-effective manufacturing processes for allogeneic cell therapies Showcase how induced pluripotent stem cells (iPSC) are being used as renewable sources of allogeneic cancer therapies Demonstrate the criticality of GMP manufacturing capabilities to the success of cell therapies, i.e. leverage biologics manufacturing framework to increase patient access to “off-the-shelf” cellular therapies. This session was broadcast as part of the CPHI North America show. -
Webinar Market Trends Impacting Practice Economics
As the healthcare landscape continues to change, the results can deeply impact the economics of a practice. This session will discuss medical vs pharmacy benefit trends, PBMs, national and government payer changes, and how to stay five steps ahead for community based physicians and infusion centers. This session was broadcast as part of the CPHI North America show. -
Webinar ESG Framework: What Investors are Looking For
As global governments and businesses increasingly make bold claims and commitments to tackle the climate emergency, investors are starting to take note by using the ESG framework to move beyond environmental concerns and focus on a company's impact on all the different ecosystems it operates in. The rise of ESG mutual funds Increase market value with SDG targeting. How are your activities impacting growth & profitability? Do you have a clear roadmap to achieve your sustainability goals in each step of the value chain? This session was broadcast as part of the CPHI North America show. -
Webinar Manufacturing of Cell and Gene Therapies – Challenges and Opportunities
This presentation will focus on: The complexities of bioprocessing and manufacturing of biologics to provide a roadmap for development of effective Efficient and scalable processes and platforms that can be adopted successfully to provide patients wide access to cell and gene therapies. This session was broadcast as part of the CPHI North America show. -
Webinar Strategic Manufacturing Development for Cell Therapy Products
This discussion will focus on four critical areas for process development and overall product life cycle management: Quality, Scalability, COGS and Sustainability Strategies for accelerating process development in efforts to initiate clinical trials quicker will also be discussed Examples, from both Imvax and other industry products, will be provided. This session was broadcast as part of the CPHI North America show. -
Webinar The Evolution of Pharma R&D Models
R&D is the lifeblood of Pharma, and the past year has really showcased the tireless work of pharma companies, start-ups and research institutions who have developed treatments, diagnostics and vaccines against COVID-19. Even in pre-pandemic times, the spiraling costs of drug development (estimated at an average ofUSD 2.6 billion*) mean that pharma companies have always had to think strategically about their R&D activities –staying competitive by reducing risk, managing costs and increasing time to market to justify their significant investments. As treatments become ever more complex, how is the R&D model evolving? Should Pharma companies shift from internal R&D to relying more heavily on CROs and other partners to increase the probability of success? What new tools and technologies can the market call upon to reduce risk? This webinar will discuss the evolution of R&D models, the outsourcing landscape, how to de-risk your development and maximise ROI. Discussion Points: Internal R&D Vs Outsourcing Vs M&A Evolving Outsourcing Landscape in Pharma R&D Strategic advantages to outsourcing your R&D needs (Pros & Cons) What factors to consider when selecting an outsourcing partner How much innovation do we see coming from start-ups? How has COVID-19 drug development diverted R&D budgets from other therapeutic areas? The R&D landscape –how can the model be more sustainable? Digital approaches to increasing drug development success WATCH NOW -
Webinar CPHI Webinar Series Driving Digital Transformation in Pharma
Increased adoption of digital technologies can offer a multitude of benefits for pharma companies –from data optimization and analysis to increased productivity in R&D, to sourcing candidates for clinical trials. How can pharma companies take a more active approach to implementing digital innovation, to help cope with new challenges in the healthcare industry? One approach to speeding up adoption is to engage with start-ups and established technology companies who can bring different expertise and a more agile approach. This webinar will explore the relationship between Pharma companies and start-ups, as the key to unlocking digital transformation. Why attend? What can digitalization do for Pharma companies? What are the benefits? Has the Covid-19 pandemic accelerated digital innovation in the pharma industry? Partnering for success –how engaging with start-ups and tech companies can help acquire expertise and accelerate development A review of successful solutions, initiatives and Start-Ups helping to solve healthcare challenges What does the future look like? Can we expect to see increased digital transformation? -
Webinar CPHI Webinar 2021 Series in partnership with Pharmapack Traceability: challenges and opportunities for Direct-to-Patient delivery
Building a comprehensive track-and-trace system for medicines, which aligns with the European Falsified Medicines Directive and ensures compliance is a must in today’s environment. Pharmacies, hospitals, HCPs, carers and patients now expect a completely secure and traceable delivery process, which requires buy-in from pharmaceutical companies and distributors involved in the supply chain of Investigational Medicinal Products (IMP), SKU’s and unique doses. This webinar provides an overview of the key trends and developments to improve the security of direct-2-patient drug delivery. Key Discussion Points A global overview of regulations for traceability of medical devices and medicines An overview of global standards that apply to medicines and medical device regulations, as well as to identification of investigational products, locations and business communication in clinical trials Technical challenges when tracking medicinal products along the product life cycle, from clinical trials to post marketing authorization What does the future look like? The potential of delivering Direct-to-Patient unique doses Why attend? Understand global regulations for traceability of drug products and medical devices Get to grips with the technical challenges of tracking medicinal products along the product life cycle, from clinical trials to post marketing authorization Learn what’s next for this sector, and explore the opportunity of delivering Direct-to-Patient unique doses -
Webinar Biomanufacturing and Moore's Law
The webinar addresses the need for new modalities, evolution of bio manufacturing of new modalities, and comparison with semiconductor manufacturing. This session was broadcast as part of the CPHI North America show. -
Webinar Considerations in Extractables and Leachables Testing of Drug-Device Combination Products
This presentation will provide an overview of the differences and similarities of E&L requirements between pharmaceutical products and medical devices. The key elements regarding E&L testing design of combination products will be discussed. Several case studies on the extractables and leachables testing of combination products will be presented. This session was broadcast as part of the CPHI North America show. -
Webinar CPHI Webinar Series Green Chemistry: Inspiring Sustainable Drug Manufacturing
What will this webinar address? Green chemistry strives to reduce theenvironmentalimpact of the manufacture and synthesis of pharmaceutical intermediates for a more sustainable drug manufacturing process. Although the industry has beentaking steps towardsthe application of green chemistryfor drug development and manufacture, there is more to be done on recognizing thecompetitive advantagesof adoption –namely cost benefits, waste reduction, reduced toxicity and increased product yield. This webinar will deliver insights into the key principles of green chemistry, highlight innovative practices in the space and outline the roadmap for adoption, helping your organization towards a more economical and ecological future. Why attend? Understand the foundations and key principles of Green Chemistry Learn how green chemistry can contribute to sustainable development Understand routes to adoption and implementation for both commercial and environmental benefits. What innovation is taking place in the green chemistry arena? What can we learn from innovators and start-ups? -
Webinar How to Drive Sustainable Packaging Development in a post-COVID world?
For Pharma Packaging companies who have not yet embedded a sustainable approach into their manufacturing and distribution processes, their hand is now being forced by drug retailers and hospitals who have come to expect more eco-friendly, cost-effective recycling solutions for plastic products and materials used in healthcare delivery. To remain competitive, Pharma companies must now collaborate with suppliers on how their packaging processes address sustainability. This webinar will provide an overview on the drive for sustainable development and improving the recyclability of healthcare products. Key Discussion Points Innovative approaches to sustainable packaging solutions Adopting a value chain approach to meet expectations of hospitals & drug retailers Meeting sustainability goals and remaining cost-effective –the pharma perspective Challenges and sustainable solutions –the supplier’s perspective Why attend? Get the complete overview on the pharma sustainable trends outlook for 2021 in just 60 minutes Hear from experts who share their advices for strategies implementing and best-practices to set-up a collaborative and value-chain approach -
Webinar Pharma Outlook 2021
Featuring three speakers from leading market intelligence organisations, this webinar outlines the key trends and developments in the pharma sector to be aware of over the coming twelve months. Daniel Chancellor, Thought Leadership Director, Informa Pharma Intelligence, provided a snapshot of pharma pipeline activity during 2020, with oncology being the therapy area that continues to dominate clinical trial activity, with immune and central nervous system jockeying for second and third position.
He also shone a light over the disruption that the COVID-19 pandemic has wrought over trials and the industry’s rapid response to the crisis in terms of developing both vaccines and therapeutics against the virus.
“We have around three and a half thousand clinical trials initiated or planned against COVID-19 since the start of the year, with around 700 pipeline drugs and not to mention some of the repurposed efforts that have been run by academic groups, too,” he told the webinar.
Sarah Rickwood, Vice President European Thought Leadership, IQVIA presented nine key issues that are going to drive change in 2021 and beyond, a blend of some directly accelerated by the current pandemic and others that were issues already in existence prior to the pandemic, “but are really going to see their full impact on changing the shape of pharma and health care in 2021 and beyond regardless.”
She told the audience that the industry is actually dealing with three crises rather than one, and that the first half of the 2020s is likely to be flavoured by the pandemic and its consequences.
“We have the infection crisis, which we hope will be addressed by the vaccines which have come through with such an amazing effort from the pharmaceutical industry,” she said, adding that the challenge is that the infection crisis has triggered two other crises which will stretch through 2021 and beyond.
“The first is a crisis of healthcare systems, the non-COVID treatments of patients in healthcare systems have been delayed, changed and negatively impacted by the fact that healthcare systems have had to pivot to COVID treatment and addressing those patient backlogs which vary by country and by therapy area, is going to take a long period.”
She also pointed to the beginning of the economic crisis, with really worrying drops in GDP in Q2: “The full impact of mass employment has yet to be realized and that will mean health care system budgets will be constrained. So at best I can say is that we’re not living in a post-pandemic environment in 2021, we're living in a vaccine plus environment.”
And rounding off the webinar, Saman Kay, Solutions Consultant for Regulatory Intelligence at Clarivate Analytics, took a look at the future of drug manufacturing technology and assessed the opportunities and challenges of adopting continous manufacturing processes in place of traditional batch manufacturing. -
Webinar The Future of Biomanufacturing: Capacity, Capability and Careers WATCH NOW
What will this webinar address? There is more emphasis than ever before on biopharma innovation – a sentiment heightened by the speed at which the industry came together to develop vaccines for COVID-19 – setting new expectations for the future. With development comes fulfillment, and with biomanufacturing capacity at an all-time low, what infrastructure is needed to keep pace with innovation? This webinar will provide an overview of biopharma trends and technologies, and address challenges around supply chain, capacity and workforce, to ensure continued productivity, efficiency, and output in the biomanufacturing sector. Key discussion points Biopharmaceutical landscape outlook - what are the short and long-term impacts of COVID-19? Rising trends and technologies – mRNA, immunology, and vaccines Building flexible manufacturing networks and increasing capacity Single-use tech availability Training the workforce of the future Why attend? Get a complete overview of trends, opportunities, and disruptions in the biomanufacturing sector, in just 60 minutes. Hear from experts on shaping biomanufacturing strategies to meet industry needs. Put your questions to our speakers during the live Q&A session -
Webinar Biosimilars in Oncology
The availability of of biologic therapies to treat oncology disease conditions has led to improved clinical outcomes, but the high cost can create barriers and disparities in treatment access, and therefore outcomes. The use of biosimilars can improve patients' access to care by decreasing treatment costs and creating price competition between the originator product and biosimilar competitors. Watch this webinar from Clarivate on-demand now to understand the following: What is the current landscape of biosimilars in oncology? How do biosimilar approvals and market access differ between the United States and Europe? What is interchangeability and what is its impact? What are the barriers to uptake of biosimilars in oncology? What is the future of biosimilars in oncology? -
Webinar Trends in Packaging Cosmetics
Packaging solutions for cosmeceuticals and pharmaceuticals share many similarities; for both, packaging needs to be functional, protective and accessible for the end user. With cosmetics however, branding plays a huge role in attracting and engaging consumers – who now have an increased consciousness and desire for eco-friendly design, zero-waste and refillable solutions. For pharma packaging innovators, adherence is at the heart of product development, but this has historically been less of a consideration on the cosmetics side – what can cosmetic companies learn from pharma in terms of accessible and adherent packaging offerings? This webinar, produced in partnership with Euromonitor International and BEAUTYSTREAMS explores some of the key trends affecting the cosmetic packaging market, and the parallels between pharma and cosmetics. Watch on-demand now! -
Webinar The Road to Pharmapack 2022
Join the Pharmapack team for an interactive and informative webinar as we map out the road to the Pharmapack 2022 event, 18 & 19 May in Paris, France. This session will outline how we are preparing for the 2022 edition of the show, share key planning milestones and the measures we are taking to bring you a safe, compliant, in-person event. We will also cover the multitude of digital products, solutions and initiatives we have developed in recent months to support you in lead generation, brand visibility and business continuity. Why attend? Understand how Informa Markets is preparing for the 2022 edition of Pharmapack, hear how best to prepare your team to participate - what are the key considerations. And learn how our digital product portfolio can support your lead-gen and visibility efforts. -
Whitepaper Sustainability Report 2024: a Guide to Growing a Greener Industry
The pharmaceutical industry is well aware of the impacts of climate change on human health, and increasingly aware of the impact the industry has in contributing to this. Connecting these dots, leaders in the field see how planet health is intrinsically linked to human health, and therefore, must be addressed as part of the mission of the healthcare industry. At CPHI, we’ve looked into the aspects of the pharmaceutical value chain that contribute to green house gas emissions, as well as the effects of waste. Now, the Sustainability Report 2024, a guide to growing a greener industry, takes these lessons to the next step and proposes how exactly pharmaceutical specialists can help to reduce climate change and preserve life. With input from experts from The PSCI, BioPhorum, Nelipak Healthcare Packaging, Pöppelmann FAMAC Pharma-Medical, Anthesis Consulting Group, Pantheon Strategies, Schneider Electric, and Singota Solutions, the report covers key challenges and solutions for all players, including valuable case studies, and information on legislation, investing, and materiality. The report covers: Chapter one: What did we learn from 2023 and where are we now? Chapter two: Wrangling with regulation Chapter three: Investing in sustainability Chapter four: What can emerging markets bring to the sustainability table? Chapter five: We’re all living in a material world Chapter six: Collaborative action through partnerships Chapter seven: At the heart of sustainable pharma We also hear from CPHI itself about the approach to making events more environmentally conscious and the role they can play in bringing sustainability conversations to the fore. -
Whitepaper CPHI Sustainability Trend Report: Towards a Greener Future
Sustainable supply chain practices within the pharma industry are an increasing concern, as pharma companies look to address the environmental impact of drug development and manufacturing, reduce reliance on non-recyclable materials and assure end-users of steps taken to minimise their carbon footprint.
In our Sustainability Report 2023, experts from the Pharmaceutical Supply Chain Initiative (PSCI), BioPhorum, IQVIA, CordenPharma and Bormioli Pharma uncover the key sustainability challenges for pharma and share recommendations for a greener future. -
Whitepaper The 2024 Pharma Outsourcing Forecast: strategic partnerships across global landscapes
Pharmaceutical outsourcing strategies have evolved to match the dynamic needs of drugmakers and biotechs. As more and more pharmaceutical companies outsource some percentage of their operations, the partnerships between contracted organisations and drug sponsors are shifting in response to changing priorities from both parties.
The future of pharmaceutical outsourcing is constantly changing, and success will depend on the pharma industry’s ability to adapt and evolve to meet these challenges. -
Podcast CPHI Podcast Series: Analysing ESG Audits with the PSCI
This episode of the CPHI Podcast Series, hosted by Digital Editor Lucy Chard, goes through the results from the recent audits from the PSCI conducted on suppliers across the pharmaceutical industry, looking into ESG outcomes. The PSCI have recently published the results from their audits and to talk us through these Digital Editor Lucy Chard is joined by Birgit Skuballa. Skuballa represents Bayer in the PSCI as Chair of the PSCI Audit Committee, and gives us a information on how these audits work, the different pillars across the pharmaceutical industry that the audits address, how this changes regionally, and importantly, how the PSCI work with their members and the participating suppliers afterwards to improve their practices. -
Whitepaper Digital Pharma Marketing Toolkit
Audiences are plugged into digital marketing more than ever. In our free eBook, we look at how to create and execute a successful digital marketing campaign. From developing an omnichannel presence to creating personalised content, we've got you covered. -
Whitepaper The 2023 CPHI Annual Report
Discover fascinating insights from over 250 global industry leaders that illuminate anticipated industry trends in 2024. And dive into a detailed analysis of major pharma markets on various vital metrics.
Plus, gain the visionary perspectives of global experts shaping the industry today and forecasting the trends of tomorrow. This includes key voices from Pharmatech Associates, Vertex Pharmaceuticals, Edgewater Capital Partners, Pharma and Biopharma Outsourcing Association, GlobalData, BioProcess Technology Group, BioPlan Associates, Ysios Capital and Sirio Europe. -
Podcast CPHI Podcast Series: the power of digital marketing in pharma
On this episode of the podcast Digital Editor Lucy Chard is joined by marketing specialist and entrepreneur, Raman Sehgal. Sehgal is the founder of several companies, including life science marketing company Ramarketing. In this podcast Raman and I discuss the different ways marketing can help companies in achieving their goals, whether it’s in how to go about finding the right partner, creating the right messaging around a companies’ ESG values, or how to attract talent. We also go on to talk about the future of digital marketing, especially the increased use of AI in today’s world, how much this has changed everyday practices already, and the scope of what is to come. -
Video Boosting DSCSA Regulatory Preparedness for FDA’s Drug Supply Chain Security Act Compliance with Copilot Integrated Generative AI/ML
Why DSCSA was created by US Congress, some history of past undertakings Who’s who and what’s what with DSCSA trading partner roles for drug manufacturers, repackagers, wholesale distributors, 3rd party logistics & service providers, dispensers Challenges for regulatory compliance with DSCSA from various FDA DSCSA pilot project participants including key findings from FDA’s Final report assessment on participant pilot reports submitted to FDA Why many remain unprepared-search gauntlet with US FDA.Gov website abysmal metadata searching specific to DSCSA vs Google browser search inherent web noise or clutter when seeking DSCSA relevant context-specific information Illustrate new AI/ML Microsoft Copilot & chatbot platform/ technology with underlying AI/ML generative technology via ChatpGPT-4 LLM under the hood Extending Copilot with your custom enterprise external data and leveraging cloud-based Microsoft Sharepoint Online Full-Text search on actual US FDA collections hosted within Sharepoint Online to fact-check generative response accuracy by You-the-Human -
Video Difficulties Meeting US FDA Regulatory Requirements
Difficulties Meeting US FDA Regulatory Requirements -
Video Unlocking Opportunities: Exploring the Middle East Pharma Market
Discover the burgeoning pharmaceutical landscape in the heart of the Middle East, where Saudi Arabia stands as a beacon of opportunity. Explore new frontiers and understand the potential for innovation and growth in this dynamic market. -
Video New Business Opportunities Between USA and Mexico - Supply Chain Resiliency
One of the problems that today US suffers is the persistent shortages in pharmaceuticals. The concentration of key products in countries as China and India is a risk for the United States and a necessity to diversify and include new partners. Special efforts are needed to have supply chain resiliency. Mexico’s pharmaceutical industry is one of the most developed in the region with not only specialized and skilled workforce but also high standards and the capacity to produce effective medicines that can help reduce those shortages. United States and Mexico can take advantage of nearshoring to improve pharmaceutical exports to the US that will benefit both countries, by making them complementary in the supply chain in this critical sector. -
Video China Drug Master File (DMF) Comparing with U.S. and EU
This session provides an overview of the Drug Master File (DMF) regulatory framework and registration pathways covering Active Pharmaceutical ingredients (APIs), Excipients and Packaging Materials. The aim is to provide an overview of the local requirements and advise on the best and quickest route to the Chinese market. Thanks to the global harmonization efforts, the Chinese DMF regulatory system is becoming more similar with US DMF, EU ASMF and CEP requirements. However, local requirements and gaps in the regulations still exists, this session shares the registration procedures, timeline and costs of China DMF and further provides a comparison of China DMF with U.S. and EU from various perspectives. Finally, this session shares the most common pitfalls in terms of technical requirements and provide hands-on tips to help minimize risks and issues with China regulatory authorities, and smooth DMF registration process in China. -
Video Bridging the Healthcare Access Gap with AI
US healthcare industry has undergone tremendous digital transformation in recent years, but critical care access to lower income ethnicities is still in its nascency. This population have historically showcased substantial hesitation to seek any timely medical assistance. While lack of sufficient financial resources plays a critical role, the existing cultural and knowledge barriers also contribute significantly to widening the healthcare access gap. Recent technological advancements have opened avenues that can identify both patients and their caregivers, who play a critical role in patients’ diagnosis, building healthcare knowledge and instilling confidence in required therapeutic procedures. A digitized go-to-market strategy featuring connected media coupled with smart wearable devices and location encoding can be collectively leveraged to reach this key audience group. The AI/ML algorithms can be thoroughly trained to identify useful targeting signals from user's digital presence. These user interactions can be further assimilated to identify contextually relevant interest topics and decipher potential audience on digital avenues to serve that brand message. In conclusion, grasping the true health access journey of any lower income ethnic group is important to design beneficial touchpoints that can alleviate patients’ concerns and allow them to break their own access barriers and opt for appropriate healthcare. -
Video The Role of Diversity in Shaping the Future
The pharmaceutical industry's reputation greatly improved during the pandemic, an era that not only underscored its critical contributions but also shone a light on the imperative need of embracing diversity, equity, and inclusion (DEI). Motivated by both social movements and the unique demands of the pandemic, the industry's commitment to DEI underscores its significance not just for enhancing corporate reputation, but also for ensuring fairness and respect within the workplace. We invite you to join us in a session designed to delve into: What do we mean by Diversity? Current State of DEI in the Pharma Industry Future Trend Outlook -
Video Lunch & Learn: Navigating Regulatory and IP issues in life sciences
The following session has been SOLD OUT. For any inquiries, please contact a member of staff on the day Take a break from the show floor and join us for a bite, network with colleagues, and stay informed with the latest updates and insights on US Pharma regulations. Join attorneys from Womble Bond Dickinson (US) LLP for a discussion about regulatory and intellectual property considerations in the life sciences arena. As part of the lunch and learn, we will discuss: Licensing of proprietary information for drugs and biologics through FDA Master Files and related regulatory issues; Collaboration agreements and data privacy When does the clock start ticking for filing a patent (and what counts as a public disclosure) In addition to our 30-minute presentation, we will have attorneys on hand to address issues involving data privacy and cybersecurity, trademark registration, and intellectual property litigation. -
Video Lightning Round: Excipients 4.0 - Novel Excipients for Drug Delivery
Excipients for pharmaceutical processes – How to identify opportunities for novel excipients that solve formulation and process challenges"; Needs of the industry for novel excipients, ways to identify current gaps and work together to resolve them, 1-2 key examples of common process challenges that might be solvable with novel excipients. -
Video Navigating the Waters of Change: The Impact of New PFAS Regulations on the Manufacture of Excipients in the Pharmaceutical Industry
The growing awareness of the environmental and health risks associated with (PFAS) has led to a global movement to phase out these chemicals from various products. In the pharmaceutical industry, PFAS have been used in some excipients due to their unique properties but concerns about potential health effects have prompted regulators and pharmaceutical companies to take action to reduce their use. This session will explore: Alternative excipients that can maintain product efficacy while reducing reliance on PFAS Challenges in removing PFAS from products and the associated regulatory aspects Regulatory compliance and adaptations in response to PFAS concerns -
Video Panel: Making Excipients Great Again
In Pharma, excipients, often underappreciated despite their essential role in drug formulation and delivery, are gaining renewed attention. With growing investments in technology and increasing consumer demand for natural ingredients, excipients are taking on an expanded role. Join our esteemed panel of experts as they delve into the evolving landscape of excipients, their increasing importance in enhancing drug effectiveness and patient outcomes, and the shifting dynamics in this field. Why excipients are regarded as unsung heroes in the industry Understand how natural excipients can be leveraged to improve the solubility and bioavailability Analyzing the potential of excipients in personalized medicine, addressing formulation challenges, and improving patient compliance -
Video Navigating the Talent Crisis: Strategies for Safeguarding the Workforce of the Future
The Pharma and Life Sciences industry confronts a pressing challenge—a substantial talent shortage compounded by the effects of the Great Resignation. To remain competitive and seize emerging opportunities, companies must prioritize both external hiring and internal mobility strategies. This pivotal issue prompts a reevaluation of talent acquisition and development approaches within the industry. -
Video Thriving Workforce, Thriving Business: Unveiling the Power of Wellness for Company Success
Discover the compelling link between employee wellness, engagement, and company success. Learn practical strategies to enhance employee well-being, boost recruitment efforts, and cultivate a positive corporate culture. Explore how wellness initiatives drive sales performance and leadership effectiveness. Gain actionable insights to create a thriving workplace where employees are motivated, engaged, and empowered to contribute to organizational growth. Don't miss this opportunity to harness the power of wellness for lasting success in your company. -
Video Discussion: The Voices of Women in Pharma
An open conversation with two women at different stages of their careers in pharma. The discussion will delve into the diverse experiences, challenges, and opportunities facing women in our industry. Prepare for a discussion that will explore the evolution of gender diversity, personal and professional growth strategies, and the shifting industry landscape. -
Video Discussion: Driving Patient-Centricity Through Diversity
How can we become truly patient-centric? This session will explore strategies for deeper collaboration between big pharma and patients, set against the backdrop of an evolving healthcare landscape where patient voices are key to innovation. Explore dynamic pathways for incorporating patient insights into drug development, featuring an industry leader and patient advocate, discussing strategies for empathetic understanding, effective collaboration frameworks, and the role of transparency and innovative technologies in building trust and measuring impact. -
Video Keynote: The Importance of Excipient Grade in Pharmaceutical Drug Development
This presentation will consider the following topics: Why is it important for pharma companies to use the correct grade of excipient in their drug formulation development? What should pharma companies do to ensure their excipient supply chain is secure and their excipients are of the correct grade? What is ‘Testing Up’ the grade of excipient and is this a concern? Can non- compendial grade excipients be used in drug products? What are the functional related characteristics of excipients and their importance in drug development? Is it important to consider excipient grade in academic pharmaceutical research? What should a pharma company consider when changing an excipient supplier? -
Video Excipients and Patient Centric Drug Product Design: A Symbiotic Relationship
Beyond traditional roles: Explore how excipients are no longer passive additives but active contributors to drug efficacy and delivery, offering diverse functionalities Precision medicine and tailored therapies: Delve into the role of excipient solutions in enabling precision medicine by allowing customized drug delivery profiles, optimized for individual patients Enhancing patient adherence and experience: Learn how excipient innovations are transforming patient experiences, from taste-masking to novel dosage forms, resulting in better medication compliance and outcomes -
Video Lightning Round: Excipients 4.0 - Cyclodextrins (CDs)
Cyclodextrins (CDs) - explore novel applications in neurodegenerative diseases, gene therapy, and biotechnology, showcasing innovative structures, mechanisms, challenges, and results, with a focus on Alzheimer's, Parkinson's, rare genetic disorders, and potential expansion to longevity and infectious diseases, presenting a non-viral gene delivery platform and demonstrating CD utilization in vaccine adjuvants and biotechnological tools. -
Video Lightning Round: Excipients 4.0 - Innovations in Novel Stabilization Technology
Changing the world one film at a time" At Jurata we are focused on eliminating the need for cold chain storage of vaccines and biologics, including mRNA-LNPs, in order to improve global health equity. Our novel stabalization technology is 10x more efficient to produce than lyophilized products and allows for sublingual and buccal delivery. -
Video Ask the Excipient Expert
Have burning questions about excipient functionalities, selection criteria, or their role in drug formulation? Join us as we bring together a seasoned excipient expert to address your queries, share insights, and offer a deeper understanding of these vital components. Whether you're a pharmaceutical professional, researcher, or simply curious about the science behind medicines. -
Video Biomanufacturing Trend Outlook
Bioproduction in the US pharmaceutical industry is crucial for delivering life-saving medications efficiently. Advancements in biotechnology and rising demand are driving rapid evolution in bioproduction. While state-of-the-art facilities and strict regulations define the sector, challenges in scalability, cost-effectiveness, and compliance persist, necessitating ongoing innovation and collaboration across academia, government, and industry for a sustainable ecosystem. Reviewing emerging technologies such as continuous bioprocessing and single-use systems and their role in transforming bio-production in the US? How can regulatory frameworks evolve to support biopharmaceutical innovation while maintaining patient safety and product quality under the FDA's emphasis on advancing regulatory science -
Video Branding Yourself in a Crowded Market
It can be difficult to stand out when you’re looking to attract new pharma partners. Reputation and referrals are key, but they aren’t always enough. To be seen as unique and increase awareness with potential partners, consider examining and better establishing your brand, so your audiences know what you stand for, why it’s relevant, and how it’s different. This interactive, engaging workshop will review the elements of branding your business and sharpening your communications. The conversation and exercises will be focused specifically around the challenges of service providers in the pharmaceutical space. We’ll get participants to start thinking about their brands more deeply, focusing on aspects including: Defining your unique value props Focusing on brand story as a differentiator Identifying what’s truly important to key decision-makers, including their pain points, hurdles, and motivations Examining positioning and communication white space Uncovering foundational insights than can help you reframe your communications -
Podcast CPHI Podcast Series: How manufacturing in Africa can improve patient access to medicine
In this episode Digital Editor Lucy Chard is joined by Daniel Mora-Brito, Engagement Manager and Thought Leader for Global Public Health at IQVIA, where they discuss the future of pharmaceutical manufacturing in Africa. Based in Geneva, Switzerland, Daniel is an Engagement Manager in IQVIA’s EMEA Thought Leadership team. Daniel’s work focuses on exploring current and emerging trends within the realms of global health security, health system strengthening, and equity in access to life-saving commodities and services. Daniel has close to 20 years of experience in the multilateral sector, having occupied advisory positions in the United Nations system, the Global Fund to Fight AIDS, Tuberculosis and Malaria, and a host of civil society organisations. His expertise covers areas such as health policy, development operations, public-private partnerships, and multi-stakeholder coordination. Daniel holds graduate degrees in public management and social policy and development from the Universidad Metropolitana in Caracas, Venezuela, and the University of Texas at Austin, respectively, with training in health policy and economics at the London School of Economics. Daniel initially gives some background to the health landscape in the continent, covering some of the greatest threats seen there over the years and that are still impacting people’s lives on a daily basis, helping to drive home the importance of improved, localised healthcare in the continent. Lucy and Daniel discuss the economic factors that underpin a lot of the transformations across the region, from the perspective of those on home turf and the companies from different regions looking to invest and expand into this space. Daniel mentions the different actors in the region and what expanding capacities in Africa will mean for patient access in a rapidly expanding population. -
Podcast CPHI Podcast Series: What to expect from novel excipients?
This podcast episode aims to explain the importance and intricacies of developing excipients and then goes into more depth regarding the excipient market, how this differs globally, what tactics different companies are now using to gain ground and push the field forward to be able to develop more innovative drug delivery systems. Lubrizol Life Science is a pioneer in the novel excipient development space so DiFranco is well placed to give a deep level of insight into the field as it stands, some of the innovative products they’re developing at Lubrizol, and the trends to look out for the in future. Nick leads the strategy, marketing, and business development efforts for Lubrizol’s solubility enhancement technologies: oral-grade Apinovex™ polymer and injectable-grade Apisolex™ polymer. He is responsible for product launch and promotion as well as external partnerships and collaborations for these novel technologies. Since 2017, Nick has held both technical and commercial leadership roles at Lubrizol spanning oral drug delivery technologies, long-acting implantables and injectables, and CDMO services. Nick holds a B.S. in Biomedical Engineering (Biomaterials focus) and a Master of Engineering and Management degree from Case Western Reserve University. -
Whitepaper Pharmapack Survey and Annual Report 2024
The Pharmapack Annual Survey and Report highlights key insights from the pharmaceutical packaging and drug delivery sectors across the globe. This year, the Pharmapack Drug Delivery Innovation Index provides in-depth observations through a collaterialised metric of drug delivery and device innovation across major pharma nations. Takeaways include: Global drug delivery innovation index hits record high with sustained growth expected in 2024 France see biggest year-on-year rise but Germany moves ahead of the USA to lead the rankings for the first time Start-Up Hub remains one of the fastest growing areas Autoinjectors narrowly supplant prefilled syringes as injectable type predicted to see greatest number of approvals in 2024 and more Download the Pharmapack Annual Survey and Report 2024 to learn more. -
Podcast CPHI Podcast Series: The 2023 Retro – what have we learnt from the past year in pharma?
Welcome to the last episode of 2023! Digital Editor Lucy Chard is joined by her team in a special retrospective episode to close out the year. Guests Vivian Xie, Editor for CPHI Online and Tara Dougal, Content Director for Pharma, discuss their highlights from the year; diversity, sustainability, and the CPHI Barcelona event cropping up as top features. The Women in Pharma series that Vivian has worked on for the past year has been a resounding success, some of our most impactful content on CPHI Online, which, as arguably some of our most important content we are so pleased to see that reflected with our audience. We also cover sustainability, drug approvals, Alzheimer's research, mRNA oncology vaccines, and digital marketing for pharma companies. -
Podcast CPHI Podcast Series: Start-ups take centre stage at CPHI Barcelona
Matthew Wise has worked with the CPHI Content team a lot over the last couple of months discussing the start-up space in the pharma industry, particularly in leading up to our annual event for CPHI which took place this October in Barcelona. In this podcast we spoke about the event, which for the first time held a specific area for Start-up companies, something that is particularly applicable to the Catalan region as a leading area for innovation in pharma and healthcare, and we also discuss what this start-up market means for the companies. We also cover how we, in our respective roles, can lend a hand from an outside perspective, and help to facilitate partnerships all across the pharma value chain and ultimately encourage innovation and progress in the field. -
Webinar Panel: The CDMO Outlook
We finished off the day by learning to understand the latest trends and developments in the CDMO space, and strategies for developing successful partnerships. Market trends and developments in the CDMO space Reviewing your in-house capabilities to determine what services you need Best practices in optimising communication with your chosen vendor for optimal results When not to outsource? -
Webinar Antimicrobial Resistance – Manufacturing Antibiotics with Environment at the Centre
BSI – set the scene of the challenge of manufacturing antibiotics and the environment - 3 mins
Centrient – what we have done so far to invest in mitigating environmental threat?– 5 mins BSI – One Health approach towards setting industry standard and certification for combatting antibiotics discharge in the environment - 10 mins BSI/Centrient – next steps – how to minimize impact of AMR through externally validated environmental compliance? – 3 mins Q&A – 5 mins -
Webinar Pharmaceutical Ecosystem Development - The UAE Journey
Evolution of the pharmaceutical industry in the UAE and the significant increase in the number of factories and diversity of products. Strategies put in place and collaboration between the government and the private sector Attraction of Innovation and medicines of the future. Opportunities and potential for investment and expansion -
Podcast CPHI Podcast Series: What does the changing US Pharma market mean for industry and patients alike?
The podcast will aim to cover the recent changes in legislature around drug pricing in the USA, and what this means for the global pharma market, as well as the impact it will have on the US population in terms of access to medicine. The price negotiations with Medicare are targeting medications used in the treatment of many chronic diseases such as cardiovascular disease and diabetes. We will take a look at what this means for people with chronic disease, and conversely the impact on the individual pharma companies and then more widely, on innovation in the industry. -
Podcast CPHI Podcast Series: Exploring neurological frontiers in Alzheimer's and beyond
Alan Palmer of Elixa MediScience joins us for this episode to talk about neurological disorders including Alzheimer’s disease and everything that is encompassed under this terminology. In the podcast we cover the history of disease and research, the social and economic impacts, the different modalities and developments in therapeutics, and what the future of research into care in this field holds. Palmer explains the intricacies of anti-amyloid medications in light of the recent therapeutic approvals, and what this could mean for patients today and in the future. -
Podcast CPHI Podcast Series: AI and drug discovery
In this episode, Bill Whitford from DPS Global speaks to Editor Lucy Chard giving context to the use of AI in different aspects of medicine and drug discovery, including how it can help revamp drug repurposing strategies and save time when searching for drug targets. We also see the implications the technology could have socially, by helping to eliminate bias in clinical trials, and the prospect of how far AI could go. -
Podcast CPHI Podcast Series: The ripple effects of nearshoring in pharma
In the most recent instalment of the CPHI Podcast Series, Digital Editor Lucy Chard is joined by Sandra Sánchez y Oldenhage, Founder and CEO of PharmAdvice, to discuss nearshoring and reshoring in the pharmaceutical industry, and the subsequent ripple effects. Sandra is a 35-year life sciences executive with significant operational and international experience operating top ranked pharmaceutical companies mainly in Latin America, including Pfizer, Merck, Sharp & Dohme, and Novartis, and was successful in establishing operations for Amgen and Biogen in Mexico as President and GM. Her entrepreneurial spirit has led her to form her own consulting firm (PharmAdvice), leveraging her extensive and in-depth experience in the commercial, regulatory, government affairs and access fields throughout Latin America. Sandra touches on manufacturing and supply, the monopoly that some regions currently have in certain fields, the various advantages and challenges that affect both the countries and the pharma companies, as well as the impacts nearshoring has economically, through things such as trade, and importantly the social impact, in equitable access to medicine, demonstrating the real ripple effect nearshoring has. -
Podcast CPHI Podcast Series: outsourcing and manufacturing trends
Gil Roth, President of the Pharma & Biopharma Outsourcing Association, delves into the most exciting aspects of outsourcing and manufacturing in this interview, including some of the biggest areas of innovation and technology, such as cell and gene therapeutics. We also get an update on the industry as a whole since the COVID-19 pandemic and how companies are adapting to changes in the market and regional trends. -
Podcast CPHI Podcast Series: Designing Drug Conjugates
In this episode from the CPHI Podcast Series Louise Duffy and Campbell Bunce from Abzena speak to Digital Editor Lucy Chard, using their cumulative knowledge to give an overview of the drug development and manufacturing processes, demonstrating all of the aspects that go into such an undertaking as making a new drug for a patient population. They highlight some of the key areas that teams have to work on, the detailed analysis that has to take place at each stage, as well as within the wider context with regulations to consider, and finally they highlight some of the key areas for innovation and for expanding knowledge in the field. -
Video A Guide to Successful Collaboration - Considerations When Choosing Your First CDMO as a Start-Up
In pharma, it is essential to identify the right partner to ensure a successful outcome. Time and time again, mistakes have been made in this process, causing clinical studies to fail and for the company to take a heavy hit. Want to avoid this? join our one-hour masterclass where we will devise a checklist for choosing your first CDMO. Discussion Points Thinking ahead before executing – why it is essential to map out what you need to achieve in all departments Why creating a business partnership case to potential CDMOs will determine if they are the right fit for you A checklist of the common pitfalls and how to avoid -
Video The Music of Process Development Science
Where QBD was the orchestral sheet music that determined how drug product development has been harmonized over the last 15 or so years, contemporary patient-centric risk-based approaches to drug development are the pop evolution. The music is still beautiful when produced well - but the focus is no longer on the full orchestra, it's on key players and moments. First - think McCartney and Lennon - one without the other and we don’t have the Beatles. The same can be said for collaboration between drug process development and commercial production colleagues. They are central to ensuring that products are easier to transfer and can be scaled, and that manufacturing challenges are identified early on and addressed proactively. In his keynote, Bernd Schümmelfeder Director-Team Leader PPD Business Development, Pfizer explores how lyrics (communication), rhythm (standardization), bass (structured development processes), and lead guitar (the use of smart technologies) throughout the drug development process come together to create beautiful music - a drug that gets to patients safely and at pace. Key areas for discussion include: Clear decision and inflection points to manage costs and prioritize projects with a higher probability of success Smart technologies like modeling to identify challenges early on and avoid late-stage pitfalls Use of platform technologies to simplify the tech transfer process and improve process robustness Areas to optimize the product development and manufacturing process to make it more efficient, cost-effective, and patient-focused -
Video Understanding Current Drivers in Drug Pricing and What this Means for the Industry
Get the inside focus on the latest drug pricing developments in the US market. Listen to the current legislator updates and how it will affect patient costs while reviewing the rising manufacturing costs from inflation and supply constraints and the market’s reaction: Examining current manufacture drug costs vs patient costs and how this reflects in current market pricing Reviewing legislator updates in the market and expected changes to the industry Calculating rising inflation and supply constraints, what will be the impact on the cost of manufacturing? -
Video Psychedelic Research: Where Do We Stand in the Global Market?
Join our thought leadership session to understand the current state of Psychedelics in the pharmaceutical industry. Delve into the potential psychedelics can bring and why the industry needs to take notice of its growing importance: How psychedelics can play a role in the treatment of mental health conditions Emerging investigator initiated and sponsored clinical trials Drug development and regulatory challenges to counter before mass adoption -
Video Forecasting and Strategy – Maximizing Product Accessibility and Uptake, Improving Commercial Forecasting Skills and Analytics
As we bring in 2023, pharmaceutical companies will undoubtedly be looking ahead. But how will the events of the past two COVID years influence future aspirations? In the past, pharmaceutical companies used a mix of bottom-up and top-down processes to budget for the next fiscal year, and then made quarterly revisions during the year. Nowadays, both on the revenue side and on the expense side, volatility is more of a rule than the exception. Brands in different diseases are impacted differently in terms of demand by COVID-19: some experience an increased need; others are temporarily less needed, and demand will spike again once hospitals are able to take on less urgent cases. The timing of the impact of post COVID-19 era varies by country and is hard to predict, which makes strategy and financial forecasting more of a crystal ball exercise. Finally, various payers in health care systems will have budget issues themselves, having to redirect their budgets on the fly. How the market is bouncing back and forecasting is aligning itself to the new normal will be understood better by this presentation The presentation will cover: A typical forecasting process- cross functional process How do you value / forecast a drug / company’s pipeline? What techniques to use? What are the forecasting models and best practices for early-stage assets in drug/IP commercialization processes? Who are the stakeholders in the forecasting process and how to influence them? -
Video Inspiring Raw Material Vendors to Apply Green Chemistry Innovations in Business: A Global Collaboration Initiative
With the industry shifting to become more sustainable, it is crucial for both end-users and vendors to work together when sourcing greener raw materials. Join and learn about the need for global collaboration and what you can do to adopt green chemistry practices to your business: Assessing the current state of sourcing sustainable materials, what changes are needed? Identifying the common pitfalls within the industry and what can be done to avoid them Reviewing the latest technologies available and how they can be best applied to traditional chemistry Improving communication with suppliers to ensure the right materials are sourced -
Video Case Study: Reviewing the Ti02 Challenges in Europe and why the US Market should Take Account
The European ban on the use of titanium dioxide for food use has called into question its use in pharmaceutical products because its pharmaceutical use was predicated on its approval for food use. The European ban is not based on hard science or a real safety issue, but on the precautionary principle. This presentation will help participants: Understand the real facts about TiO2 safety and the possibilities of the precautionary EU food ban being extended to pharmaceuticals Realize what this could mean to patient access to critical drugs in the EU and what the potential for a similar ban occurring in other countries Identify what a ban of TiO2 in pharma would mean to the potential for serious drug shortages in the EU and global pharmaceutical development regarding the reformulation of thousands of existing products and the associated regulatory reviews which would be needed -
Video Case Study: Putting ESG into Practice - Decarbonizing Supply Chains in the Pharma Industry
Join our case study to learn more about the current supply chain set-up across the industry and gain takeaways on how to reduce your environmental footprint: How does the PSCI make a positive impact towards the creation of sustainable supply chains? Audit Program Capability Building Special Projects -
Video The Link Between Supply Chain Integrity and Auditing During and Post Pandemic
Join our focused session, where our market expert will provide an insightful analysis of the supply chain set-up from the pandemic to now. Understand the industry learnings from Covid and what has evolved within the supply chain industry: Reviewing current developments in pharmaceutical supply chain security and data integrity Why remote pharmaceutical auditing will play a key role in supply chain set-up Comparing pharmaceutical supply chain Integrity and Material quality during and after the Pandemic (COVID-19) Joint Audit Programs® for the Pharmaceutical Industry -
Video Understanding Current Quality Challenges in Cell and Gene Therapy and How to Resolve Them
Manufacturers face unique quality challenges that can hinder progress. In this session hear about quality challenges in Cell and Gene Therapy (CGT) and learn common practices for overcoming them: The amc was formed in 2010 to focus on manufacturing when everything at the time was focused on clinical How CGTs have different challenges from a quality perspective to other biologics and small molecules Challenges over the last 13 years of the amc being in existence and how they've been overcome Current challenges o How to achieve the right quality at the right time with manufacturing readiness o Cell line development for allogeneic cell therapies o Variability of starting materials for autologous cell therapies o Analytical challenges in viral vector manufacturing -
Video GUCY2C Self-Antigen Vaccines and Adoptive Cell Therapy Development for GI Cancers
GUCY2C is a mucosally-restricted, immunologically compartmentalized self-antigen overexpressed in GI adenocarcinomas GUCY2C Vaccination in MRD (minimal residual disease) setting for recurrence prevention GUCY2C adoptive cell therapy for treatment of metastatic GI cancers -
Video Fireside Chat: Reviewing Quality and Technical Manufacturing in Allogenic Cells
Watch this fireside chat, where Stephen Chen of Tevogen Bio will go through the opportunities and challenges in Allogeneic Cells: Current quality and manufacturing procedures Reviewing short- and long-term challenges Forecasting the opportunities for Allogenic Cells -
Video The Future is Now: Best Practices and Considerations when using AI in Drug Development
AI an ever growing in pharma and is playing a prominent role in drug development. Listen to the latest developments in AI and what you need to know to utilize its potential in drug development. Join our focused session to get key takeaways from our expert: Understanding what AI can bring to your organization and identifying the areas you want to implement the technology. Reviewing regulatory considerations for successfully implementing AI into your business. Forecasting cost-effective measures to stay within budget. -
Video Partnering for Success: How USP is Working Together with Next-Generation Partners to Advance the Pharma Industry
Pharmaceutical and other healthcare innovations have extended and improved countless lives in recent decades. Today, advanced manufacturing technologies such as pharmaceutical continuous manufacturing are gaining increasing interest for their potential to serve as one more way to improve global health by bolstering medicines supply chain resilience and bringing products to market faster. However, obstacles to industry adoption remain. Recognizing the need, USP is working with partners like Phlow Corp., a U.S.-based public benefit corporation and contract development and manufacturing organization, to lower technical and knowledge barriers to advanced and enabling technologies adoption through quality-focused solutions. Working together, pharmaceutical and biotech companies are now positioned to reduce production costs, decrease development time, and lower their environmental impact through the use of advanced manufacturing technologies. -
Video Panel Discussion: Best Practices in Implementing a Successful ESG Framework
With US Pharma pledging to become net-zero by 2050, you must have the right processes. Join our focused panel where our experts will discuss the best ways to implement ESG in your organization: Defining ESG in Pharma to understand what makes it different from other industries Establishing what is needed for implementing ESG practices in your organization Reviewing common pitfalls and how to avoid them Evaluating the role of the Chief Substantiality Officer in helping the industry move in the right direction What will ESG in pharma look like in five years? What should the industry’s KPIs be? -
Video Panel Discussion: Considerations for a Successful and Prolonged Partnership with your CDMO
Companies must have the right partner to avoid potential setbacks in clinical development. To avoid disappointment, join our panel and gain expert analysis and best practices in sourcing the right CDMO for your study: Strategies for getting the most out of our CDMO partnership – mapping objectives, defined ways of working and clear communication Understanding what services and resources you need to ensure your targets are met Why mapping out deadlines is crucial to avoid untimely delays Assessing why having multiple partnerships can help manage expectations -
Video Panel Discussion: Redesigning the Supply Chain – The Road to Increased Resilience
With constraints on clinical supplies due to high demand, the industry is scrambling to find a solution. Join our focused panel, where our experts will offer insights and tips for designing a supply chain to help meet current supply needs: Investing in smarter processes and forecasting technology to avoid timely delays Establishing why having local and international suppliers will help reduce the burden of scarce materials Re-aligning your business practices to ensure you stay within budget and meet global sustainability initiatives Reviewing current technological solutions to help address supply chain constraints -
Video Panel Discussion: A National Hotspot – What’s happening with Cell and Gene Therapy in Philadelphia
Finish off your day by hearing the latest trends and excitement occurring in Philadelphia. Join our panel, where our speakers will dive into the current state of the industry and why Philadelphia is a hotspot for C>: Overview of the C> market in Philadelphia, investments, launches and developments Why Philadelphia is such a hotbed for C> How to ensure success as an emerging biotech -
Video Panel Discussion: Workplace 4.0: How to Equip your Team for Digital Transformation in Pharma
With the industry digitally evolving, you and your team need to keep up and not be left behind. Join and take part in our panel and learn how to equip and prepare yourself with the latest digital tools in pharma: Assessing what resources and training services are available Introducing practical assessments for understanding current and new hire skill sets and experience Why building skills and not compliance training will provide better insight into your team How universities can play their part by merging traditional subjects with technology -
Video Reviewing the Latest Product Development Trends in the Current Market
M&A plays a pivotal role in pharma growth as it enables companies to streamline their operations and strategies while reducing overhead costs, which can contribute to profit growth. To understand more, join our informative session, where our market experts will highlight and discuss the latest trends in the market and the expectations for the market: Discussion Points: Reviewing M&A trends in the North American Pharma Market Optimization of API process and selection of primary and secondary suppliers Drug development considerations for product development understanding scale, formulations for large volume Analytical Method optimization -
Podcast CPHI Podcast Series: An insight into the Chief Sustainability Officer
In this episode Digital Editor Lucy Chard finds out more about being the Chief Sustainability Officer in the pharmaceutical industry from Nic Hunt, Global Head of Sustainability at Nelipak Healthcare Packaging. In the podcast we discuss what it means to be in the role of a Chief Sustainability Officer, how the role is set to develop along with more established sustainability teams. The role they provide is so integral to the overall health of a pharma company, the pharma supply chain and subsequently the health of the planet. Hunt's optimism about the industry making real changes to how they work and meeting goals such as Net Zero really comes across, with the outlook for the future of sustainability in pharma a bright one. -
Podcast CPHI Podcast Series: Ursatec – celebrating 30 years of pioneering preservative free
The podcast focused on the 30 year anniversary of Ursatec in the provision of preservative-free pharmaceuticals, medical devices and cosmetics. We took a look through the Ursatec portfolio, the importance of safety and sustainability in their products, and what the future holds for the company. Ursatec values the ideology of constant improvement, whether that's keeping patients in mind, or the environment, or both. This is built into the core of the company and the products, the very state of being preservative free demonstrating this holistic vision. -
Podcast CPHI Podcast Series: Celebrating International Women’s Day by championing women’s health
As part of celebrating International Women’s Day on the 8th March 2023, we wanted to highlight the importance of supporting women’s health. Historically, women’s medicine, in relation to diseases, and sexual and reproductive rights, has been chronically overlooked in the wider field. We hope to use this podcast to open up a wider conversation about educating and empowering women so they can access the healthcare they need. -
Podcast CPHI Podcast Series: Rare Disease Day – Pharma fulfilling an unmet need
This February 28th, we have a special feature episode to mark Rare Disease Day. Rare disease is an oft overlooked field of medicine, but one which is steadily becoming more recognised. Rare diseases affect around 300 million people globally. By their nature, rare diseases are incredibly difficult to research and develop medicines for, but this just makes it all the more urgent to fulfil this unmet need for rare disease patients. In this podcast I am joined by Rachel Smith; Rachel is the Executive Director, Global Head of Rare Diseases at Parexel (MA, USA). Rachel brings more than a decade of experience in every development phase of rare disease and cell and gene therapy clinical trials to her work with Parexel’s rare disease clients. Previously, she served as Vice President of Clinical Operations, Portfolio Director of Rare Disease, and Global Head of the Cell & Gene Center of Excellence at Veristat LLC (MA, USA). She led programmes in ADA-SCID, Canavan disease, congenital adrenal hyperplasia, Fabry disease, Gaucher disease, metachromatic leukodystrophy, and Wiskott-Aldrich Syndrome. Rachel has expertise in clinical development strategy, novel and adaptive trial design, decentralised trials, and other creative solutions for rare disease products with a non-classical route to market.
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance



.png)
-file131961.png)
.png)



.png)

.png)








.png)














.png)



.png)








-file131280.jpg)

-file131355.jpg)

-file131351.jpg)

-file131353.jpg)


-file131356.jpg)





-file131359.jpg)
-file131276.jpg)
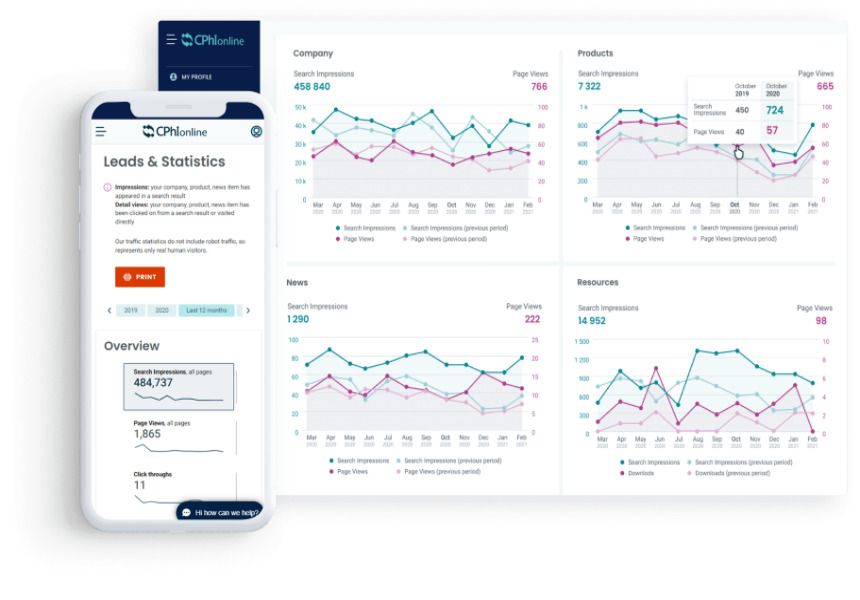



















































-file133848.png)
-file132873.png)

-file131229.png)
-file130360.png)

-file128337.jpg)
-file128336.jpg)

-file128334.jpg)


_scaled-file128330.jpg)


-file128327.jpg)






















-file128186.png)
-file128188.png)

-file128181.png)


-file128184.png)






-file128415.jpg)



-file128187.png)






-file149194.png)

-file149482.png)



-file149077.png)

-file148137.png)












-file148117.png)
-file148118.png)





-file148143.png)

%20(1)-file148148.png)
-file147144.png)
-file146349.png)
-file145456.png)


-file144426.png)
-file143226.png)
-file143227.png)