CPHI Online is the largest global marketplace in the pharma ingredients industry
-
Products551,744
-
Companies7,781
-
Articles11,636
-
Events8
-
Webinars342
Laboratory and Analytical Services
Laboratory and Analytical Services Companies (75)
Laboratory and Analytical Services News
-
News CPHI North America a ‘one stop shop’ for professional and business development
CPHI events are renowned for connecting pharma players from across the supply chain to learn, grow, and conduct business all over the world.25 Apr 2022 -
News Lonza Switzerland site to undergo expansion of microbial development capabilities
The expansion includes the installation of a pilot suite with a 50-L fermenter and automation upgrades to accelerate clinical and commercial projects17 Nov 2021 -
News SGS completes expansion of its Biosafety Centre of Excellence in Glasgow
Clients to receive accelerated quality testing and reduced turnaround times1 Nov 2021 -
News Recipharm expands its analytical services offering in India
The CDMO inaugurates its new analytical laboratory under Recipharm Analytical Solutions25 Aug 2021 -
News Skyepharma selects Veratrak platform to ensure secure data storing and sharing
Using Veratrak's platform, the CDMO will have full oversight of data, including an audit trail of data and document processes20 Jul 2021 -
News LGM Pharma launches analytical and stability testing as standalone service
The integrated CDMO rolls out services to the broader community of drug manufacturers and developers12 Jul 2021 -
News China's Raybow Pharmaceuticals invests $15.8 million to expand US facility
The company will triple its capacity and workforce at its North Carolina site to increase its global capabilities and reach23 Mar 2021 -
News Vectura expands capabilities to develop highly potent inhaled drugs
The expansion will enable the CDMO to widen the nature of inhaled drug development projects it can undertake for customers14 Dec 2020 -
News Skyepharma Analytical Services : Specialized teams
Skyepharma is a full CDMO. We have an analytical service that covers all aspects from development to manufacturing. Packaging, raw materials and finished products are all controlled by this service.17 Sep 2020 -
News Sai Life Sciences opens new Research & Technology Centre in Hyderabad
One of India's fastest growing CDMOs goes "beyond the norm" to reinvent itself as a new generation global CDMO.17 Aug 2020 -
News EuTech announces new management team additions
EuTech has recently added two strong professionals in its Management Team. Shekhar Yerramilli has joined as COO and Rick Yglesias has joined as Head of Business Development.12 Feb 2020 -
News CDMO WuXi Biologics says drug supply unaffected by coronavirus
Amid heightened global concern over the recent coronavirus outbreak, Chinese CDMO WuXi Biologics has sought to reassure customers and patients that its drug supply lines have been unaffected.30 Jan 2020 -
News Eli Lilly unveils shared innovation laboratory in San Francisco
Lilly Gateway Labs will provide biotech companies direct access to Lilly scientists and expertise.3 Dec 2019 -
News Thermo Fisher Scientific opens Customer Solution Center in China
The new center will provide expertise in critical analytical processes and specialized workflows.20 Nov 2019 -
News Method Development, Verification & Validation of NDMA & other impurities
Accuprec Research Labs Pvt. Ltdhas successfully doneMethod Development, Verification & Validation of NDMA impurities in Ranitidine through LCMS/MS [API & Finished Dosage Form] as per USFDA method.6 Nov 2019 -
News The Leader in Spray Drying
Hovione continues to lead in Spray Drying, thanks to the largest capacity, the most experienced team of scientists that keep coming up with innovative solutions and the best-scale up science.29 Oct 2019 -
News OneVision: Creating a Global Standardized Laboratory Network for SGS Life Sciences
SGS launches OneVision, a global digitalization initiative to standardize record-keeping procedures across all Life Sciences’ testing laboratories.
27 Aug 2019 -
News How real-world data can revolutionise drug development
Utilising the current data environment and its data pools for the benefit of advancing research.9 Aug 2019 -
News MedPharm expands relationship with Novan on formulation science
New agreement allows MedPharm to react quickly to changes in requirements and makes full use of its comprehensive services for de-risking Novan’s product development.23 Jul 2019
Laboratory and Analytical Services Products (180)
-
Product Analytical Services
With 55+ years of experience, our expert teams develop 1,000+ analytical methods and validate 250+ methods annually. Drawing upon an extensive range of analytical technology, combined with a wealth of analytical knowledge, we can add real value to your drug development and commercialisation programs....
-
Product FINISHED FORMULATION , PHARMACEUTICALS
We are Manufacturing Solid/ liquid orals, Topicals, Sterile preparations (SVP/ LVP/ Eye drops ) in all therapeutic segments .
Global Pharma Healthcare has a wide range of Therapeutic segments
Cardiovascular, Oral Diabetic, Respiratory drugs, Neuro – Psychiatric, Genito – U...
-
Product Outsourcing
Patheon by Thermo Fisher Scientific has a broad manufacturing platform for pharmaceutical and biologic products which provides sustainable solutions for mammalian cell-based and microbial-based manufacturing, green chemistry R&D and manufacturing technologies, and finished dosage production of biopharm...
-
Product R&D Services
Our R&D Services are categorized into 8 streams: • Material Science Services
• Metal and Organic Scavenging Screening Services
• Organic Synthesis Services
• Catalysis Services
• Process Services
• Chromatography and Purification Services
• Method D...
-
Product Foreign Particles
Merieux NutriSciences has developed various strategies and complementary approaches for the identification of foreign visible and subvisible particles, thanks to sophisticated instrumentation combined with a pool of experts in different fields.Download our brochure: https://bit.ly/48HKnVs
-
Product Intertek Pharmaceutical Services
Intertek's pharmaceutical contract laboratory services, regulatory assistance, and supply chain assurance deliver quality and safety to meet your unique pharma and biopharma outsourcing needs. We bring quality and safety to life!
With our pharmaceutical experts working with you at every ...
-
Product Solid state characterization services
cGMP services include:- Particle size by laser diffraction on powders, sprays/aerosols, liquid dispersions - Particle size, shape & Chemical identification in powder mixtures, creams, liquids by MDRS- BET surface area and porosity- Thermal analysis DSC
Other services include:- Density (...
-
Product Disease Activity Models/Tissue Culture
MedPharm has developed proprietary models relevant for major disease areas such as psoriasis, atopic dermatitis, onychomycosis and continue to develop new ones through our dedicated innovation group. These sophisticated ex vivo experiments provide additional confidence to clients and their in...
-
Product Formulation of lyophilized products
A correct formulation means greater stability of the active ingredient and provides protection against the stress suffered during the lyophilization process itself.
The selection of the appropriate excipients is really important in the development of a freeze-dried product.
-
Product Certification of Suitability (CEP)
Book a private, one-to-one session with the Certification team to discuss issues such as the CEP procedure, CEP applications or inspections, please complete the online form and the team will be in contact regarding an appointment.
Please book using this url:
https://www.edqm...
-
Product SANAL® SQ Pharmaceutical Sodium Chloride (API)
SANAL® SQ Sodium Chloride Pharmaceutical Quality is our chemically pure product which is suitable for all pharmaceutical applications. Additionally this product is suitable for laboratory testing and chemical applications in manufacturing procees - This product comply with American Chemical Socie...
-
Product Analytical Techniques
Comprehensive analysis during formulation development and GMP manufacture is vital to ensure that your drug has optimal delivery properties and stability profile as well as supporting your regulatory submissions.
-
Product Regulatory Testing and Research based services for Pharmaceuticals, Chemicals, Phytochemicals, Herbal Formulations, Food and Medical devices
1) Analytical Testing Of Pharmaceuticals & Cosmetics2) Biotechnological Services
3) Microbiological Services
4) Bio - Compatibility Studies of Medical Devices(As per ISO 10993USFDA and MHLW Guideline)
5) Preclinical & Toxicological Services
6) Phytochemical & AYUSH Testing Se...
-
Product Method Development, Validation and Transfer
Kymos Pharma Services, S.L. offers wide range of chemistry, manufacturing & control analytical services in medicinal chemistry which includes method development, validation and transfer. Kymos has comprehensive knowledge in the development and validation of analytical proprietary and non-proprietary method...
-
Product Drug Product Development
We support our clients from early-stage research and preclinical phase to and beyond market approval by providing comprehensive development services, including
• Developability assessment • Pre-formulation screening • Formulation development • Lyophilization process development • Forced d...
-
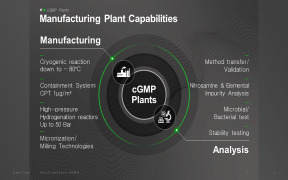
Product cGMP Manufacturing
1. Manufacturing facilities ensure: 1) Production flexibility at various scale 2) Total Production Capacity: 726 m3 including pilot production 3) Potent compound manufacturing (OEL Category 3A, CPT 1 Mug/m3) 2. Various Analytical Capabilities: 1) cGMP QC lab with HPLC, UPLC, GC, LC-MS, GC-MS, NMR, X...
-
Product Laser Micromachining Services & Systems
From pinholes in stainless steel discs to complex arrays in semiconductor guide plates, we provide a wide range of ultra-high precision laser drilling, cutting, milling, scribing and ablation contract services (and systems) for numerous applications in industry & academia. With over 45 years of experie...
-
Product Services - Lab analysis
We offer the quality standards that we apply to our own environment, machines, solutions and processes as services to our customers as well.
We make continuous investments in our specialist expertise. Quality assurance is the focus of our work. To ensure that we can meet your requirements r...
-
Product QACS Pharmaceutical services
QACS Lab is GMP/GLP certified and provides contract laboratory testing for Pharmaceuticals. Methodologies stay in accordance with EMA & FDA. QACS is equipped with Microbiological - Chemical - Molecular - Packaging laboratory premises to provide variety of Pharmaceutical testing solutions &...
-
Product J.T.Baker® Viral Inactivation Solution
J.T.Baker® Viral Inactivation Solution Biotech Reagent is a highly effective, ready-to-use product, specifically developed for detergent-based viral inactivation in biopharmaceutical operations and plasma derived therapy processes. J.T.Baker® Viral Inactivation Solution is formulated with non-ionic deterge...
-
Product Analytical Services
Our Analytical Services encompass critical aspects of drug development and manufacturing, beginning with Analytical Testing to ensure product quality, employing advanced instrumentation and experienced teams to facilitate compliance. We excel in Method Development & Validation, utilizing HPLC and UHPLC...
-
Product Core Technologies and Services
• API / GMP Manufacturing • Rapid Process Development, Flawless Upscaling, and Economy of Scale-Production • Simulated-Moving Bed (SMB) Chromatography • Heterocyclic, Hazardous and Malodorous Chemistries • Organometallic and Cryogenic Chemistry • Transition-Metal Catalysis • High-Pressur...
-
Product Solid & Liquid Dose Drug Manufacturing & Development
From OTC and Rx to diagnostics and dietary supplements, Avéma manufactures a full range of solid and liquid dose products, all manufactured under strict FDA guidelines and cGMP compliance. With an ever-growing portfolio of innovative formulas and a diverse mix of state-of-the-art equipment, our offerings a...
-
Product Syringe and Vial Filling for Clinical and Commerical
Lifecore’s experts have gained the know-how to build processes around your requirements for smooth scale-up and high-quality manufacturing.
Whether we’re your primary partner or a secondary source for manufacturing, we ensure you're fully satisfied with the final produ...
-
Product Impurity Synthesis
Pharmaffiliates Analytics & Synthetics (P) Ltd, is an integrated CRO (Contract Reserach Organisition) established in year 2001. Presently our group consists of more than 130 Scientists with 3 R&D Centres offering its expertise in Custom Synthesis, Impurity synthesis, Isotoped lebelled compound...
-
Product Analysis Service
Lomapharm GmbH provides wide range of services including analysis service. It involves chemical and physical analysis of raw materials, intermediate and finished products according to national and international specifications as well as customer-specific information (microbiological purity, stability t...
-
Product VialArch
The Gasporox sensor module VialArch offers non-destructive Headspace Gas Analysis to be integrated directly onto the production line or into an inspection machine.
This unique laser-based solution is installed on the production line for 100% quality inspection and container closure ...
-
Product Analytical Development
BioDuro-Sundia’s Analytical Testing team offers high quality analytical services including method development and validation, qualification of reference standards, testing and release studies, stability studies, and CMC dossier preparation services. ...
-
Product Dissolution Testing - R&D services
For us dissolution testing is never a routine test but an essential tool in pharmaceutical formulation development to:
• evaluate the physicochemical properties of drug candidates to select the most appropriate solid form for further development (pre-formulation)
• compare pro...
-
Product PEKUWhite - Cleanroom packaging
Cleanroom packaging:- flat film
- tubes
- half tubes
- cuttings
- bottom seam bags
- side gusseted bags
-
Product Analytical Services
The complete integration of analytical sciences into the process of API development is fundamentally understood at CARBOGEN AMCIS. Our scientists, technique ranges, systems (including a fully validated LIMS) and procedures facilitate a full understanding of the unique characteristics of the complex molecul...
-
Product Research & Development
Discovery - Biology, Lead Optimization, Libraries, Synthetic & Medicinal ChemistryDevelopment - Chemical Process R&D, Fermentation, Formulation Development, High Potency, Kilo Lab & Small Scale Manufacturing, Lipid Nanoparticle, Method Development/Material Science, Rare/Orphan, Separation Scien...
-
Product Highly Potent Pharmaceutical Principles
Olon relies on one of the most extensive expertise in handling highly potent pharmaceutical principles at global level, has raised the level of containment up to the actual standards and very large scale putting in place a set of primary and secondary containment designed to avoid the active substances...
-
Product Analytical Development and expertise
Our analytical experienced team support the pharmaceutical development during every phase of the development process. Analytical development transfer and validation, characterization capabilities, stability analysis, ICH Q3D (residuals solvent risk assesment), ICHQ3C (elemental impurities risk assesment...
-
Product Pharmacopea Analysis
Neotron SpA provides wide range of analytical control according to EP, USP, JP, BP, CHP.
Contact us to ask for tests in relation to your monograph of interest.
-
Product Analytical Services - Inhaled Products
With extensive, state-of-the-art analytical testing facilities and equipment, our expert teams are able to develop and validate the methodologies required to characterise inhaled delivery platforms, especially DPI, pMDI and nebulised products.
To ensure seamless support for your development...
-
Product Analytical development
Alcami offers a fully-integrated analytical method development, method validation, and testing solution with a full complement of advanced analytical and information technologies. From Alcami’s electronic laboratory notebook (ELN) data collection system to its integration with our formulation development, ...
-
Product NETWORKING OF Excellence and innovation of the health sector. Made in France.
A team of experts serving the network.
POLEPHARMA stimulates industrial and economic development of actors of the French first pharmaceutical sector with actions that promote competitiveness, innovation and attractiveness to the advantage of the job market.

-
Product Quality Control
Our quality control department offers a broad range of services from incoming goods testing, in process controls to release of final product and stability testing. Our workflow ensures that all products are safe for market release and simultaneously functions as a feedback tool to help in the optimizat...
-
Product Analytical Services
ChemCon’s analytics team takes care of your inquiries if you are looking for ICH-compliant quality control, GMP validation, release analysis, impurity determination, reference standards or answers to other analytical queries. Close, outcome-oriented communication is the key to a successful partnership:...
-
Product Wasdell Manufacturing
Wasdell’s specialised pharmaceutical manufacturing facilities in the North East and the East Midlands are MHRA approved for the manufacture of non-sterile oral liquids with the capability and capacity to extend its services to other dosage forms. We can support the manufacture and packaging for cl...
-
Product Netpharmalab Consulting Services
Contract Research Laboratory. Analytical Services (physicochemical analysis, microbiological analysis and stability studies) and EU Import Lab.
-
Product Analytical Services
Mabion offers a extensive suite of Analytical Services to ensure safety, efficacy and quality of your biologic product. Our portfolio includes development, optimization and validation of analytical methods, as well as stability studies, GMP release testing and in-depth drug characterization. In addition to...
-
Product CMC analytical support
Leveraging state-of-the-art technology and rigorous scientific methodologies, we provide a full spectrum of CMC analytical support, including:
· Method Development and Validation
· &nb...
-
Product Analytical Services
From a drug product’s first raw materials to its release-ready batches, our experts rigorously evaluate and verify a product’s quality at key steps throughout the manufacturing process. Analytical services include material testing to verify product quality from the early stages of development, cGMP-com...
-
Product CONSULTING SERVICES ON PARTICLE CHARACTERIZATION
Consulting services which create value by combining our expert scientific background with highly qualified staff, equipments and facilities, to deliver advanced particle characterization studies such as:
- Development validation and transfer of analytical methods
- Comparative...
-
Product Contract Manufacturing and Development, from Clinic to Commercial Scale
CoreRx provides comprehensive drug product pre-formulation, formulation, analytical, and GMP manufacturing and packaging solutions, enabling our partners to meet their drug program goals. CoreRx delivers these solutions from it’s development and manufacturing campus in Clearwater, Florida and it’s...
-
Product Licensing for Partners
PharmaMatch represents companies from all over the world and we have always shown ourselves to be a reliable and trustworthy partner. Our sales team has an excellent network and they are continually assessing the market per product to find the best match. Our partners can expect our Sales team to appro...
-
Product Packaging and Supply Chain Solutions
Tjoapack's packaging and supply chain solutions help our customers to make the world a healthier place. Our expert team work to facilitate your life sciences research, production and the delivery of medicines to patients across the globe.
We offer: - Packaging solutions for; oral solid blisters a...
-
Product Raw Material Services
Supply of Active Pharmaceutical Ingredients (APIs) and excipients to pharmaceutical manufacturers
Services include:
Purchase of active pharmaceutical substances, excipients, primary packaging materials and reagents Analytical services to ensure identity and compliance
I...
-
Product Laboratory & Analytics
Samples, bulk products or drugs: Our experts analyze your product and evaluate whether it meets all individual and regulatory requirements.
Among others our modern laboratory is equipped with:
• HPLC-systems • Dissolution testing • UV/Vis- and atomic absorption spec...
-
Product Microbiological Analysis
Oasis Test House caters to the microbiological requirements for its clients serving them with the below analysis. • Preservative Efficacy Analysis • Sterility Test • Sterility Validation • Microbial Limit Test Validation • Microbial count and pathogens • Bioburden testing • Antibiotics assay • Vitam...
-
Product Analytical method development and Validation
Stability indicating assay Related substances Residual Genotoxic impurities Residual solvents Residual metals Dissolution studies Content uniformity
-
Product Cgmp Quality Control
PolyCrystalLine offers a wide range of analytical services which includes cgmp quality control. Its provide a full complement of state-of-the-art gmp analytical testing resources and expertise to support cgmp quality control testing for raw materials, active pharmaceutical ingredients (api) and intermediat...
-
Product Analytical Development And Validation Services
Pharma-Data sa offers wide range of services which includes analytical development and validation services. It includes justification of specifications, drug product release testing, method transfer, validation of microbiological methods (sterility, bet etc), filter validation (for sterile products – leach...
-
Product In-Vitro Release & Permeation Studies
DALPL Pharma Division performs IVRT & IVPT Studies using Logan Instrument
-
Product Instrument-Based Analytics
The pharma laboratories within the Tentamus Group are equipped with state of the art analytical instruments offering an unparalleled array of analytical and bioanalytical services. The range covers simple Pharmacopeia methods (e.g. Ph.Eur, USP) to very complex analyses of biomolecules. We offer all kin...
-
Product API Services, Chemical Development & Manufacture
Almac’s strength in API development and manufacture is proven by being the partner of choice for many pharma and biotech companies seeking integrated drug development solutions from molecule to market.
Our technical expertise and extensive facilities enable us to offer integrated API contract ma...
-
Product 14C Radiolabelling
Almac has extensive experience in the synthesis and analysis of stable (non radio-active) and 14C isotope labelled compounds – from drug discovery through to launch – we can label any compound at any stage, including small molecules, peptides and larger bio molecules.
Our custom radiolabell...
-
Product Physical Sciences - Preformulation & Solid State Services
Almac's Physical Sciences groups' comprehensive range of services and expertise will help strengthen the success of your product. Identifying and consistently producing a drug in its optimal physical form is vital to any successful drug development programme. It also avoids the need for bridging toxicologi...
-
Product Biologic drug substance CDMO services
From pre-clinical development to commercial supply, Patheon by Thermo Fisher Scientific is an industry leader in the development and manufacture of mammalian cell culture drug substances. Patheon offers biotech and pharmaceutical companies the ability to pursue opportunities around the globe with a fully i...
-
Product Sterile drug product CDMO services
Thermo Fisher Scientific's flexible aseptic manufacturing and sterile fill finish solutions for your molecule’s unique needs and challenges will enable success in early development, late-phase, and commercial manufacturing.
Thermo Fisher offers extensive sterile product development and commercial ...
-
Product Mérieux NutriSciences | Pharma & HealthCare complete services
Download our brochure: https://bit.ly/4dzryoA
Our complete range of Pharma services include: • Quality controls • Investigation studies • Absorption studies • R&D and validation activities • Stability & storage • Cleaning and disinfectants validation • Environmental monitoring
-
Product Merieux NutriSciences | Absorption and efficacy in vitro studies
Download our brochure: https://bit.ly/4dzryoA
In vitro models mimicking different biological barriers can be adopted to perform absorption studies and evaluate efficacy of products, ingredients or new formulations before to proceed with the next step evaluation (e.g. clinical trial).
q...
-
Product Merieux NutriSciences | Nitrosamines analysis on pharmaceutical products
Download our brochure: https://bit.ly/4dzryoA
Thanks to the long-standing experience, Mérieux NutriSciences has been developing various strategies and approaches for the determination of nitrosamines residues in different matrices through sophisticated mass spectrometry combined with a pool of ex...
-
Product Merieux NutriSciences | Extractables and Leachables studies
Download our brochure: https://bit.ly/4dzryoA
Pharmaceutical packaging can release chemicals into the drug product that can not only impair its effectiveness, but also be harmful to the patient. Similarly, medical devices can undergo leaching processes during their use that could negatively affec...
-
Product Merieux NutriSciences | Medical devices services
Download our brochure: https://bit.ly/4dzryoA
Thanks to a valuable pool of experts, Mérieux NutriSciences supports you in the development of the testing plan to determine which studies are necessary to ensure that the device is safe and effective, meeting the essential requirements for affi...
-
Product Liquid, cream & gel forms physical characterization services
cGMP services include:- Particle size by laser diffraction on sprays/aerosols and liquid dispersions
- Particle size, shape & Chemical identification in gels, creams, liquids by MDRS
- Thermal analysis DSC
- colloids, liposomes and nanoparticle size distribution and zetapotential by DLS ...
-
Product EVs extracellular vesicles characterization services
Alfatestlab is equipped with the latest technology platform to provide rapid, reliable and accurate characterization of EVs following MISEV 2023 guidelines. Based on Nanoparticle Tracking Analysis (NTA - Nanosight) and Single-Vescicle Multiparametric Analysis (Leprechaun), our services for EVs of...
-
Product Aerosol & Spray particle size distribution analytical services
We provide cGMP analysis of sprays and aerosols: droplets particle size distribution over time of sprays and aerosols, using laser diffrcation technique (Spraytec by Malvern Panalytical)
-
Product LNPs production services by microfluidics
We offer method development services for the production of lipid nanoparticles LNPs using the microfluidics platform Sunshine from Unchained Labs.

-
Product Lyophilization Process Development (Quality By Design approach)
With the aim of obtaining a product that meets the critical quality attributes, but with the most optimized freeze-drying process.
Our focus is the total control of the process. We develop freeze-drying processes in a previously defined design space: Quality By Design approach.
-
Product European Pharmacopoeia Supplements 11.6 to 11.8
The 2025 subscriptions to the European Pharmacopoeia (Ph. Eur.), including the Supplements 11.6 to 11.8, are now available for purchase in the webstore. Two subscription formats are available: print and online versions.
-
Product Nitrosamine Detection
We provide Method Development and Method Validation for new analytical methods or Method Transfer for Routine Testing according to existing validated methods for raw materials and final products. QACS is GMP/GLP certified.
• performed with UPL...
-
Product Leachables Extractables
QACS tests for organic and inorganic components, Volatile, Nonvolatile residues and perform Elemental impurities analysis. Packaging Migration/Compatibility studies are also provided. Studies are performed with state of the art equipment.
▶️ [email protected] +30 21...
-
Product Stability Studies
QACS Labs offer drug development services. Pharmaceutical testing includes pharmaceutical stability testing, storage stability services and custom solutions based on international standards. We test API’s, raw materials and final products in recommended environmental storage conditions such as Temperature,...
-
Product Method Validation & Transfer
Pharmaceutical testing services during and after drug development from QACS ensure GMP compliance and product safety. Method validation studies aim to demonstrate analytical suitability of procedures according to their intended use/purpose. QACS transfer method protocols ensure, that results generated by s...
-
Product J.T.Baker® Direct Dispense packaging system
Enhance the effectiveness of your products and streamline your biopharmaceutical manufacturing processes with the J.T.Baker® Direct Dispense packaging system. This novel packaging platform makes it easy to deliver powdered performance materials and excipients directly to your process, whether you are using...
-
Product Avantor OmniTop Sample Tubes® adjustable volume sampling system (AVSS)
AVSS helps our customers overcome complex scale-up challenges, particularly in sensitive applications like cell and gene therapy production and mAbs downstream processing. The single-use sampling device, with reusable adjustment tool and rack, can easily be used stand alone or configured in a manifold ...
-
Product Avantor® Magnetic Mixing System
The Magnetic Mixing Bag offers a sterile, single-use mixing container designed to maximise mixing performance while minimising disruptive shear. The 5-bladed impeller is floating freely due to magnetic forces minimising risk of particle generation and ensuring a safe and homogenous mixing of product. A...
-
Product MasterSense™ Smart Peristaltic Pumps
NEW integrated sensor technology, intuitive touchscreen and MasterflexLive® remote monitoring for the biopharma industry of tomorrow
-
Product Reference Standards
Pharmaffiliates is Supplying all the Pharmacopoeial Reference Standards (i.e. USP, EP, BP, IP, JP).Apart from this we offer Pesticide Standards, Phyto-chemical Standards, Food & Environmental Standards, etc.
For more details please send your enquiry on [email protected]
-
Product Analytical research & development
Our analytical experts know what it takes to bring a pharmaceutical product to the market, from discovery, feasibility to GMP. Having worked with more than 100 unique molecules and even more formulations, we may state that we have faced the most difficult CMC challenges that are involved.
...
-
Product Excipient Quality Testing and Selection Services
Excipient testing, composition and variabilityExcipients are either natural / naturally derived or synthetic / semi-synthetic. In all cases the exipients are obtained through chemical processing of a raw material source that usually has an animal, vegetable or mineral origin.
...
-
Product Fermentation
Relying on an experience gained over more than 50 years, OLON represent one of the most extensive know how of microbial fermentation in Europe. The Group, global leader in biomanufacturing, has two Biotechnology Centres located in Italy and is one of the first companies in Italy producing via microbial ...
-
Product Research & Development
Avéma offers formulation, process and analytical development support of new or existing products using the most current and effective active ingredients and delivery systems. Our team of scientists bring decades of pre-formulation development expertise to your products, combined with the knowledge of how t...
-
Product Elemental Impurities
In light of the growing interest of the pharmaceutical world on the issue of determining Elemental Impurities in accordance with tables 1, 2a, 2b and 3 of the ICHQ3D, Neotron Pharma laboratory, thanks to its decades of experience in the analysis of metals, has developed a series of analytical proposals to ...
-
Product Extractables & Leachables
There are many contaminants that could be released inside a drug during the production process or by contact with the packaging material. Neotron Pharma will be able to support you from the study of Extractables, to the toxicological evaluation up to the control of the Leachables. What distinguishes us is ...
-
Product Nitrosamine impurities
Neotron Pharma, on request of several customers, has provided an effective methodological approach for Nitrosamine alerts management to support the pharmaceutical industry. Currently the laboratory is able to perform screening and validation activities for more than 11 Nitrosamine residues in API, FP and e...
-
Product Pyrrolizidine Alkaloids
Neotron Pharma has an HPLC-MSMS method for the determination of 28 pyrrolizidine alkaloids on different matrices. We currently collaborate with numerous herbals producers for these routine checks for both the Pharma and Food markets
-
Product Integrated offerings
Alcami is a Us-based, contract development, testing, and manufacturing organization for pharma and biotech companies. Our goal is to support our clients in making their projects go from potential to reality day-after-day.
Core Capabilities:Sterile-fill finish development and manufacturingO...
-
Product Fine Chemicals
SPECIAL TECHNOLOGIES AND REACTIONS
Special Technologies
• High-pressure reactions up to 64 bar: H2, CO, NH3, Amines, CO2 • Cryogenic: -80°C • Supercritical Fluid Chromatography (SFC), Simulated Moving Bed Chromatography (SMB) • Ultra low vacuum distillation • ...
-
Product Regulatory Support
Our experienced regulatory team has a good understanding of the regulatory framework in every region. We’ve obtained registrations globally through our clients. Our team provides clients with quality dossiers compiled in accordance with the latest guidelines, which result in numerous successful registr...
-
Product API Sourcing
PharmaMatch has an extensive network of API Active Pharmaceutical Ingredients (API) suppliers, ensuring our clients are matched with the right partner based on quality, price and regular supply. We can also provide third party audits or pre-audits for our clients to ensure the facilities are up to st...
-
Product Contract Manufacturing Organisations
PharmaMatch has global contacts with contract manufacturing organisations (CMO’s). These state of the art CMOs are EU and/or US approved and follow the GMP standards of production. As clients demand high quality standards and a reliable supply chain with the best commercial conditions, the CMO par...
-
Product Lab Services
Through one of our partners, we can provide comprehensive analytical support during the development of drug formulations. By combining innovative approaches, modern instruments and advanced knowledge in the field of analytical science we are able to achieve high performance in terms of selectivi...
-
Product Laboratory Services
Curida offers Lab services1 chemical lab
3 microbiological labs, including sterility lab and lab for test organisms
Main services:
Quality Control
Method development and validation
Stability testing
Contract laboratory
-
Product Dossier Writing Service
Pharma-Data sa offers wide range of services which includes dossier writing service. It includes ectd dossier preparation, dcp/mrp submission and follow up, life cycle dossier management. Contact us for more information.
-
Product Microbiology
Microbiological tests are mandatory for non-sterile and sterile pharmaceutical products as well as medical devices. Tentamus Pharma Labs provide all standard test methods in this field and support your products in development or commercial phases. For non-sterile products, microbial enumeration tests (...
-
Product Pharmaceutical Products
Bringing a pharmaceutical product to the market entails many complex, time consuming and expensive activities. Analytical expertise is a cornerstone in this development process. Tentamus Pharma Labs can support our customers throught the entirety of the development process and commercialization by prov...
-
Product Medical Devices
The EU Medical Device Regulation (MDR) requires a more stringent characterization of products in the Medical Device field. The MDR is madatory for all new products and existing products have to comply by May 26th 2020. Tentamus Pharma Labs are equipped to support you in fulfilling MDR requirements. Bio...
-
Product Biomolecules and ATMPs
Biomolecules like antibodies have revolutionized the pharmaceutical industry during the last decade and new products (e.g. Advanced Therapy Medicinal Products – ATMPs) are evolving in clinical studies and on the market. Tentamus Pharma Labs are among the leaders of analytical support for these product ...
-
Product Preclinical Laboratory Services
MYER's team counts with 30+ years of experience conducting nonclinical efficacy in vitro and in vivo experiments. Small molecules, biologics, devices, diagnostics. Contact us to get a quote!
-
Product Nitrosamines - QC / Development / Validation
LC-MS/MS methods are nowadays commonly used for nitrosamines analysis in all types of materials, since it allows analyte-specific detection based on both retention time and structurally specific fragmentation information in conjunction with high sensitivity.
After the establishment of the ni...
-
Product Analytical Services
Physico-Chemical testing
- General identification (TLC, HPLC)
- Assay (UV/VIS, HPLC-UV, GC-FID, LC-MS/MS) and dosage uniformity
- Related substances identification and quantification (HPLC-UV, GC-FID, LC-MS/MS)
- Physical determinations (pH, viscosity, density)
- Moisture...
-
Product Analyses of your chitin, chitosan or chitosan derivatives
Do you need further analyses for your ordered products? We would also love to analyse your own chitin, chitosan or chitosan derivative.
In addition to our standard analyses like degree of deacetylation, viscosity, ash content and content of heavy metals, HMC+ offers different other analyses. ...
-
Product Quality control laboratory
Batch testing and GMP EU batch release for commertialization of pharmaceutical products within the European market.
Import and batch certification
Physicochemical analysis: • General Identification Test (UV, IR, TLC, HPLC, colourants) • Physical Tests (pH, viscosity, hardness, disintegrati...
-
Product In Vitro Dissolution and Release Testing - R&D Services
Support formulation development with our experience in drug release testing. We offer a range of methods to:
• Evaluate the physicochemical properties of drug candidates to select the most suitable dosage form for further development • Elucidate drug release mechanisms, such as diffusion, erosion, etc...
-
Product Analytical Testing
Glatt Pharmaceutical Services develops and produces solid pharmaceutical dosage forms. Our focus lies on multiparticulate systems such as pellets, micropellets and granules. Whether you are looking for optimal bioavailablity or taste masking, improved solubility or stabilization of the dosage form, we have...
-
Product Analysis of biocides for registration in BPR
Analysis of active ingredients and impurities.Accelerated and long-term storage 5-patch analysis
-
Product Analysis of coatings of medicine products
Analysis of EG silver coated products
e.g. implant meshes,
nails and discs, wound dressing,
-
Product Trace Metals Testing and Elemental Analysis
Our experienced analysts apply a strategic approach to sample preparation for difficult samples and a range of analytical technology, relevant to your needs, including inductively coupled plasma – mass spectrometry (ICP-MS) or inductively coupled plasma – optical emission spectroscopy (ICP-OES) or ion chro...
-
Product GMP and CMC Laboratory Services
We provide regulatory-driven, phase-appropriate laboratory services in support of CMC programs seeing you through pre-formulation, formulation and product release. Our capabilities include centers of excellence for method development and validation, analysis, stability studies,...
-
Product Stability Testing
With a network of ICH stability storage facilities in the UK, US and Australia, we offer an extensive capacity and a range of conditions including climatic walk-in chambers, cabinets and refrigerated as well as freezer storage, covering all ICH stability conditions. These are fully controlle...
-
Product Analitycal Method Development
• Method development, optimization, validation and verification. • High skill operating with HPLC/UPLC • Cost and time focused • Working according to GLP standards • R&D Support

-
Product Other analytical and related services
• Stability Studies Design and Execution (chambers available) • Probiotic testing • Degradation studies • Animal Studies • Cell Research • Market Research • Supplier Qualification

-
Product Precious Metal Recovery Service
Mastermelt recover precious metal from heterogenous and homogeneous catalyst waste streams. We specialise in difficult material to find new routes to recover metal from complex waste streams.

-
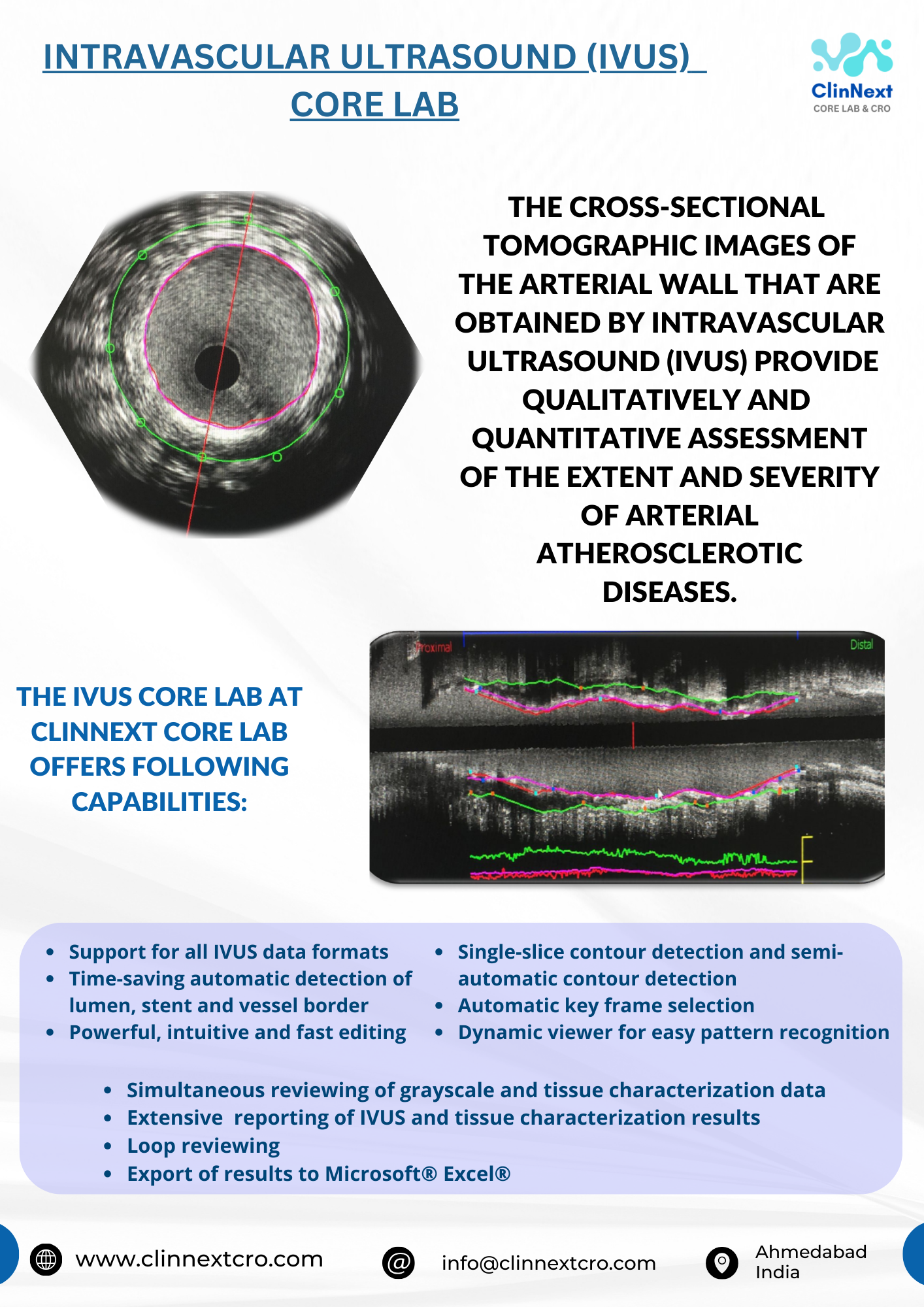
Product CORE Lab Services
ClinNext is established with the objective of serving the need of the global pharmaceutical, medical devices & clinical research industry in the areas of central reading of QCA, Echo, Q-IVUS, QVA, QFR, ECG, OCT, CT & MRI images

-
Product Analytical Services
• Analytical Method development • Validation, Co-validation, Transfer • Quality control • Stability studies (ICH/VICH) • Cleaning validation
-
Product LVA Pharma
We offer our international customers specialized services which are tailored to their requirements, from early stage of development to approval and quality control of their products. - LVA Pharma and EL spol. are international providers of analytical services - 2 sites in Austria and Slovakia - more than 2...
-
Product LVA Cosmetics
Laboratory Analysis and Expertise “ one-stop laboratory services
Cosmetics and personal care products are subject to accurately defined guidelines regarding marketability and claims on benefits and effects. In any case, manufacturers of cosmetics must ensure the safety of their products.
We work to...
-
Product Clinical BA/BE Studies
Our clinical pharmacology unit comprises of: • 3 spacious independent Clinics with dedicated recreation and dining area having a total bed capacity of 100. • In-house 2 bed ICU and a tie up with a multi-specialty hospital for handling medical emergencies. • Emergency Alarm on each bed side /Exclusive Ele...
-
Product Laboratory Services
Laboratory Services
Our 30 years of experience and our continual quest for improvement ensure we deliver analytical services of the highest level for active ingredients, excipients, medicinal products and drug-related products. In addition to developing and validating analytical methods, batch-...
-
Product LVA Service
As a Part of the CONSCIO GROUP, LVA is your partner for the areas of food and feed, cosmetics, environment and pharmaceuticals.
This includes, in particular:
• food at every stage of the production chain
• packaging and basic commodities
• hygiene products (diapers, napkins, etc.)
...
-
Product RESEARCH AND DEVELOPMENT
We offer research and development services at all stages of the production of capsules, from idea to finished product for medicinal products, medical devices, food supplements, cosmetics and veterinary products.
We offer our clients a proper identification of their needs and requirements, r...
-
Product Analysis
Excerpt from the range of services offered by our analytical department: • physico-chemical testing • chromatography • spectroscopy/spectrometry • determination of TOC/TNb (water analysis/cleaning validation) • particle measurement
• elemental analysis
• dissolution testing
• pho...
-
Product Microbiology
Excerpt from the range of services offered by our microbiological department: • microbial contamination testing (bioburden) • identification of microorganisms • sterility testing • biological indicator testing • bacterial endotoxin testing (LAL) • assay of antibiotics • efficacy of preserva...
-
Product Bioanalytics/Molecular Biology
Excerpt from the range of services offered by our bioanalytical/molecular biological department:
• determination of mycoplasma • biological contamination testing • determination of content and identity of nucleic acids • determination of biological activity
• protein/antibo...
-
Product Solid Form Screening
We know that the needs in early development are different from the needs in late development and we tailor our screening approach based on information from the client and our experience with a wide range of compounds. At Crystal Pharmatech, we utilize a unique screening approach that co...
-
Product Crystallization
Crystallization is a critical unit operation in API manufacturing for both final product and intermediates. Desired product quality attributes can be obtained economically with high efficiency through well-designed and controlled crystallization processes.
-
Product Chiral Separation by Crystallization
More than 70% of drug candidates worldwide are chiral. Typically, for chiral API, only one enantiomer or diasteromer is biologically active or desirable. Therefore, the production of enantiopure compounds or diastereomers is imperative. In the production of small molecule drugs with desired chirality, sepa...
-
Product Analytical Method Develpment and Validation
Providing Analytical method development, validation and stability study from Phase I-III to commercial prodcution and registration.
-
Product Clinical Supply Manufacturing
NCEs • Phase I-III formulation process development and clinical supply manufacturing Generic drugs
• BE sample manufacturing • Process Optimization
-
Product Delivery systems / formulations for drugs with low water solubility
Develop delivery systems for drugs with low water solubility
-
Product Liposome
Development of liposome delivery systems for both small molecule drugs and biologics
-
Product GC 456 System
SCION 456 GC Gas Chromatograph When your requirements demand more functionality or you want room to upgrade, the SCION 456 easily meets your need. Supporting three injector and four detector positions (including the mass spectrometer) with full independent access, no other gas chromatograph system pr...
-
Product Batch Testing & EU Batch Release / Certification
QP Declaration
QPLab will evaluate the information regarding active substances manufacturing sites of the product, to assess the EU GMP compliance and issuing the QP Declaration.
QP Batch Certification SetupIn this step QPLab will have a complete oversight ...
-
Product M&A
With over 29 years of experience and operating across all key life science sectors, PharmaVentures offers deep sector expertise to support your M&A and funding transactions.
We support our clients with strategic advice and full end-to-end process management capabilities to achieve best o...
-
Product MODDE® | Explore. Improve. Advance!
MODDE® offers Design of Experiments Software that Accelerates Progress.
Design of Experiments (DOE) is the fastest and most cost-efficient way to design effective experiments, increase productivity, and tackle your toughest challenges in development and manufacturing. With MODDE®&n...
-
Product Stability Storage Testing
As the evaluation of stability is a critical aspect of drug substance and drug product testing, Integrated Analytical Laboratories offers Stability Services and Sample Testing to our partners. Whether you have predetermined stability protocols or require IAL to develop the protocol, IAL will be an im...
-
Product Compendial Testing Services
Ensuring that your raw materials meet specifications, whether it be excipients or Active Pharmaceutical Ingredients (APIs), is essential to prevent product failure. Raw materials that do not meet specifications can create problems with finished product quality and uniformity. We at IAL understand the i...
-
Product Certified toxicological studies
Toxicological tests performed at the Lukasiewicz - IPO:
-allow a toxicological evaluation of chemical substances contained in pharmaceutical products, veterinary medicinal products, plant protection products, food and feed additives, industrial chemical substances, etc.,
-make it possi...
-
Product Certified ecotoxicological studies
Ecotoxicological studies conducted by Lukasiewicz - IPO:
-chemical analyses in the range of environmental studies:analysis of concentrations/residues of chemical substances in water, soil, plant and animal material, food, fate and behaviour of chemical substances in the environment
-evaluation ...
-
Product Analytical chemistry&Physico-chemical properties
Determination of physical, chemical and technical properties of substances and chemical products

-
Product Analytical Development
Chemical analysis and development of methods to identify, characterize and test materials is an essential part of drug product development. Pensatech Pharma offers a wide range of analytical services to support your drug substance, excipients and drug products testing requirements.
Con...
-
Product GMP Quality Control & Batch Certification
Pensatech Pharma is authorised to do quality control testing (physicochemical and pharmaceutical analysis) as well as batch certification of finished and investigational medicinal products in its GMP-certified laboratories. • Comprehensive and well-documented quality control services • Testing and release...
-
Product Small scale synthesis
• Route scouting • Feasibility studies • Synthesis of compounds in mg/g scale • Metabolite synthesis • Preparation of standards / reference substances • Impurity profiling • Impurity reference synthesis • Synthesis of stable isotope labelled substances
-
Product Contract manufacturing and analytical services
• cGMP manufacturing of APIs and key intermediates • Analytical method validation • Process validation • ICH stability studies • Degradation studies • Full CMC package • Drug master files: compilation, maintenance • Comprehensive documentation
-
Product Analytical Development
Idifarma’s analytical development services include the following activities:
- Solubility studies in different media (organic, aqueous and different PH levels).
- Characterization of the thermal profile and identification of polymorphic forms by DSC.
- Impurities profile characterization. q...
-
Product Project Management and Consulting service
The KIPA service portfolio in Asia:
1.Facility planning 2.GMP Consulting for pharmaceutical manufacturers and Herbal Product manufacturers up to highly regulated market certifications (EU / PICs / WHO / GACP; GMP for Herbal medicines)
3.GMP certification manage...
-
Product Contract Development & Manufacturing (CDMO)
Our capabilities include: • Development Services: Process, Analytical and Formulation Development, and Non-GMP Lab-Scale Manufacturing • Drug Substance: Upstream Manufacturing and Downstream Processing • Drug Product: Clinical and Commercial Manufacturing, Drug Product cGMP Lyophiliza...
-
Product Cannabinoids Analysis
The Quality of Cannabis and Hemp products in all matrices is guaranteed by Eutech Scientific Services. Our highly trained staff will support your business with the expertise and reliability needed to handle your CBD analysis.
EuTech Scientific Services has been in business for over 2...
-
Product Extractables and Leachables studies
Extractables/Leachables (E&L) profiles have as a primary purpose identifying any potential chemicals derived from the chemical interactions of a drug with the packaging components ,or the interaction within the pharmaceutical drug formulation.
In addition, E&L is a FDA, ICH, and USP studies...
-
Product Contract Analytical Services
•EuTech Scientific Services has been serving the industrial and academic scientific community since 1994.
•EuTech focuses on the development of new analytical methods and utilization of existing methods. •GLP/cGMP and DEA Certified Lab, Schedules I through V
•FDA Regulated
-
Product Assistance with FDA Inspections
EAS Consulting Group is available to provide a mock FDA inspection of domestic and foreign facilities to determine deficiencies and/or to develop a corrective action plan either before FDA makes its initial inspection or if FDA finds deficiencies that need correction.

-
Product Contract Analytical Services
Established in 2004, Parsolex is a world-class contract services company delivering analytical, development, manufacturing, and strategic advisory solutions to clients who are involved with non-sterile pharmaceutical products for clinical trials as well as commercialization. Our clients include leading aca...
-
Product Formulation Development
We have particularly strong expertise in developing oral dosage forms (powder in capsule/vials/bottles) and aseptic liquids. We combine formulation development with physical characterisation and analytical method development. The team works together to determine how best to formulate APIs for both non...
-
Product Clinical Services
Sharp Clinical Services, a division of UDG Healthcare plc, services Pharma and Biotech clients from phase I all the way through to commercialization. With a highly experienced team, we can handle every aspect of your clinical trials project. At Sharp, we offer a full compliment of clinical trial ser...
-
Product Manufacturing Services
At Sharp, we work closely with you to build an in-depth understanding of your goals, challenges and requirements before we begin our state-of-the-art manufacturing process. This enables us to tailor our clinical manufacturing services to meet your specific needs, making sure we deliver your product on time...
-
Product Accelerated Stability Assessment Program (ASAP) and Laboratory Services
FreeThink is a global leader in the science and technology of stability. In addition to lab services, FreeThink licenses ASAPprime®, a proprietary software which we developed based on the Accelerate Stability Assessment Program (ASAP) to determine shelf-life. ASAP can speed and streamline p...
-
Product Raw Materials Testing Services, Compendial
Pace analytical life sciences llc offers wide range of services which includes raw materials testing service. It provides raw material testing services to the pharmaceutical, biopharmaceutical and medical device industries. It offer excipient testing to support usp/nf, ep, bp, jp, fcc and acs monograph tes...
-
Product Pharmaceutical / Biopharmaceutical Testing Services
Pace analytical life sciences llc offers wide range of services which includes product release testing for pharmaceuticals service. It provides pharmaceutical product release testing for both qa pharmaceutical release testing and clinical release testing. It offer quality assurance/quality control product ...
-
Product Structural Characterisation of Biologics
Structural characterization of proteins, monoclonal anti-bodies (mAbs) and antibody-drug conjugates (ADCs):- HR-MS based molecular weight measurement of intact proteins - Reduced / non-reduced forms (heavy and light chains of mAbs) - Peptide mapping
-
Product Electron Beam
There is much to consider when choosing a sterilization technology. Sterigenics’ expert advisors look forward to working with you to understand and achieve your unique product goals. We offer all major sterilization technologies: Gamma Irradiation, Ethylene Oxide & Electron Beam as well as innovativ...
-
Product Chemical and Material Testing
Alliance Technologies is a DEA licensed and FDA registered contract chemical testing laboratory and consultancy. Our senior analytical chemists provide a wide range of analysis and material testing services to a diverse, international client base with expertise in problem solving, method development, fail...
-
Product Speed Laboratory Inc
Speed Laboratory is a QC/QA laboratory that supports both FDA-regulated and non-regulated products and companies, with value, integrity, and commitment. In today’s dynamic marketplace, organizations can be reassured that they will get exceptional laboratory services in the pharmaceutical, consumer goo...
-
Product Clinical Services
CENTRO DE ESTUDIO CIENTÍFICOS offers wide range of services which includes clinical services. Contact us for more information.
-
Product Analytica Services
CENTRO DE ESTUDIO CIENTÍFICOS offers wide range of services which includes analytica services. Contact us for more information.
-
Product Regulatory Affairs
CENTRO DE ESTUDIO CIENTÍFICOS offers wide range of services which includes regulatory affairs. Contact us for more information.
-
Product Analytical Development And Validation Services
Pharma-Data sa offers wide range of services which includes analytical development and validation services. It includes justification of specifications, drug product release testing, method transfer, validation of microbiological methods (sterility, bet etc), filter validation (for sterile products – leach...
-
Product Marketing And Sales Service
DKSH offers wide range of services which includes marketing and sales service. It provides a complete array of marketing and sales services for consumer goods, luxury and lifestyle products, healthcare products, performance materials, and technology products. It has a long-standing track record in brand-bu...
-
Product Distribution And Logistics Services
DKSH offers wide range of services which includes distribution and logistics services. It provides custom-made logistics solutions, uniquely tailored to your needs. it has its own warehouses and offer third-party logistics services. It can outsource many additional specialized services with it including pr...
-
Product After-Sales Services
DKSH offers wide range of services which includes after-sales services. It ensures top quality standards, fast problem resolution and the ability to establish a high value image. The expertly trained teams provide customer service, repairs and maintenance, on-the-spot training and know-how transfer, offeri...
-
Product Development Sciences
Our development laboratories are well equipped for conducting a diverse range of experiments. These encompass formulation development, cycle design and process refinement, as well as evaluating finished product. Ease of scale-up is accomplished by completing process development studies within a pilot scale...
Looking to outsource analytical pharma studies? Find 100+ global CDMOs offering analytical & laboratory services and discover the latest news from the contract services sector.
References
Upcoming Events
-
CPHI & PMEC India 2024
India Expo Centre, Greater Noida, Delhi NCR
26 Nov 2024 - 28 Nov 2024 -
CPHI Middle East 2024
Riyadh Front Exhibition & Convention Center Riyadh, Saudi Arabia
10 Dec 2024 - 12 Dec 2024 -
Pharmapack Europe 2025
Paris Expo, Porte de Versailles - Hall 7.2 | Paris, France
22 Jan 2025 - 23 Jan 2025
Pharmaceutical Industry Webinars
-
Webinar Key to Success: A CDMO's Pathway to Biologics Excellence
-
5th November 2024
-
3pm BST/ 4pm CET
-
-
Webinar The Changing Dynamics of Global API Manufacturing
-
17th September 2024
-
3pm BST/ 4pm CET
-
-
Webinar Shaping the Future of Italy’s Pharma Market: Trends and Opportunities
-
4th September 2024
-
4pm CET/ 10am EST
-
-
Webinar Fragment-Based Oligonucleotide and Oligopeptide Synthesis
-
30th Jul 2023
-
4pm CET / 10am EST
-
-
Webinar GMP Rationale for Sterile High-Potency/Toxic Pharmaceuticals
-
18th June 2024
-
4pm CET / 10am EST
-
-
Webinar Unlocking Opportunities in the Growing Pharma Landscape of The Middle East
-
5th June 2024
-
3pm CET / 9am EST
-
-
Webinar Exploring Technological Trends in the Future of Pharmaceutical Manufacturing
-
23rd May 2024
-
4pm CET / 10am EST
-
-
Webinar Achieving Manufacturing Excellence Through Digital Transformation
-
16th April 2024
-
4pm CET / 10am EST
-
-
Webinar Made in Africa: What’s Driving Pharma Manufacturing
-
28th March 2024
-
4pm CET / 10am EST
-
-
Webinar Case Study: Risk Management for Annex 1 Sterile Production EMS
-
28th February 2024
-
4pm CET / 10am EST
-
-
Webinar Innovative Strategies for B2B Pharma Marketeers: Driving Value through Content
-
20th February 2024
-
4pm CET / 10am EST
-
-
Webinar Revolutionizing Pharma: Data and AI Unleashed
-
18th January 2024
-
4pm CET / 10am EST
-
-
Webinar Optimal Temperature: Elevating Biologics Cold Chain Excellence
-
16th January 2024
-
4pm CET / 10am EST
-
-
Webinar Market Outlook – The Biggest Pharma Trends of 2024
-
12th December 2023
-
4pm CET / 10am EST
-
-
Webinar The Next Frontier – Emerging Opportunities in the LATAM Pharma Market
-
21st November 2023
-
4pm CET / 10am EST
-
-
Webinar Vistamaxx™ MED - imagine the possibilities for healthcare product performance
-
10th October, 2023
-
4pm CET / 10am EST
-
-
Webinar Co-processing: A Multifaceted Approach for Enhancing Density & Powder Flow
-
19th September 2023
-
4pm CET / 10am EST
-
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance









































































.jpg)

.jpg)





























.jpg)
















.jpg)
















































.jpg)














.png)







.png)

.png)



.jpg)




