- Home
- Contract Services - Contract Services
CPHI Online is the largest global marketplace in the pharma ingredients industry
-
Products551,744
-
Companies7,781
-
Articles11,636
-
Events8
-
Webinars342
Contract Services - Contract Services
- Analytical Development
- Blending
- Cell-culture Development
- Chemical synthesis/analysis
- Computer software
- Consulting Services
- Contract Manufacturing
- Contract Packaging
- Cool Chain
- Data services
- Development & Validation of Analytica...
- Drug Discovery
- E-Business
- Formulation development
- Logistics and Distribution
- Lyophilisation
- Marketing Services
- Pharmacovigilance
- Process Optimisation
- Product Characterization
- Recruitment/human Resources
- Regulatory Affairs
- Synthetic Organic Chemistry
- Synthetic Route Design
- Vial/ Syringe Filling
Contract Services - Contract Services Companies (104)
Contract Services - Contract Services News
-
News 2024 Pharma Industry Trends Outlook: Collaboration, Market Maturity, and Digital Futures
The annual CPHI Online 2024 Pharma Trends Outlook, in partnership with Arvato Systems, identifies 12 key industry trends shaping the life sciences industry in the coming year.9 Jan 2024 -
News CPHI Barcelona 2023: Partnering for Success – Managing Outsourcing Relationships to Optimise Manufacturing Operations
During CPHI Barcelona 2023, insightful content sessions offered attendees the chance to explore trending topics with expert speakers and panellists. Here, we summarise what the pharma industry and supply chain are talking about the most.18 Dec 2023 -
News CPHI Barcelona 2023: The CDMO Outlook – a new definition of partnerships
To conclude the ‘Partnering for Success’ track at CPHI Barcelona 2023 (October 24–26th) we held a panel, with a range of experts, on the CDMO outlook.30 Nov 2023 -
News On track at CPHI Barcelona – The Track Sponsor interview: Seqens
In our packed out content sessions at CPHI Barcelona this year, we focus on some of the hottest topics coming up in the pharma industry, with each track sponsored by a leading expert in the field.1 Nov 2023 -
News The Patient-Centric Synergy of Pharmaceutical CDMO and CRO Collaborations
Pharmaceutical collaborations are nothing new to the industry. Increasingly complex drug development programs, calls for supply chain resiliency, and the involvement of all key stakeholders throughout a drug’s development lifecycle are pushing co...4 Oct 2023 -
News An Altogether Different Kind of CDMO
Discover what makes Pfizer CentreOne an Altogether Different Kind of CDMO.19 Sep 2023 -
News CPHI Barcelona Keynote Preview - Mapping the journey to commercial starts at proof of principle completion
Ahead of his session at CPHI Barcelona, which explores the current state of the CDMO landscape, Vinay Saluja (VS) – global head development services at Novartis Global Biotech Cooperations, contract manufacturing unit of Novartis – discusse...19 Sep 2023 -
News CPHI North America 2023: from the experts - Cambrex
Throughout CPHI North America (Philadelphia, PA; April 25–27, 2023), we caught up with some of the exhibiting organisations to ask what the show brings to the North American pharmaceutical sector, and what is driving industry innovation in this r...30 May 2023 -
News CPHI North America 2023: from the experts – Pfizer CentreOne
Throughout CPHI North America (Philadelphia, PA; April 25–27, 2023), we caught up with some of the exhibiting organisations to ask what the show brings to the North American pharmaceutical sector, and what is driving industry innovation in this r...22 May 2023 -
News A Start-Up’s Guide to Choosing Their First CDMO: Breakfast Session Bulletin
Throughout CPHI North America 2023, several Breakfast Sessions were held to start the day off right with a complimentary breakfast and a networking and learning session for our attendees. With insightful presentations from industry experts and partners...3 May 2023 -
News CPHI North America Anniversary Ball – celebrating The Art of Pharma
This year marks the 5th anniversary of CPHI North America regional events. To celebrate this remarkable achievement, the CPHI show team hosted The Art of Pharma Anniversary Ball at the illustrious Philadelphia Museum of Art, with a keynote speech from ...26 Apr 2023 -
News Rebuilding the United States' pharma infrastructure – CPHI North America 2023 preview
As we prepare for CPHI North America from April 25–27 in Philadelphia, USA, CPHI Online caught up with some of the track sponsors to discuss how the show connects vital players within the North American pharmaceutical landscape. ...18 Apr 2023 -
News Seqens Group Expands CDMO Offerings to Include CRDO
Meet with Seqens team members at CPHi, April 25-27, 2023 to learn how we have extended services to rapidly and sustainably advance drug development. VISIT CPHI Booth 44114 Apr 2023 -
News To recruit or to retain - where is the pharma talent going?
Discover how the pharmaceutical workforce has evolved past the pandemic, and the challenges faced by the pharma industry during a talent acquisition and retention crisis.12 Apr 2023 -
News Sai Life Sciences opens new HPAPI manufacturing facility in Bidar, India
A new HPAPI development and manufacturing block is now open at Sai Life Sciences’ Bidar Manufacturing Campus, expanding the CRDMO’s capabilities in small molecule discovery and commercialisation.17 Jan 2023 -
Sponsored Content 2023 Pharma Trend Outlook: Innovation, Resilience, and Pharma 4.0
Download our 2023 Pharma Trends Outlook report to discover the trends set to shape the pharmaceutical landscape in the new year, with expert opinions and insight from across the pharmaceutical value chain.2 Jan 2023 -
News Aragen’s formulation manufacturing facility to begin operations in 2023
Aragen is set to open their new manufacturing facility at their Mallapur campus in Hyderabad, India in January 2023, enhancing the company’s ability to provide scaled manufacturing solutions and clinical supplies to their customers.25 Oct 2022 -
News CDMO - Health is a Joint Project: Connect to Frankfurt on-demand
In this Connect to Frankfurt session, Emanuele Agnese, Business Development Manager and Pietro Allegrini, R&D Director at Indena, present the ways in which a reliable manufacturing partner in the pursuit of better health outcomes for patients and pharm...11 Oct 2022 -
News Streamlined Development for Efficient Production of Bispecific Molecules: Connect to Frankfurt on-demand
In this Connect to Frankfurt session, Séverine Fagète, VP, Cell Line Development Services at Selexis (Geneva, Switzerland), and Brandon Brino (Durham, USA), Process Development Scientist and Group Leader at KBI Biopharma, present an overv...7 Oct 2022 -
News Expansion of Quotient Sciences' UK drug development and manufacturing facilities
Quotient Sciences’ Nottingham and Reading facilities have announced the completion of their facility expansions, providing further capabilities to support their flagship program Translational Pharmaceutics®.26 Sep 2022
Contract Services - Contract Services Products (269)
-
Product Finasteride tablets USP 1 mg - Venopecia
VENOPECIA 1mg is having the drug ingredient called Finasteride 1 mg and is available in a Tablet form packed 10 tabs in one strip. Contact us for more Information.

-
Product Dydrogesterone 10 mg Tablets - Dydroloop
Shree Venkatesh International Ltd provides a wide range of pharmaceutical products which includes Dydrogesterone 10 mg Tablets. Dydrogesterone 10 mg Tablet is useful in the treatment of menstrual disorders such as absent, irregular or painful menstrual periods, infertility, premenstrual syndrome and e...
-
Product 1,2-Dioleoyl-sn-glycero-3-phosphoethanolamine
Based on our unique lipid processing and isolation techniques Merck’s helper lipid DOPE demonstrates outstanding solubility and thus distinctive physico-chemical properties in comparison to conventional amorphous DOPE’s. Those improved handling properties facilitate your LNP formulation process
-
Product (R)-1,2-di-O-octadecenyl-3-trimethylammonium propane chloride
Cationic lipid used in the preparation of liposomes and lipoplexes. Specifically, R-DOTMA chloride with its positive charge allows the protective loading of nucleic acids into lipid nanoparticles and promotes their endosomal release. Merck as the solely manufacturer of DOTMA-Cl can provide you with high-pu...
-
Product Outsourcing
Patheon by Thermo Fisher Scientific has a broad manufacturing platform for pharmaceutical and biologic products which provides sustainable solutions for mammalian cell-based and microbial-based manufacturing, green chemistry R&D and manufacturing technologies, and finished dosage production of biopharm...
-
Product Biologic drug substance CDMO services
From pre-clinical development to commercial supply, Patheon by Thermo Fisher Scientific is an industry leader in the development and manufacture of mammalian cell culture drug substances. Patheon offers biotech and pharmaceutical companies the ability to pursue opportunities around the globe with a fully i...
-
Product SOFTGELS
At Softigel, we offer much more than traditional softgels, including everything from a single unit dosage form, to multiple delivery systems in a single dose. Softgels are an effective delivery system for oral drugs, especially those with low solubility and/or permeability (BCS Classes II, III and...
-
Product Commercial Support
Selectchemie AG provides a wide range of services including commercial support. Our experienced professionals are at your service at locations in 18 countries to deliver tailor-made solutions according to your needs. Contact us for more information.
-
Product CONTRACT MANUFACTURING - SMALL MOLECULES
KD Pharma’s professional expertise, state-of-the-art assets and proven track record in regulatory compliance allow us to become your preferred CDMO solutions partner.
Your Agile Experts
• Responsive to our customers’ needs in a more dynamic customer-centric way • Adaptive to the most diverse re...
-
Product Analytical Services
Our state-of-the-art analytical testing labs support drug substance (API) and drug product (finished product) analytics across all phases of clinical development and into commercial release.
We employ over 170 highly skilled analysts working in GMP environments across the UK, Europe, and the...
-
Product Drug Substance Manufacturing for Viral Vaccines, Gene & Immune Therapeutics, Oncolytic Viruses and Viral Vectors
Drug Substance Manufacturing for pre-clinical, clinical and commercial supply- Validation of aseptic processes
- Cell and virus banking
- Using own seed cell banks: VERO and DF-1, Duck cell line (AGE-1)
- API production using different TechnologiesMVA Know-How
IDT Biolo...
-
Product Pregabalin Capsules
Each hard gelatin capsule containsPregabalin EP 25 mg / 50mg /75mg /150mg / 300mg
-
Product Injectables (Aseptic Filling)
Specialist in filling and packaging SVP (small volume parenteral) in prefilled syringes and vials. Our injectable plants export over 50 countries worldwide.
- 5 high speed lines for syringes (aseptic filling)- 3 high speed lines for vials (aseptic filling)
- Terminal sterilization
- Fully equipped...
-
Product Biotechnology-oncology-immunology-cns
Rhenochem Ag is the pharmaceutical trading, develops packaging solutions for the pharmaceutical industry and a global supplier of production machines and accessories for the pharmaceutical industry, which includes biotechnology-oncology-immunology-cns it belongs to pharmaceutical. Contact us for more infor...
-
Product Process Development and Support Services
Experic offers a consultative and collaborative approach to technical transfer and process development based on a foundation of deep scientific knowledge and the ability to solve problems and facilitate scale-up. Process development activities are augmented with analytical services, provided by qualifi...
-
Product Unique Fruit Matrix
Delicious fruit matrix meets well-established Probiotics: With our fortified fruit gums we‘ve finally found a convenient and delicious way to end pill fatigue. The brand new product in our health division merges several trends: Global markets for gut health products are growing rapidly, as is the ...
-
Product Cell Culture Services
FUJIFILM Diosynth Biotechnologies is a leading provider of cell culture services for biologics, advancing anddelivering life changing therapies. We offer complete solutions for cell line development, process development, late phase activities, clinical and commercial manufacturing of a wide variety of biop...
-
Product Rontameal Nutri-pro Adult fomulas
Rontameal Nutri-pro ACTIVE
Nutritional supplement formulated especially for Healthy ageing!Rontameal Nutri-pro BALANCED
Nutritional supplement with High Calcium formulated especially for Healthy Joints!
Rontameal Nutri-pro LGI
Nutritional supplement formulated especially for Type-2 Di...
-
Product Contract Development and Manufacturing Organization (CDMO)
As a contract development and manufacturing organization (CDMO), Formosa Laboratories (Formosa Labs) offers full-service for both general and high potency API (HPAPI) for all stages of drug development for both clinical trial material (CTM) and commercialized (NDA). Our GMP compliance has been approved by ...
-
Product Unifarma Herbals Triphala
Herbal product / Authentic Ayurvedic product. The pro kinetic cleanserTriphala , which combines the best properties of three plants,provides distinct cleansing and renewing effect on the entire body. It has anti-inflammatory ,anti-bacterial ,anti-septic and tonic emptying the stomach .Triphala hel...
-
Product Complete Characterization Testing Solution For Iron Carbohydrate
Method Development & Validation @ACCUPREC
1) Iron Chelation Assay (labile iron by U.V spectroscopy) Validated method
2) Polymorphic estimation of Iron carbohydrate by XRD
3) Estimation ratio of Fe3+ to Fe2+ ion validated method
4) Morphology estimation of iron carbohydrate
5)...
-
Product Contract Development & Manufacturing Services (CDMO)
Over 65 years of experience in developing & manufacturing pharmaceutical products for different markets. Operational Excellence, Flexibility & Adaptability.
Salvat offers integrated CDMO services. We have 3 manufacturing facilities in Barcelona, Madrid, and Miami with the capacity to ...
-
Product Vials
Mefar ilac sanayii offers wide range of products including vial filling between 2 ml through 300 ml.
Mefar's all services have EMA certification.
Contact us for more information.
-
Product Enzyme screening & proof-of-concept studies
We offer tailor made screenings of internal and external enzyme panels to identify applicable off-the-shelf biocatalysts for your target reaction. We integrate analytical services such as method development and validation throughout the process to get a reliable statement about the feasibility of your ...
-
Product Impurity Synthesis
Pharmaffiliates Analytics & Synthetics (P) Ltd, is an integrated CRO (Contract Reserach Organisition) established in year 2001. Presently our group consists of more than 130 Scientists with 3 R&D Centres offering its expertise in Custom Synthesis, Impurity synthesis, Isotoped lebelled compound...
-
Product Facility Planning
Csv Life Science can support customers in all phases of the project, right from the very beginning. This includes facility planning activities, aimed to a better and conscious defnition of your new or upgraded process needs

-
Product PEKUWhite - Cleanroom packaging
Cleanroom packaging:- flat film
- tubes
- half tubes
- cuttings
- bottom seam bags
- side gusseted bags
-
Product Highly Potent Pharmaceutical Principles
Olon relies on one of the most extensive expertise in handling highly potent pharmaceutical principles at global level, has raised the level of containment up to the actual standards and very large scale putting in place a set of primary and secondary containment designed to avoid the active substances...
-
Product Integrated offerings
Alcami is a Us-based, contract development, testing, and manufacturing organization for pharma and biotech companies. Our goal is to support our clients in making their projects go from potential to reality day-after-day.
Core Capabilities:Sterile-fill finish development and manufacturingO...
-
Product Electron Beam
There is much to consider when choosing a sterilization technology. Sterigenics’ expert advisors look forward to working with you to understand and achieve your unique product goals. We offer all major sterilization technologies: Gamma Irradiation, Ethylene Oxide & Electron Beam as well as innovativ...
-
Product NIANEED-500 (NIACINAMIDE TABLETS 500 MG)
NIANEED-500 (NIACINAMIDE TABLETS 500 MG)
MAINTAINING HEALTHY SKIN AND PROPER BLOOD CIRCULATIONREDUCING CHOLESTEROL LEVELS IN THE BLOOD
-
Product API Escrow
Take the risk out of working with a new supplier with Savnedor's escrow service. This way you know that what you are paying for is what you will receive or your money back.
The process is as follows:
- Companies agree to use escrow service
- Buyer and seller sign Savendor's escrow agreem...
-
Product Tray and Tub&Nest
Our Tub & Nest trays represent ready-to-fill (RTF) solutions meticulously designed to maintain the order and sterility of glass prior to the filling process, effectively eliminating the need for subsequent sterilization procedures.

-
Product CONTRACT PARTICLE ANALYSIS - Routine Analysis and R&d Studies
Analytical services for Drug Substance & Drug Product advanced particle characterization.
GMP services which include: • Particle Size Distribution (PSD) by Laser Diffraction • Morphological and Particle Size Parameters by microsocopy • NanoParticle Size and Concentration by MADLS • Z-Potentia...
-
Product Effervescent Tablets
As the expert in user-friendly dosage forms we can make effervescent tablets that have a pleasant taste, even if they contain bitter or difficult-to-process APIs, ensuring they leave neither residue nor foam when they dissolve. The composition of our effervescent tablets helps to speed up the absorption of...
-
Product Contract Manufacturing and Development, from Clinic to Commercial Scale
CoreRx provides comprehensive drug product pre-formulation, formulation, analytical, and GMP manufacturing and packaging solutions, enabling our partners to meet their drug program goals. CoreRx delivers these solutions from it’s development and manufacturing campus in Clearwater, Florida and it’s...
-
Product Licensing for Partners
PharmaMatch represents companies from all over the world and we have always shown ourselves to be a reliable and trustworthy partner. Our sales team has an excellent network and they are continually assessing the market per product to find the best match. Our partners can expect our Sales team to appro...
-
Product MyInfla
Pharmascience Inc provides a wide range of key products which includes MyInfla. Form: colchicine extended-release tablets. MYINFLA is indicated for the reduction of atherothrombotic events in adult patients with existing coronary artery disease, in addition to standard therapies...-comp241600.jpg)
-
Product Custom Protein Synthesis
We provide a Custom Protein Synthesis Service, using a chemical method that synthesises proteins amino acid by amino acid and making modifications on an atomic-scale. We work closely with our partners in designing, customising and optimising the proteins that is synthesized in an automa...
-
Product Comparator Supply
CREAPHARM CLINICAL SUPPLIES offers wide range of pharmaceutical services which includes comparator supply. It includes sourcing, import-export, eu qp release, blinding, clinical labelling, clinical packaging, storage, clinical distribution, etc. Contact us for more information. -
Product Packaging and Supply Chain Solutions
Tjoapack's packaging and supply chain solutions help our customers to make the world a healthier place. Our expert team work to facilitate your life sciences research, production and the delivery of medicines to patients across the globe.
We offer: - Packaging solutions for; oral solid blisters a...
-
Product SoftGroup® SaTT RU Hub Gateway
SoftGroup® SaTT RU Hub Gateway provides a connection between the Site level and IS MDLP (Chestny ZNAK track & trace digital system) allowing an exchange of information about each product. The application allows MAHs, Manufacturers and Parallel Importers to upload the product information, to reques...
-
Product Asset management and disposition (operating facilities)
Pharmabiosource inc offers a pharmaceutical services which includes asset management and disposition (operating facilities). It belongs to transaction-based services category. Contact us for more information.
-
Product Contract Manufacturing & Services
Abiogen Pharma S.p.A offers a wide range of services which includes contract manufacturing & services. It is developed through the availability of high quality, significant and attested technical-scientific competencies and technological capabilities available in the various strategic areas of production, ...
-
Product Finished Dosage Forms Manufacturing Service
Doppel Farmaceutici Srl offers finished dosage forms manufacturing service. Features: it includes manufacturing and packaging of soft and hard gelatin capsules, coated and uncoated tablets, granules, creams, gels, lotions, suppositories, vaginal suppositories, drops, syrups filled in plastic or glass etc. ...
-
Product Metabolic & Cardiovascular Services
Chempartner/china gateway offers metabolic & cardiovascular services. Extensive experience in drug discovery research for disease of diabetes, renal failure and cardiovascular disorders. Contact us for more information.
-
Product Process R&D
Ubichem Pharma Manufacturing Kft offers wide range of pharmaceutical services which includes process r&d. It minimises the risks associated with the scale-up of medicinal chemistry routes, giving our customers the confidence that their chemistries are viable for commercial scale production. Contact us for ...
-
Product Hydrogenation
Ka Malle Pharmaceuticals Ltd offers a wide range of services hydrogenation contract manufacturing. It provides hydrogenation contract manufacturing for products like Aroma & Perfumery, Pharmaceutical Industries, Agro Products, Fine Chemicals, etc
-
Product FSH/Urofollitropin for Injection BP - Ovitropin FSH 150 IU
Urofollitropin is a human follicle-stimulating hormone (FSH) analogue that is used to treat infertility in women. Available as combipack of Urofollitropin for Injection BP 150 IU & Sodium Chloride Injection BP 0.9%w/v

-
Product Progesterone Injection USP 100 mg/ml-Pregaloop
Shree Venkatesh International Ltd provides a wide range of pharmaceutical products which includes Progesterone 100 mg/ml Injection. Contact us for more Information
-
Product Hydroxyprogesterone Caproate Inj. 500 mg/ml - TRIUMPH
Hydroxyprogesterone caproate injection is a progestin indicated to reduce the risk of preterm birth in women with a singleton pregnancy who have a history of singleton spontaneous preterm birth. The effectiveness of hydroxyprogesterone caproate injection is based on improvement in the proportion of wom...
-
Product ADC & Bioconjugation CDMO services
With 15+ years of contract development and manufacturing experience, Millipore® CTDMO Services offers leading expertise in the conjugation of drug linkers to monoclonal antibodies (mAbs). As ADC and Bioconjugation CDMO, we have comprehensive experience in conjugation technologies backed by broad analytical...
-
Product ChetoSensar™
ChetoSensar™ is our novel solubilization technology for hydrophobic ADCs.
Increased ADC solubility, important for reaching high DARWider therapeutic index and higher drug efficacyImproved bioconjugation efficiencyChetoSensar™ and ready-to-conjugate drug linkers available as samples or within ADC...
-
Product Small Molecule CDMO services
With Millipore® CTDMO Services we support small molecule API synthesis projects from milligram to commercial quantities. We are the partner to address the most difficult challenges, whether the target is difficult to handle, synthesize, or source. With a true consultative and partnered approach, our servic...
-
Product Viral Vector CDMO services
Millipore® CTDMO Services delivers expertise and flexible solutions for viral vector development and manufacturing to advance cell and gene therapies from preclinical through commercial production.
Our proficiency with adeno-associated virus (AAV), lentivirus, adenovirus, and other vectors helps...
-
Product mAbs and rProteins CDMO services
With Millipore® CTDMO Services we provide deep expertise and flexible solutions across all stages of development and manufacturing for monoclonal antibodies, bi-specific antibodies, fusion proteins, or antibody fragments. We work with our clients to balance risk, optimize speed, and support all aspects of ...
-
Product mRNA / Lipidic Nanoparticles CDMO services
As part of our Millipore® CTDMO Services offering, we pave the way for robust, integrated, and consistent processes along all stages from pre-clinical to commercialization. Bringing together our capabilities, technical expertise, and regulatory know-how, we enable our clients to achieve their goals— to get...
-
Product Biosafety Testing & Analytical Development Services
Choosing the right partner for analytical and biosafety testing is critical in the race to approval. Our BioReliance® Contract Testing Services offer exceptional, risk-mitigating solutions with technical and regulatory expertise, to help bring life-changing drugs to market.
-
Product Dimyristoyl glycerol polyethyleneglycol-2000
High-purity and high-quality DMG-PEG2000 enabling a consistent supply from development to commercial scales.
-
Product Health, Safety and Environment Services
Our experienced HSE experts offer various health, safety and environment services: Safety data sheet (SDS) support (SDS classification, labelling) in most EU languages, including online access and QR links; Controlled substances, precursors, highly active substances and other restricted chemicals (in-clud...
-
Product ASYMMETRIC HYDROGENATION
We extensively address the specific needs of the pharmaceutical, food, cosmetics, agrochemicals and specialty chemicals industries with our capabilities in Transition Metal Catalysis (TMC) as applied to asymmetric hydrogenation. Complex active pharmaceutical ingredients (API’s) are often stereochemica...
-
Product Core Technologies and Services
• API / GMP Manufacturing • Rapid Process Development, Flawless Upscaling, and Economy of Scale-Production • Simulated-Moving Bed (SMB) Chromatography • Heterocyclic, Hazardous and Malodorous Chemistries • Organometallic and Cryogenic Chemistry • Transition-Metal Catalysis • High-Pressur...
-
Product Functional Polymers
The CDMO Division of KD Pharma offer well-established experience in the chemical functionalization of custom and designer polymers. We are specialized in polymer production used in sensitive applications, where a high level of confidence in production consistency, regulatory compliance and technical perfor...
-
Product Physical Sciences
We are experts in solid state chemistry.Through the creation of synergy between solid state chemistry and formulation development, our experienced scientists have the ability to
-
Product Packaging, Quality Control, Storage, Cold Chain
Within the contract development and manufacturing, IDT Biologika GmbH provides a wide range of other pharmaceutical services for our clients products, which include- Labeling & Packaging of vials, prefilled syringes, autoinjectors
- Serializiation (Track & Trace)
- Quality Systems
- ...
-
Product Process Development for Production of Viral Vaccines and Vectors
Process Development for API Production, Fill/Finish and Secondary Packaging- Cell line development
- Transfer of customer technologies
-Upstream / Downstream -iCellis Fermenter: development of scalable upstream and downstream technologies for adherent cells with higher yield.Major ...
-
Product Pellets (ec, cr, sr) - micro pellets mups - sterline ready-to fill products
Rhenochem Ag is the pharmaceutical trading, develops packaging solutions for the pharmaceutical industry and a global supplier of production machines and accessories for the pharmaceutical industry, which includes pellets (ec, cr, sr) - micro pellets mups - sterline ready-to fill products it belongs to sem...
-
Product Vials-ampoules-syringes-tablets-capsules-ndds
Rhenochem Ag is the pharmaceutical trading, develops packaging solutions for the pharmaceutical industry and a global supplier of production machines and accessories for the pharmaceutical industry, which includes vials-ampoules-syringes-tablets-capsules-ndds it belongs to finished formulations. Contact us...
-
Product Global accredited products from first-class partners in competent hands from a-z (Services)
Rhenochem Ag is the pharmaceutical trading, develops packaging solutions for the pharmaceutical industry and a global supplier of production machines and accessories for the pharmaceutical industry, which includes global accredited products from first-class partners in competent hands from a-z it belongs t...
-
Product Clinical Trial Supply Services
Experic provides comprehensive solutions to support clinical trials from study planning to close-out, including clinical trial supply management services, commercial drug sourcing, and clinical-scale manufacturing services. Manufacturing services include powder blending; powder, pellet, or tablet fil...
-
Product Specialty Commercial Manufacturing Solutions
Experic provides a variety of services to support Phase 3 to commercial-scale process transitions to create a customized solution for your specialty, niche, orphan, or combination product, or support novel pharmaceutical particle engineering technologies. Build-to-suit dedicated suites, a flexible...
-
Product Me n' Mil UHT milk
Me n’ Mil is a fresh, semi-skimmed and UHT treated cow milk with 1.5% fat In 1000ml and 200ml bricks.Me n’ Mil is an excellent quality cow’s milk, with high nutritional value and unique taste. If you love milk but prefer a balanced lifestyle, you can now experience the rich, creamy texture of low-fat ...
-
Product Blow-Fill-Seal Technology (BFS) Single-dose vials CDMO
Salvat specialize in developing and manufacturing sterile liquid pharmaceutical products in single-dose vials, using Blow-Fill-Seal technology (BFS).
Advantages of using Blow-Fill-Seal technology in your products:
• Innovation in the format, giving differentiation to your produc...
-
Product Lyophilization
Mefar offers wide range of products which includes lyophilization in vials with its 20 million + capacity with its state of the art freeze dryers.
Mefar's all services have EMA certification.
Contact us for more information.
-
Product Reference Standards
Pharmaffiliates is Supplying all the Pharmacopoeial Reference Standards (i.e. USP, EP, BP, IP, JP).Apart from this we offer Pesticide Standards, Phyto-chemical Standards, Food & Environmental Standards, etc.
For more details please send your enquiry on [email protected]
-
Product Fermentation
Relying on an experience gained over more than 50 years, OLON represent one of the most extensive know how of microbial fermentation in Europe. The Group, global leader in biomanufacturing, has two Biotechnology Centres located in Italy and is one of the first companies in Italy producing via microbial ...
-
Product PROSCOF CHILDREN (CHESTY COUGH RELIEF)
PROSCOF CHILDREN (CHESTY COUGH RELIEF) IVY LEAF EXTRACT
SOOTHES THE THROATHELPS CLEARS THE AIRWAYSWELL TOLERATED HERBAL FORMULASUITABLE FOR CHILDREN UNDER 6 YEARSALCOHOL FREE NON-DROWSYSUGAR FREENO COLOURING
-
Product PROTICS (PRE & PROBIOTICS)
PROTICS (PRE & PROBIOTICS)PROVIDES FAVOURABLE BALANCE OF FRIENDLY PROBIOTIC BACTERIA ESSENTIAL FOR MAINTAINING HEALTHY GUT & DIGESTIVE SYSTEM
-
Product WESCOVITA (PROTEIN POWDER WITH DHA & D3)
WESCOVITA (PROTEIN POWDER)PROTEIN POWDER WITH DHA ,BRAHMI,SANKHPUSHPI FOR BRAIN DEVELOPMENT PROTEIN FOR PHYSICAL GROWTH MULTIVITAMINS FOR ENERGY MINERALS FOR IMMUNITY
-
Product API Update Program
Know the market with our API Update Program. Savendor has developed this program to give your sourcing team the data they need to make the best API sourcing decisions quickly. Savendor will gather pricing, Certificates of Analysis, lead time, and capacity info from the manufacturers of any API. This c...
-
Product Gene Therapy Services
Our gene therapy process development and manufacturing capabilities are designed to confidently and rapidly progress your products from gene to market through development of robust and reproducible cGMP practices.
-
Product Orally Disintegrating Granules
ODGs are small granules containing one or more APIs that are packaged into small sachets ‘stick packs’. ODGs can be poured directly into the mouth and immediately swallowed without the need for any additional fluid. We use hot melt coating (HMC) technology for poor tasting APIs to ensure that ODGs are plea...
-
Product Chewable Tablets
HERMES PHARMA offers a wide range of user-friendly dosage forms which includes chewable tablets.Chewable tablets are a convenient, more agreeable alternative to conventional tablets. As they are chewed, they spend longer in the mouth and are therefore tasted more thoroughly. We use flavorings, as required,...
-
Product Hot & Cold Instant Drinks
HERMES PHARMA offers a wide range of user-friendly dosage forms which includes instant drinks. Instant drinks come in hot and cold versions and contain a single dose of powder or granules that dissolve in water, making them both easy to dose and consume. With extra rehydration benefits, instant drinks are ...
-
Product HERMES NutriCaps
HERMES NutriCaps - the new generation of supplements
HERMES NutriCaps are a novel way to deliver food supplements. People can use the Nespresso**(hard-pod) coffee machine in their kitchen to prepare supplements as a delicious and nutritious hot drink. Simple, fast and convenient, HERMES NutriCaps help c...
-
Product Oral Solid, Oral Liquid, and Topical Drug Product Development and GMP Manufacturing
CoreRx provides comprehensive drug product pre-formulation, formulation, analytical, and GMP manufacturing and packaging solutions, enabling our partners to meet their drug program goals. CoreRx delivers these solutions from it’s development and manufacturing campus in Clearwater, Florida and it’s...
-
Product Regulatory Support
Our experienced regulatory team has a good understanding of the regulatory framework in every region. We’ve obtained registrations globally through our clients. Our team provides clients with quality dossiers compiled in accordance with the latest guidelines, which result in numerous successful registr...
-
Product API Sourcing
PharmaMatch has an extensive network of API Active Pharmaceutical Ingredients (API) suppliers, ensuring our clients are matched with the right partner based on quality, price and regular supply. We can also provide third party audits or pre-audits for our clients to ensure the facilities are up to st...
-
Product Contract Manufacturing Organisations
PharmaMatch has global contacts with contract manufacturing organisations (CMO’s). These state of the art CMOs are EU and/or US approved and follow the GMP standards of production. As clients demand high quality standards and a reliable supply chain with the best commercial conditions, the CMO par...
-
Product Lab Services
Through one of our partners, we can provide comprehensive analytical support during the development of drug formulations. By combining innovative approaches, modern instruments and advanced knowledge in the field of analytical science we are able to achieve high performance in terms of selectivi...
-
Product Contract Development Manufacturing Services
Our expert R&D and manufacturing team delivers FDA & EMA approved turnkey solutions delivering CDMO and CMO competitive advantages for sterile cytotoxic injectables and solid dosage forms.-comp241600.jpg)
-
Product Olican
Pharmascience Inc provides a wide range of key products which includes Olican. Form: Capsules of 0.5 mg of fingolimod (as fingolimod hydrochloride). OLICAN is indicated as monotherapy for the treatment of patients with the relapsing-remitting form of multiple sclerosis (MS) to r...-comp241600.jpg)
-
Product Clinical Primary Packaging
CREAPHARM CLINICAL SUPPLIES offers wide range of pharmaceutical services which includes clinical primary packaging. It includes deblistering / blistering, bottling, etc. Blisters are available in a variety of materials: pvc/alu, pvc pvdc/alu, pvc pe pvdc/alu, alu/alu, blisters can be directly printed and p... -
Product Clinical Labelling & Secondary Packaging
CREAPHARM CLINICAL SUPPLIES offers wide range of pharmaceutical services which includes clinical labelling & secondary packaging. It includes IMPs and non-IMPs (rescue medications and co-medications), ivps including vaccines, controlled drugs and highly potent medicines, cosmetics and food supplements,... -
Product Clinical Distribution
CREAPHARM CLINICAL SUPPLIES offers wide range of pharmaceutical services which includes clinical distribution. It includes carrier qualification and management, import and export, including customs clearance, GMP/GDPstorage, distribution and returns management, etc. Contact us for more information. -
Product Cold Chain Management
CREAPHARM CLINICAL SUPPLIES offers wide range of pharmaceutical services which includes cold chain management. It includes cold-chain carriers with limited temperature excursions, express carriers with validated insulated shippers, point-to-point cold-chain deliveries. Contact us for more information. -
Product SoftGroup® SaTT EU Hub Gateway
SoftGroup® SaTT EU Hub Gatewayoffers an upload/update of PMD (Product Master Data) and PPD (Product Pack Data) to the EU HUB (EMVO). Via this Cloud solution, the MAH / Parallel distributor could select which serialized batch should be reported to EMVO. The system allows status verification o...
-
Product Capacity management
Pharmabiosource inc offers a pharmaceutical services which includes capacity management. It belongs to consulting services category. It includes financial performance review, benchmarking / best-in-class performance, time-to-market strategy, etc.
-
Product Business case development
Pharmabiosource inc offers a pharmaceutical services which includes business case development. It belongs to consulting services category. It includes launching a new product, options for acquiring / disposition of a facility, options for acquiring / disposition of an on-going concern, options for partners...
-
Product Benchmarking
Pharmabiosource inc offers a pharmaceutical services which includes benchmarking. It belongs to consulting services category. It is is often used in the exploration of capacity options and supports the development of the business case. It has conducted national and international assessments of bulk biologi...
-
Product Acquisition due diligence
Pharmabiosource inc offers a pharmaceutical services which includes acquisition due diligence. It belongs to consulting services category. It methods include protocols and categories that are specific to the pharmaceutical industry and the regulatory agencies. It has the disciplines to support various leve...
-
Product Process Development
Aether offers unique chemistries paired with project-ready, cutting edge process technologies at both pilot and manufacturing scale. Featured chemistries: high pressure hydrogenations, Grignard reactions, alkoxide (EO) derivatization, isobutylene chemistry, organo-lithiation, coupling reactions (Suzuki, He...
-
Product Integrated Services For Pharmaceutical Development
Abiogen Pharma S.p.A offers a wide range of services which includes integrated services for pharmaceutical development. The department is comprised of a production area and a quality control unit for investigation medicinal products (imps) operating in accordance with the eu-gmp good manufacturing practice...
-
Product Formulation Development Services
Doppel Farmaceutici Srl offers formulation development services. Features: it include analytical development studies, formulation development, stability studies (ich), process development, scale-up, process validation, regulatory affairs, technology transfer, ctd documentation and product manufacturing for...
-
Product General Test Service
Chempartner/china gateway offers general test service. Contact us for more information.
-
Product Chiral Separation
Chempartner/china gateway offers chiral separation. Contact us for more information.
-
Product Purification and Structure Elucidation service
Chempartner/china gateway offers purification and structure elucidation. Contact us for more information.
-
Product Instrumentations service
Chempartner/china gateway offers instrumentations. Contact us for more information.
-
Product cGMP API Manufacture
Ubichem Pharma Manufacturing Kft offers wide range of pharmaceutical services which includes cgmp api manufacture. Contact us for more information.
-
Product Intermediate Manufacture
Ubichem Pharma Manufacturing Kft offers wide range of pharmaceutical services which includes intermediate manufacture. It provide custom synthesis services on lab to pilot plant scale to support our customers' development programmes. Contact us for more information.
-
Product Discovery Chemistry
Ubichem Pharma Manufacturing Kft offers wide range of pharmaceutical services which includes discovery chemistry. With particular expertise in the synthesis of heterocyclic compounds and offer a broad range of novel building blocks for use in your medicinal chemistry programmes through discovery catalogue....
-
Product Pharmaceutical Chem. Support
Ubichem Pharma Manufacturing Kft offers wide range of pharmaceutical services which includes pharmaceutical chem. Support. It offers services to support the synthetic development projects. Such services include: analytical method development and validation, impurities & reference standards, salt screening ...
-
Product Experts in Regulatory, Pharmacovigilance & Quality
TOSCA has developed out of the Regulatory and QualityDepartment of a regional pharmaceutical companyactive in Central Eastern Europe (CEE) and the CIScountries.With the experience of almost two decadesand handling of more than 200 registrations in thisregion we have a profound knowledge of the regulationsa...
-
Product GMP Storage
GMP Storage of IMP, GMO and API at a Variety of Temperature Ranges 15-25 °C, 2-8 °C, -20°C, -80 °C, -150°C, LN2
Controlled substance Storage at 15-25 °C
-
Product IODOBENZNE DIACETATE
General InformationA. Product Name: Iodobenzene DiacetateB. Product Number: P15C. Cas No: 3240-34-4D. Synonyms: (Diacetoxyiodo) Benzene, (HydroxyIodo)-Diacetate Bis(acetato)phenyliodineE. Molecular Formula: C 10 H 11 IO 4F. Molecular Weight: 322.10G. Physical State: Solid White to off white crystallinepowd...
-
Product 2-Phenyl Butyric Acid
General InformationA. Product Name: 2-Phenyl Butyric AcidB. Product Number: P40C. Cas No: 90-27-7D. Synonyms: 2-Phenylbutanoic acid,α-Ethylphenylacetic acidE. Molecular Formula: C 10 H 12 O 2F. Molecular Weight: 164.20G. Physical State: Solid White to off white crystallinepowder.H. Purity: Not less than 99...
-
Product 2-Amino-1,3-Propanediol (Serinol)
General InformationA. Product Name: 2-Amino-1,3-Propanediol (Serinol)B. Product Number: P26 C. Cas No: 534-03-2D. Synonyms: 2-Amino-1,3-propaned,2-Aminoglycero2-Aminopropan 1,2 DiolDL-SERINOLE. Molecular Formula: C 3 H 9 NO 2F. Molecular Weight: 91.11G. Physical State: Solid White to off white crystallinep...
-
Product INOSIM Consulting & Services
When it comes to using simulation tools efficiently, experience goes a long way. The more precise the input, the more realistic the model and the more reliable the simulation results.
INOSIM can be your partner for simulation services: whether you need training combined with start-...
-
Product Aseptic Fill & Finish
As specialized CDMO we bring our expertise to sterile processing and aseptic Fill & Finish of complex formulations, such as High potent and Cyto molecules (OEB 4–6), temperature/oxygen/light sensitive and organic solvents.

-
Product Freeze-drying
Freeze-dryers are equipped with Automatic Loading and Unloading System placed in Class A. Loading directly onpre-cooled shelves assures protection of temperature sensitive products. Freeze dryers have a capacity of 12 m² and 17 m².
-
Product HPAPI handling & processing
Highly potent and toxic substances (OEB 3 – OEB 6) are handled using HIGH-TECH containment technology and processes which are OHSAS 18001 certified.

-
Product CLINICAL RESEARCH
- Support for strategic evaluations and regulatory alignment on clinical trials
- Qualification of study centres and researches
- Preparation of research protocols
- Preparation of Clinical Drug Development Dossiers (CDDDs)
- Monitoring of clinical studies
- Review of scientific literature
-
Product GOOD MANUFACTURING PRACTICES
- Quality Audits
- Risk assessment and management
- Review of Documents
- Training
- Preparation of technical reports

-
Product Technical Due Diligence
For companies that are:
• Buying a Product outright • In-licensing a Product • Acquiring or Licensing Technology • Acquiring a Division • Acquiring a whole Company
To cover the wide range of disciplines involved in Technical Due Diligence, Explicat® Pharma provides clien...
-
Product Imikro Nano Tablets
PROMISE OF LIFE FOR A LIFETIME
We believe that the right to live healthy is for all, and no one must be devoid of it. So, each of our services promises to offer extended and quality life to patients. Our first-of-its-kind iMickro and iMickro Nano technology serve as a testament to our business p...
-
Product Dosecare
Dosecare is a dosing syringe for oral liquids.
It differs from the others as it allows you to simply and intuitively save the dose of liquid to be administered. Therefore once the dose is set, dosing errors are prevented, time is saved, there is no waste and soiling.
Thos...
-
Product GMP manufacturing
505 m3 GMP manufacturing capacity in ChinaGMP capabilities available in Pennsylvania, USA
-
Product Clinical solutions: Trials
Conscio's in-house clinical Unit has been operating since 2007, with more than 600 BA/BE/PK studies completed and proprietary of over 270 validated bioanalytical methods. Equipped with a 60-bed on-site clinical unit in Prague and a recently opened 36-bed unit in Ostrava, we provide comprehensive services w...
-
Product Ultra compact tablet press for personalized medication
Ultra compact tablet press with patented weight-controlled dosing system for individual tablet production.
Especially designed but not limited to pediatric medication, psychoactive API blends, Diabetes, nutraceutical tablets and a lot more.
-
Product High Speed Door
1. Specially designed fabric door for indoor applications requiring high frequency of operations.
2. High speed doors are CE certified
3. Special EPDM seal between lateral parts and curtains, providing complete seali...
-
Product Active pharmaceutical ingredients
CBC (Europe) GmbH offers a wide range of products which includes active pharmaceutical ingredients. Contact us for more information.
-
Product Intermediates
CBC (Europe) GmbH offers a wide range of products which includes intermediates. Contact us for more information.
-
Product GMP Audit & Consulting Services
We aim to provide the most reliable and preferred service platform to pharmaceutical companies worldwide for R&D cycle, regulatory affairs, and GMP compliance with a higher benchmark of quality, integrity, and ethics.
...
-
Product Analysis
Excerpt from the range of services offered by our analytical department: • physico-chemical testing • chromatography • spectroscopy/spectrometry • determination of TOC/TNb (water analysis/cleaning validation) • particle measurement
• elemental analysis
• dissolution testing
• pho...
-
Product Microbiology
Excerpt from the range of services offered by our microbiological department: • microbial contamination testing (bioburden) • identification of microorganisms • sterility testing • biological indicator testing • bacterial endotoxin testing (LAL) • assay of antibiotics • efficacy of preserva...
-
Product Bioanalytics/Molecular Biology
Excerpt from the range of services offered by our bioanalytical/molecular biological department:
• determination of mycoplasma • biological contamination testing • determination of content and identity of nucleic acids • determination of biological activity
• protein/antibo...
-
Product Pharmaceutical Development
PreformulationSolubility/stability assessmentExcipient screeningAPI properties for processability
Formulation developmentSolid dosePowdersTablets: sublingual, dispersible, effervescent, gastro-risistant, sugar and film-coatedCapsules, hard gel: pellets, double-component, granules, powders, e...
-
Product GMP Services(Good Manufacturing Practices)
We offer various GMP services which can help the clients across globe, ranging from audits to various consulting services.
We provide following comprehensive GMP Quality Compliance services to the pharmaceutical industry.
• Audits
• API Audit Reports
• QMS/Complia...
-
Product Solid Form Screening
We know that the needs in early development are different from the needs in late development and we tailor our screening approach based on information from the client and our experience with a wide range of compounds. At Crystal Pharmatech, we utilize a unique screening approach that co...
-
Product Crystallization
Crystallization is a critical unit operation in API manufacturing for both final product and intermediates. Desired product quality attributes can be obtained economically with high efficiency through well-designed and controlled crystallization processes.
-
Product Chiral Separation by Crystallization
More than 70% of drug candidates worldwide are chiral. Typically, for chiral API, only one enantiomer or diasteromer is biologically active or desirable. Therefore, the production of enantiopure compounds or diastereomers is imperative. In the production of small molecule drugs with desired chirality, sepa...
-
Product Biotage Initiator+ Microwave Synthesis System
Biotage® Initiator+ represents a new generation of synthesizer instruments for organic, medicinal, materials, nano and polymer chemistry professionals. It is an upgradeable and reliable platform allowing chemists to make great discoveries in less time.Heating reactions to high temperatures and pressur...
-
Product QC and R&D tests
We provide quality control analysis according to Ph. Eur., USP and in-house methods and requirements for raw materials and finished products in the range of :
- Identification testing
- A...
-
Product Engineering & Services
ZETA offers consulting and contract work in the areas of designing and engineering, process development and optimisation, smart engineering, qualification services, verification and validation, the installation of piping and equipment, and individual maintenance packages, all with a comprehensive customer ...
-
Product Mammalian manufacturing of biologics
GTP Bioways’ activities cover the full development cycle of your biotherapeutics, from cell line development to complete process & analytical development and cGMP manufacturing.
Our best-in-class cGMP facilities are equipped with single-use technologies to allow for flexible production of dr...
-
Product Transdermal Patch "Transfilm®" – topical & transdermal
Our Transfilm® is a non-dissolving patch system for dermal or transdermal delivery of your API. The delivery of a drug via a transdermal route of application offers the best of oral and intravenous administration. Avoidance of the first-pass effect leads to better bioavailability and may result in fewe...
-
Product Oral Film "Rapidfilm®" – our orodispersible film
Our orodispersible Rapidfilm® works like a conventional or orodispersible tablet. The film is placed on the tongue and dissolves within a few seconds. The active ingredient is swallowed together with the saliva and takes the same route as a tablet via the liver where the drug is metabolized. D...
-
Product Marketing Services
Years of pharma industry experience working with clients to help identify the best channels and messaging to maximize ROI, raise brand awareness and keep within budget.
-
Product Custom Intelligence
Work with our Pharmaceutical Consulting team, and draw on the strength of the entire Informa family. Get best-in-class datasets and insights from over 500 industry-leading analyst experts. Plus benefit from years of experience in delivering custom biotech and life sciences consulting projects.
-
Product Certified ecotoxicological studies
Ecotoxicological studies conducted by Lukasiewicz - IPO:
-chemical analyses in the range of environmental studies:analysis of concentrations/residues of chemical substances in water, soil, plant and animal material, food, fate and behaviour of chemical substances in the environment
-evaluation ...
-
Product Analytical chemistry&Physico-chemical properties
Determination of physical, chemical and technical properties of substances and chemical products

-
Product Formulation & Process Development Services
Pensatech Pharma uses its extensive experience in formulation and process development to offer the following services: • Contract formulation development services, which include tailor-made formulation solutions to the specific needs of our clients; solution of complex formulation challenges • Formulation...
-
Product Small scale synthesis
• Route scouting • Feasibility studies • Synthesis of compounds in mg/g scale • Metabolite synthesis • Preparation of standards / reference substances • Impurity profiling • Impurity reference synthesis • Synthesis of stable isotope labelled substances
-
Product Oral film "Mucofilm®" - mucoadhesive film
Our Mucofilm® offers intra- or transmucosal delivery. Our mucoadhesive film technology is designed for immediate release, directional or controlled release of your API. A thin film, which harbors your API, is applied to the mucosa of the oral cavity. After application, the film is small, light and ca...
-
Product Formulation Development
Idifarma offers solutions in different dosage forms with the ultimate goal of optimizing its future industrial production. The formulation development services encompasses:
- Selection of dosage form.
- API and excipient compatibility studies.
- Definition of qualitative and quantitative f...
-
Product Land in Europe- EU Market Access Support Services
Azierta provides global support in accesing to European Market to Non-European companies
European Pharma market access support services: There are three main strategies that may be implemented on an individual basis, or alongside each other, depending on your company needs...
-
Product Quality Assurance
At Azierta, we implement and maintain Quality Systems according to the needs of each company for enhanced resource management and results.
• In-company traininig • GxP Support: GMP/GDP Certification and GMP Compliance Adviser • Assessment and implementation of Quality Systems: Audits and/or self-...
-
Product Brand and Promotional Analysis
Powered by the IQVIA CORE™, our promotional analysis can help you understand the trajectory of your brand and measure its progress along the way. We can help you see where you are today, where you’re going and help you make adjustments as part of your path forward. Find out what you need to know about •...
-
Product Managed Markets Data & Analytic Solutions
Fundamental to the success of your market access strategy is clear visibility into payer, provider and patient behavior. IQVIA’s comprehensive real-world database includes lifecycle transactions, which offer the critical level of detail required to untangle the complex interaction of payer utilization mana...
-
Product Clinical Trial Optimization
Faster market access by optimized clinical trials Every study is unique and stakes are high. In order to ensure success IQVIA uses all available resources - therapeutical expertise, latest technology, advanced analytics and proactive approach. This allows us to effectively search for research centers an...
-
Product Standards Connect
Standards Connect provides an effective, money-saving, multi-user solution for accessing standards. By selecting content from multiple publishers, you create a subscription that is tailored to your business while only paying for the standards your company needs.
Standards Connect is the subscrip...
-
Product CIP/SIP
Hyde Engineering + Consulting provides wide range of pharmaceutical services which includes cip/sip. It includes cleaning program strategy, development of cleaning process cip, development of parameters for sip, cip & sip validation, validation of manual cleaning processes, etc. Contact us for more inf...
-
Product MENTHOL AND MINT OILS
AOS Products Pvt Ltd offers impeccable quality of Menthol Crystals, Mint Oils, Spearmint Oil, Peppermint Oil, Indian Piperita Oil, Basil Oil and derivatives of all these Oils. We offer these products as per Indian Pharmacopeia, British Pharmacopeia, US Pharmacopeia etc.
-
Product Process Development and GMP Manufacturing
3P Biopharmaceuticals is focused on process development and GMP manufacturing of biopharmaceuticals (microbial or mammalian) and cell therapy products at all stages (early development through commercial manufacturing). In addition, 3P supports through process transfers, late-stage project development, ...
-
Product Commercial Manufacturing
3P Biopharmaceuticals is a trusted partner for commercial manufacturing of biologicals. As part of our one-stop-shop service, we also offer a wide variety of in-house analytical testing and release capabilities to support the quality manufacturing of commercial-stage products. Thanks to our comprehensive Q...
-
Product Analytics and Quality Control
3P Biopharmaceuticals offers a wide range of services, including development and/or transfer of analytical methods at all project stages while applying detailed regulatory knowhow and the latest innovation. We also provide a wide range of in-house analytical methods.

-
Product Drug Substance Development and Manufacture
We provide our clients with a full offering of drug substance development, scale up and manufacture, along with comprehensive support services. Our skilled scientists are highly experienced in the development of bespoke and innovative solutions for technically difficult challenges. We pride ourselves on of...
-
Product Drug Product Development and Manufacture
We leverage our extensive experience in the Chemistry, Manufacturing and Controls (CMC) aspects of molecule development for drug product manufacture. Our all-in-one facility allows us to develop, manufacture, test, release and supply a range of clinical product types. All of our individual offerings can be...
-
Product 1. TP G - High Shear Mixer Granulator
It has been designed for the optimization of a wet granulation process. Used to turn a heterogeneous mixture of fine powder into a uniform granulation with an increase of the particle size and density. Moreover, it improves the fluidity of the product and its compactness properties, within a short process ...
-
Product 2. TP S - Fluid Bed Dryer
Designed to perform 3 different processes:
• Drying of wet granulate
• Top drive granulation
• Bottom spray coating for micro granules
Main Features:
• Standard capacity: 30 L to 1.540 L. • PLC + HMI for machine control i...
-
Product 3. TP R - Perforated Pan Coating
The perforated pan coating is an equipment based on a film coating for tablets and production of pellets. Its high technology allows an automatic flow control, avoiding the damp, saving coating and optimizing the process time.
TP R COATER | Main features: • Standard capacity: 5Kg ...
-
Product Discovery Biology
Sundia Meditech Company, Ltd. offersa wide range of drug discovery and development services which includes discovery biology. It belongs to drug discovery and research category. Features: it includes in vitro biology and in vivo pharmacology. Sundia's state-of-the-art biology facilities include an animal f...
-
Product Drug Metabolism and Pharmacokinetics (DMPK)
Sundia Meditech Company, Ltd. offersa wide range of drug discovery and development services which includes drug metabolism and pharmacokinetics (dmpk). It belongs to drug discovery and research category. Features: it has established various in vitro assays that help clients efficiently and cost-effectively...
-
Product Analytical Development
Sundia Meditech Company, Ltd. offersa wide range of drug discovery and development services which includes analytical development. It belongs to pharmaceutical development category. Features: it is to support chemical process development and formulation development during the pharmaceutical development. Ar...
-
Product API‘s, Lipid Excipients for Bioavailability Enhancer, Lipid Enhancer for Cell Culture, Carbopol, Contract Service (low molecular・ADC), 2MeTHF...
Besides the aforementioned CBC Group offers a wide range of products, thus kindly contact us for more information.

-
Product Capsule for Probiotic Supplement
Morishita Jintan Co. Ltd offers a wide range of health food products which includes probiotic strain. It is encapsulate live strains and reach the intestines without being killed by stomach acid. Shell formulation: gelatin or vege with acid resistance / enteric. .
-
Product Capsule for DHA / EPA
Morishita Jintan Co. Ltd offers a wide range of health food products which includes dha / epa. It is encapsulate mixture of dha and epa, adding enteric function. Reduced returning smell. Shell formulation: gelatin with acid resistance / enteric function. Content substance: lipophilic substances. Applicatio...
-
Product Launch Performance by Patient Segment
Each launch has a million moving pieces. Almost all are based on what you know about your market. Which patients are using which products? What is their patient journey? What types of physicians prescribe them? How do treatments and costs vary by geography or provider? Primary market research provides ...
-
Product Brand and Promotional Analysis
Powered by the IQVIA CORE™, our promotional analysis can help you understand the trajectory of your brand and measure its progress along the way. We can help you see where you are today, where you’re going and help you make adjustments as part of your path forward. Find out what you need to know a...
-
Product Managed Markets Data & Analytic Solutions
Fundamental to the success of your market access strategy is clear visibility into payer, provider and patient behavior. IQVIA’s comprehensive real-world database includes lifecycle transactions, which offer the critical level of detail required to untangle the complex interaction of payer utilization ...
-
Product Pharmaceutical Forms (Development)
Nextpharma offers wide range of pharmaceutical services which includes pharmaceutical forms (development). It includes forms such as solid, semi-solid, liquid, softgel capsule, vial, syringe, etc. Contact us for more information. -
Product Manufacturing
Nextpharma offers wide range of pharmaceutical services which includes manufacturing. It includes manufacturing of imps (solids, semi-solids and liquids) and matching placebos, dosator for powder / granules, de-blistering and de-packaging of medication, dosator for powder/ granules, blinding/ manufacturing... -
Product Packaging & Labelling
Nextpharma offers wide range of pharmaceutical services which includes packaging & labelling. It includes primary packaging, secondary packaging, special label design and printing, etc. Contact us for more information. -
Product SIPS
1- Consulting services 2- Pharmacovigilance 3- Regulatory Affairs 4- Anti-counterfeit 5- Training
-
Product Api services
Marken LLP offers a wide range of services which includes api services. It belongs to clinical trial logistics category. Marken's network fulfills the global movement of api from the point of manufacturing to cmos and primary packagers. Contact us for more information.
-
Product Biomarkers
Marken LLP offers a wide range of services which includes biomarkers. It belongs to biological samples category. Marken understands the critical link between biomarkers and a clinical study. Time sensitive logistics coupled with stability ensure that the samples arrive on time and within specification, eve...
-
Product Active/passive packaging
Marken LLP offers a wide range of services which includes active/passive packaging. It belongs to packaging solutions category. It ensures that biological samples stay within the proper temperature ranges at all times during transit or storage. Contact us for more information.
-
Product Active Pharmaceutical Ingredient Development Service
Consort Medical plc offers CDMO service which includes active pharmaceutical ingredient development service. Contact us for more information.
Search global Pharma Contract Service providers by region, sector, product or company name at CPhI Online and discover the latest news, interviews and reports from the CDMO sector.
Pharma Contract Services
The pharmaceutical industry is rapidly advancing, and as it continues to grow, the need for pharma contract services and manufacturing providers is simultaneously rising too. Over the past few years, pharmaceutical outsourcing services have grown popular and brought significant changes to the pharmaceutical industry.
Several pharmaceutical and biotech companies have adapted to outsourcing various pharmaceutical processes to pharma contract service providers to meet on-demand services effectively. By doing this, these companies have been able to cut costs, lessen supply chain risks, prevent unnecessary capital expenditures, broaden their geographic reach, and boost operational cost and effectiveness.
The activities of these services providers can be administrated by pharmacy benefit managers. Pharma outsourcing services cover every pharmaceutical process, including drug discovery, research, manufacturing etc., and they are contract-based. Learn more about pharma contract services, types, benefits and its impact on the pharmaceutical and healthcare industry.
Types of Pharma contract Service
Given the multiple complicated steps involved in drug development, there are various types of pharma contract services that handle each of the steps in the pharmaceutical manufacturing process. These pharma contract service types include Pharmaceutical contract manufacturing services, contract research and development services, pharma contract packaging services, contract clinical trial services, and sales and marketing services.
These pharma contract services are made available to the pharmaceutical and healthcare industry to reduce the burden of in-house drug development and manufacturing cost. Outsourcing services are an essential strategy utilized by pharmaceutical companies to help reduce competition from generic drugs, low productivity and the number of blockbuster drugs with patent expiration.
What are the most common types of pharma contract services?
Some of the most common pharma contract services include manufacturing outsourcing and research and development outsourcing. The contract research organizations (CROs) and contract manufacturing organizations (CMOs) meet the majority of the outsourcing demands of pharmaceutical companies.
The CROs provide pharmaceutical companies with preclinical drug development, development of the production process, clinical trial services and the development of drug delivery system. CMOs, on the other hand, handle the manufacturing process of APIs, formulations, drug dosage, intermediates, stability storage, and pharmaceutical raw materials.
How much do pharma contract services typically cost?
The cost of pharma contract services is dependent on several factors. Some determining factors include geographic location or region, type of pharma contract services required, and the length of the contract among many others. Although the cost of these services varies, they usually cost less compared to in-house industry activities.
There are no fixed prices for these services due to the variability of the determining factors. However, due to high demand, CMO and CRO services tend to cost more. For instance, based on a recent report., in 2017 the global pharmaceutical contract manufacturing market had an estimated value of USD 92.314 billion and is expected to attain a value of USD 146.41 billion by 2023. During the forecast period, this is likely to grow at a CAGR of 8.08%. Although there are various regions influencing this global market, the Asia-Pacific market has had the most impact in recent years.
Why are pharma contract services important to the industry?
The global pharmaceutical industry and life sciences are faced with several challenges associated with regulatory policies, pricing, research and development processes, etc., and this has stunted the growth of these industries. With pharma contract services, pharmaceutical companies and life science industries are presented with an opportunity to address these challenges while gaining numerous benefits as well.
Pharma contract services, help to boost manufacturing efficiency, net earnings and cash flow, compliance with regulatory processes, and hasten research and drug development at a cheaper rate. They help reduce overall costs and the need for investments in capital-intensive facilities and maintain product quality. Also, contract services allow pharmaceutical companies to divert funds and resources to other areas and give room for innovations.
What to consider when hiring a pharma contract service provider
When deciding whether to hire a pharma contract service provider or not, it is crucial for pharmaceutical and biotechnology companies to consider the risks and benefits associated with it. These industries will need to consider certain factors to avoid any ambiguity or conflict that may arise during the agreed contract period.
Necessary factors to scrutinize include the financial capability of the pharmaceutical company and service provider, the experience of the contract service provider, payment terms, quality standards of the project deliverables, and comparative costs between in-house services and contracted services. The payment currency, validity period and validity of the contract prices, trade terms, and purchase quantities should also be considered when hiring a contract service provider.
What does a contract pharmaceutical sales representative do?
A contract pharmaceutical sales representative is responsible for handling the education, distribution, and sales of pharmaceutical drug products. Also, the sales rep helps in the development and establishment of contracts with drug manufacturers and healthcare professionals. They are employed by pharmaceutical companies to promote the use of pharmaceutical products and medical device between healthcare professionals and also monitor clients to get feedback after use.
Pharmaceutical sales representatives are deployed to work with specific drugs, conduct analysis on the sales and administration of those drugs, the effects on patient care, and introduce new specialized drugs to healthcare professionals within a targeted location. Pharmaceutical sales reps can either work independently (freelance) or partner with a pharmaceutical company.
How does contract manufacturing work?
In contract manufacturing, a pharmaceutical industry makes an agreement with a contract manufacturing organization (CMO). Here, pharmaceutical products are manufactured by the CMO under the brand name or label of the partnering pharmaceutical company. However, contract manufacturers offer these services based on their formulas, consumer designs, and specifications.
Given the cost of production of certain chemicals and pharmaceutical products, most pharmaceutical companies prefer to allocate the manufacturing process of these products to CMOs. Once the process is completed, the manufactured product is collected by the pharmaceutical company and combined with other chemicals to produce the desired end-result. In the majority of cases, the CMOs are contracted to handle the purchase and shipment of raw materials and the maintenance of the manufacturing facilities and equipment. With contract manufacturing, pharmaceutical companies and firms can reduce in-house production cost.
What is the difference between a contract service provider and a pharmaceutical company?
Contract service providers are organizations or firms that provide a specific or a variety of services to pharmaceutical and biotechnology companies based on a previously established contract. These services range from drug discovery and development services to drug manufacturing and packaging services. These organizations, however, have little to no control over data security and are difficult to regulate.
Unlike pharmaceutical companies, the products and services they render are manufactured under the label or brand name of the contracting pharmaceutical company. These firms handle third-party local operations. On the other hand, a pharmaceutical company is one that is licensed to discover and research drugs, develop and market or distribute generic or brand medications within the healthcare industry. These companies have higher compliance with FDA regulatory policies and have better control over data security.
Top pharma contract services companies
Pharma contract service companies perform several functions and help to reduce the burden of production and manufacturing be pharmaceutical companies. They are several specialized contract companies such as contract development and manufacturing companies, contract research organizations, and contract testing laboratories, etc., that help to reduce the overall cost of production through outsourcing research, testing, drug development, and many others. There are top pharma contract service companies that provide these services and more, while also impacting the pharmaceutical industry. The majority of these key players have been listed below.
Who are the top pharma contract service companies?
The pharma contract service market has experienced exceptional growth over the years. Some of the key players have influenced the development of this global market in the pharma outsourcing industry. They include Catalent Pharma Solutions, Catalytica Pharmaceuticals, Dr Reddy's Laboratories, Pfizer, Cambrex, Boehringer Ingelheim Pharmaceuticals, Siegfreid USA, AbbVie Inc., Cambrex Corp., Patheon, Lonza, Inc., Contract Pharmacal Corp, Xcellerex LLC, Teva API, Roche, Delpharm, Alcami Corporation, etc.
How is success measured in pharma contract service companies?
Measuring the benefits and success of pharma contract service companies is a complex process. It involves a precise analysis of quantitative data to compare cost, return on investment, scalability, revenue, health, effectiveness, and efficiency as well as other qualitative factors such as feedbacks and reviews. Assessment of business economics influenced by the pharma contract services, comparative study for sales force effectiveness, etc., can aid in measuring the success pf pharma contract service companies in delivering consistent quality services.
The way the success of pharma contract service companies is measured is also dependent on the degree of outsourcing performed, a model for monitoring and measurement, and even the function it is leveraged on.
Why is it important to partner with a trusted pharma contract service company?
Bringing new drugs into the global pharmaceutical market is a complex and lengthy task that will require thorough research and development, the use of specialized manufacturing equipment, and experience with regulatory policies, etc. As a result of this, several companies are continuously seeking the services of pharma contract service providers such as contract manufacturing organizations and contract research organization.
Regardless of what you need a pharma contract service company for, you need a trusted one to provide quality services, as a wrong choice can result in devastating consequences. With unverified pharma contract services and lack of transparency, you may face low-quality standard and value production, partnership problems, and regulatory compliance problems (policy issues, medical device and equipment used). This can, in turn, affect the sales of drug products, give rise to significant health care issues, and hinder the growth of your pharma company.
How to find the right contract services for your pharma company?
Pharmaceutical contract services can only be beneficial to your pharma company if you find the right one. Pharmaceuticals and biologics companies are in a constant search for reliable partners who will boost their drug production process through a consistent supply of products and services. However, in most cases, pharmaceutical companies find it challenging to choose the right contract manufacturing services needed for the success of their pharmaceutical product and the company’s overall performance.
Although pricing is a vital factor to consider, several other essential factors must be considered. To find the right contract services for your pharma company, you must first establish your drop development needs, then scrutinize your list of partners based on certain factors. Relevant qualification and experience in producing pharmaceutical products at different clinical and commercial phases, manufacturing capacity and quality, and financial stability should be considered when searching for a suitable contract service provider.
Also, the geographical location, GMP and regulatory compliance, reliability, personnel, and technical capabilities of your desired manufacturing partners should be critically reviewed. The kind of partnership or relationship desired, short-term or long-term should also be put into consideration.
Where can you find the right contract manufacturer for your pharmaceutical product?
As technology continues to evolve, accessibility to various services and information is made even more comfortable. Almost every service provider can be found on the internet, and contract manufacturers aren’t an exception. Pharma contract manufacturers can be found online, through manufacturing consultancy companies, through recommendations from pharma company owners. After contract manufacturing organizations are found, relevant research and comparisons can be carried out to make the final choice.
How do you determine your contract drug development needs?
After deciding to work with a pharma contract service company, the next step is to determine which pharma contract service is right for your pharma company. The outcome of this decision is dependent on your company's existing capabilities and your drug's specifications.
What type of drug does your company intend to produce and what is its estimated market size? The type and class of drug will determine the technical proficiency and technology needed for effective drug development and contract manufacturing. Sometimes, CMOs only have specific manufacturing and development capability available; therefore, market size is an important factor that helps you determine your needs and choose a suitable partner. A standard contract manufacturing organization should be able to assist your pharma company in moving from the clinical to commercial scale.
What are your pharma company’s in-house capabilities? Your drug developments needs will influence the kind of manufacturing and development solution you are in search of. You may be in search of complete or incomplete contract development and manufacturing solutions or a contract partner to fill in for your pharma company in places that it is lacking. Also, the drug phase is essential. The stage of clinical trials and commercialization of your drug will also determine your development needs in a contract manufacturing partner. In-depth knowledge of your contract drug development needs and the designated timeline is essential for the identification of the right contract manufacturing organizations for you.
Why should companies research and compare contract services companies before entering into an agreement?
The decision-making process is a long and arduous one requiring thorough research and comparison to determine the best and most trusted contract services for your pharmaceutical company. In-depth research into the history and activities of contract services providers give pharma company owners a well-informed idea of the experience, reliability and capability of service providers to provide the desired end-result consistently. With due research, pharma companies can compare skills and expertise, experience, pricing, and other factors based on their unique needs before establishing a contract agreement. The final choice will determine drug costs, general expenditure, efficiency, etc.
References
https://www.msh.org/sites/msh.org/files/mds3-ch39-contracting-mar2012.pdf
https://www.thomasnet.com/articles/top-suppliers/contract-pharmaceutical-manufacturers-companies/
https://astrixinc.com/best-hiring-practices-for-contract-manufacturing-organizations/
https://astrixinc.com/best-hiring-practices-for-contract-manufacturing-organizations/
References
Upcoming Events
-
CPHI North America 2024
Pennsylvania Convention Center, Philadelphia
07 May 2024 - 09 May 2024 -
-
CPHI South East Asia 2024
Queen Sirikit National Convention Center, Bangkok, Thailand
10 Jul 2024 - 12 Jul 2024
Pharmaceutical Industry Webinars
-
Webinar Exploring Three Technological Trends in the Future of Pharmaceutical Manufacturing
-
23rd May 2024
-
4pm CET / 10am EST
-
-
Webinar Achieving Manufacturing Excellence Through Digital Transformation
-
16th April 2024
-
4pm CET / 10am EST
-
-
Webinar Made in Africa: What’s Driving Pharma Manufacturing
-
28th March 2024
-
4pm CET / 10am EST
-
-
Webinar Case Study: Risk Management for Annex 1 Sterile Production EMS
-
28th February 2024
-
4pm CET / 10am EST
-
-
Webinar Innovative Strategies for B2B Pharma Marketeers: Driving Value through Content
-
20th February 2024
-
4pm CET / 10am EST
-
-
Webinar Revolutionizing Pharma: Data and AI Unleashed
-
18th January 2024
-
4pm CET / 10am EST
-
-
Webinar Optimal Temperature: Elevating Biologics Cold Chain Excellence
-
16th January 2024
-
4pm CET / 10am EST
-
-
Webinar Market Outlook – The Biggest Pharma Trends of 2024
-
12th December 2023
-
4pm CET / 10am EST
-
-
Webinar The Next Frontier – Emerging Opportunities in the LATAM Pharma Market
-
21st November 2023
-
4pm CET / 10am EST
-
-
Webinar Vistamaxx™ MED - imagine the possibilities for healthcare product performance
-
10th October, 2023
-
4pm CET / 10am EST
-
-
Webinar Co-processing: A Multifaceted Approach for Enhancing Density & Powder Flow
-
19th September 2023
-
4pm CET / 10am EST
-
-
Webinar The Outlook for Cell & Gene Therapy Manufacturing
-
28th June 2023
-
4pm CET / 10am EST
-
-
Webinar Contract Packaging Outlook: Growth Trends within the Commercial Packaging Sector
-
25th May 2023
-
4pm CET / 10am EST
-
-
Webinar The HPAPI Boom: How Pharma is Rising to the Manufacturing Challenge
-
23rd February 2023
-
4pm CET / 10am EST
-
Position your company at the heart of the global Pharma industry with a CPHI Online membership
-
Your products and solutions visible to thousands of visitors within the largest Pharma marketplace
-
Generate high-quality, engaged leads for your business, all year round
-
Promote your business as the industry’s thought-leader by hosting your reports, brochures and videos within your profile
-
Your company’s profile boosted at all participating CPHI events
-
An easy-to-use platform with a detailed dashboard showing your leads and performance



































-comp241600.jpg)






















































.png)

.png)



.png)





.png)
.png)
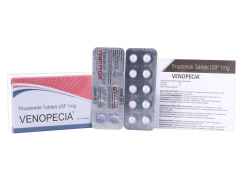


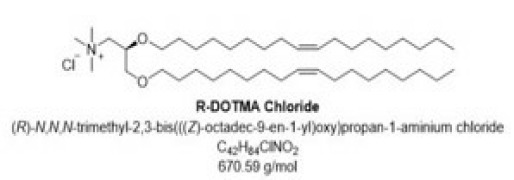




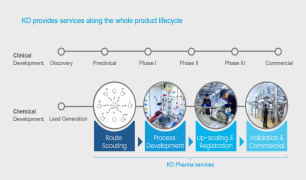












































































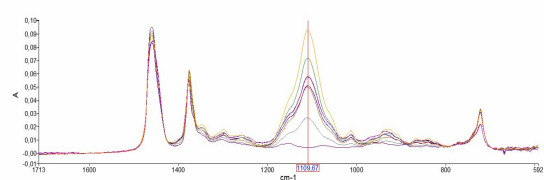


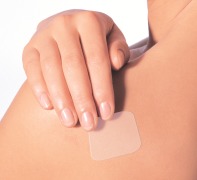




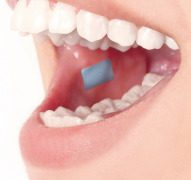




















.png)


.png)



.jpg)